Scientists Warn That COVID-19 Can Cause Idiopathic Multicentric Castleman Disease with TAFRO Syndrome
Nikhil Prasad Fact checked by:Thailand Medical News Team Apr 09, 2026 1 month, 3 weeks, 5 days, 11 hours ago
Medical News: In the wake of the global COVID-19 pandemic, clinicians worldwide have documented a growing number of once rare post-infectious complications that challenge diagnostic frameworks. Among these is the sudden onset of idiopathic multicentric Castleman disease (iMCD) accompanied by TAFRO syndrome, a severe inflammatory lymphoproliferative disorder. Emerging case reports suggest that SARS-CoV-2 infection may act as a potent trigger in susceptible individuals, unleashing a cytokine-driven storm that mimics or precipitates this once-obscure condition.
 Cases of Idiopathic Multicentric Castleman Disease with TAFRO Syndrome that were once rare are rising in many that were exposed to COVID-19
Cases of Idiopathic Multicentric Castleman Disease with TAFRO Syndrome that were once rare are rising in many that were exposed to COVID-19
This Thailand
Medical News report examines the clinical profile of iMCD-TAFRO, its hallmark symptoms, and the plausible mechanisms by which COVID-19 may precipitate its development, drawing on recent peer-reviewed case studies and mechanistic insights.
What is Idiopathic Multicentric Castleman Disease (iMCD) with TAFRO Syndrome?
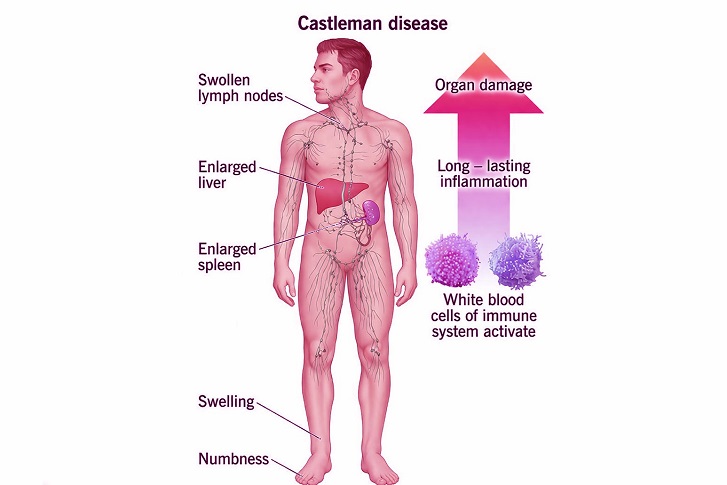
Idiopathic multicentric Castleman disease (iMCD) is a rare, non-malignant lymphoproliferative disorder characterized by widespread enlargement of lymph nodes and systemic hyperinflammation without evidence of human herpesvirus-8 (HHV-8) infection or other identifiable causes. Unlike unicentric Castleman disease, which affects a single lymph node region and is often curable by surgical resection, iMCD involves multiple nodal sites and produces profound constitutional symptoms driven by excessive cytokine release, particularly interleukin-6 (IL-6).
TAFRO syndrome represents the most aggressive clinical subtype of iMCD. The acronym stands for Thrombocytopenia (low platelet count), Anasarca (generalized severe oedema with pleural effusions and ascites), Fever (persistent or recurrent), Reticulin fibrosis (bone marrow fibrosis) and/or Renal dysfunction, and Organomegaly (enlargement of the liver, spleen, and lymph nodes).
Patients typically exhibit normal or only mildly elevated immunoglobulin levels, distinguishing TAFRO from the more common iMCD-not otherwise specified (iMCD-NOS) variant, which often features thrombocytosis and polyclonal hypergammaglobulinaemia. Histologically, lymph nodes show hypervascular or plasmacytic features with atrophic germinal centres and prominent high-endothelial venules. Bone marrow biopsy frequently reveals reticulin fibrosis and atypical megakaryocytes.
The condition once exceedingly rare, with limited epidemiological data globally is raising concerns in the Post-COVID era. Studies in Asian populations, including China and Thailand, indicate a notably high proportion of TAFRO cases among adult Castleman disease patients.
