COVID-19-News: Spanish Study Shows That Gut Microbiome Composition And Function Play A Role In COVID-19 Progression. Probiotics Help!
COVID-19 - News - Gut Microbiome - Disease Progression Nov 20, 2022 3 years, 6 months, 1 week, 2 days, 20 hours, 31 minutes ago
COVID-19 News: A new study by researchers from the Institute of Agrochemistry and Food Technology, Spanish National Research Council (IATA-CSIC), Valencia – Spain has found that gut microbiome composition and function play a role in COVID-19 disease progression.
Despite SARS-CoV-2 mainly affecting the respiratory system, it has been found that the gastrointestinal tract is also a target.
Worryingly, prolonged gut disorders, in COVID-19 patients, were correlated with decreased richness and diversity of the gut microbiota, immune deregulation and delayed viral clearance.
Though there are no definitive conclusions, ample evidence would suggest that the gut microbiome composition and function play a role in COVID-19 progression.
Microbiome modulation strategies for population stratification and management of COVID-19 infection are under investigation, representing an area of interest in the ongoing pandemic.
The study team in this meta-analysis and review, present the existing data related to the interaction between gut microbes and the host’s immune response to SARS-CoV-2 and discuss the implications for current disease management and readiness to face future pandemics.
The study findings were published in the peer reviewed journal: Critical Reviews in Food Science and Nutrition.
https://www.tandfonline.com/doi/full/10.1080/10408398.2022.2143476
This is the first review study that provides an overview of the role of gut microbiota in shaping the host immune response to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
Already, Thailand Medical News in its previous
COVID-19 News coverages had covered studies that shows that COVID-19 causes dysbiosis or an imbalance between the types of organism present in a person's natural microflora, especially that of the gut that is thought to contribute to a range of conditions of ill health.
https://www.thailandmedical.news/news/new-york-university-study-finds-that-sars-cov-2-infection-causes-gut-microbiome-dysbiosis-that-increases-risk-for-bacterial-infections
https://www.thailandmedical.news/news/post-covid-19-individuals-advised-to-take-probiotics-as-study-shows-sars-cov-2-induced-gut-microbiome-dysbiosis-increases-risk-for-colorectal-cancer
https://www.thailandmedical.news/news/warning-dysbiosis-induced-by-sars-cov-2-infections-presents-risk-of-antibiotic-resistance
<
a href="https://www.thailandmedical.news/news/breaking-autopsy-studies-by-king-s-college-london-reveal-that-covid-19-is-causing-severe-gastrointestinal-damage-especially-on-the-ileal-peyers-s-patc">https://www.thailandmedical.news/news/breaking-autopsy-studies-by-king-s-college-london-reveal-that-covid-19-is-causing-severe-gastrointestinal-damage-especially-on-the-ileal-peyers-s-patc
https://www.thailandmedical.news/news/covid-19-research-disruption-of-gut-microbiome-could-be-also-be-a-contributing-factor-to-disease-severity-in-covid-19-patients
https://www.thailandmedical.news/news/gut-microbiome-composition-possibly-altered-by-sars-cov-2-resulting-in-increase-of-il-18,-a-pro-inflammatory-cytokine-in-covid-19-patients
https://www.thailandmedical.news/news/gut-microbiota-possible-link-with-covid-19-susceptibility
Thailand Medical News would also like to add that many studies have also showed that
probiotics are useful in preventing or treating COVID-19.
https://www.thailandmedical.news/news/covid-19-treatments-probiotics-could-be-used-as-adjuvants-to-treat-covid-19-
https://www.thailandmedical.news/news/emerging-studies-showing-role-of-gut-microbiome-in-covid-19-and-benefits-of-probiotics-for-treatments-and-as-prophylactics-
https://www.thailandmedical.news/news/u-s-clinical-trial-shows-that-probiotics-like-lactobacillus-rhamnosus-gg-can-reduce-covid-19-symptoms-when-taken-as-a-post-exposure-prophylaxis
https://www.thailandmedical.news/news/post-covid-19-individuals-advised-to-take-probiotics-as-study-shows-sars-cov-2-induced-gut-microbiome-dysbiosis-increases-risk-for-colorectal-cancer
https://www.thailandmedical.news/news/breaking-italian-study-shows-that-oral-probiotics-comprising-of-lactic-acid-bacteria-and-bifidobacteria-decreases-risk-of-respiratory-failure-in-covid
https://www.thailandmedical.news/news/study-in-bmj-journal-says-women-do-modestly-benefit-from-multivitamins,-omega-3-supplements-and-probiotics-in-terms-of-decreasing-risk-of-covid-19
https://www.thailandmedical.news/news/gut-microbiota-hong-kong-researchers-say-that-certain-probiotics-could-enhance-the-immune-system-to-fight-against-covid-19
The SARS-CoV-2 coronavirus, the causative pathogen of the COVID-19 pandemic, is an RNA virus that primarily affects the upper and lower respiratory tract. The virus is also known to potentially target the gastrointestinal (GI) tract and impair the composition and diversity of gut microbiota.
The human host gut microbiota is a collection of microbes that naturally live in the GI tract. The GI tract contains trillions of microbes that interact with one another to regulate