Clinical Symptoms and Disease Course
Onset is often acute or subacute. Core symptoms include high spiking fevers, profound fatigue, weight loss, and night sweats. Generalized anasarca leads to marked swelling, pleural and pericardial effusions, and ascites, frequently causing respiratory distress. Thrombocytopenia predispos
es patients to bleeding tendencies, while renal impairment can progress to acute kidney injury requiring dialysis. Organomegaly is evident on imaging as hepatosplenomegaly and diffuse lymphadenopathy. Laboratory hallmarks comprise elevated C-reactive protein (CRP), hypoalbuminaemia, anaemia, and markedly raised IL-6 levels. Without prompt intervention, multi-organ failure can ensue, with historical mortality rates exceeding 50% in severe untreated cases.
How COVID-19 Possibly Triggers iMCD-TAFRO
SARS-CoV-2 infection is known to provoke profound immune dysregulation, including a hyperinflammatory “cytokine storm” that overlaps mechanistically with the pathophysiology of iMCD-TAFRO. The virus induces massive release of pro-inflammatory cytokines such as IL-6, which drives the lymphoproliferation, vascular permeability, and systemic inflammation central to TAFRO. Post-COVID endothelial damage, autoantibody formation, and persistent immune activation may unmask latent genetic vulnerabilities or trigger de novo autoreactive responses in predisposed individuals.
Several case reports have now documented iMCD-TAFRO emerging weeks to months after confirmed COVID-19 infection. In one notable Japanese case, a 62-year-old woman developed classic TAFRO features shortly after recovering from COVID-19, with lymph node histology confirming iMCD and rapid response to IL-6 blockade.
Broader literature reviews have catalogued additional instances of TAFRO-like syndromes or iMCD flares following SARS-CoV-2, supporting a temporal and biological link. While causality is not definitively proven in every case, the shared cytokine profile and exclusion of alternative triggers strengthen the hypothesis that COVID-19 acts as an environmental precipitant.
Diagnostic Challenges and Management
Diagnosis requires integration of clinical criteria, laboratory findings, imaging, and lymph node or bone marrow histology, with exclusion of malignancies, infections, and autoimmune diseases. The Castleman Disease Collaborative Network provides consensus guidelines emphasizing the TAFRO triad (thrombocytopenia, anasarca, fever/inflammation) plus supportive minor criteria.
First-line therapy typically involves high-dose glucocorticoids. For steroid-refractory cases, IL-6 inhibitors such as siltuximab or tocilizumab have revolutionized outcomes, achieving rapid symptom control and sustained remission in many patients. Rituximab, cyclosporine, or combination chemo-immunotherapy may be added in severe disease. Early recognition is critical, as delays can prove fatal.
Relevance to Global Implications
The medical community has already noted a higher-than-expected incidence of TAFRO features within local Castleman disease cohorts. With millions of COVID-19 cases recorded nationally, clinicians should maintain vigilance for post-viral lymphoproliferative syndromes presenting with unexplained fever, oedema, and cytopenias. Heightened awareness, coupled with access to IL-6 targeted therapies, could improve outcomes in this rare but increasingly recognized entity.
Conclusion
While iMCD-TAFRO were once rare, the accumulating evidence of its emergence following COVID-19 underscores the virus’s capacity to induce long-term immunological sequelae. This Medical News report highlights the need for continued surveillance, multidisciplinary collaboration, and further research into post-infectious triggers of lymphoproliferative disorders. Prompt diagnosis and IL-6-directed therapy offer hope for patients, transforming what was once a frequently fatal condition into a manageable one. As the medical community navigates the long shadow of the pandemic, recognizing such links will be essential to safeguarding public health globally.
References:
https://rs.yiigle.com/cmaid/1673182
https://academic.oup.com/mrcr/article-abstract/9/1/174/7739789
https://linkinghub.elsevier.com/retrieve/pii/S0165247825001567
https://www.mdpi.com/2227-9059/12/6/1287
https://www.mayoclinic.org/diseases-conditions/castleman-disease/symptoms-causes/syc-20543017
For the latest COVID-19 News, keep on logging to Thailand
Medical News
Read Also:
https://www.thailandmedical.news/articles/coronavirus
https://www.thailandmedical.news/articles/long-covid
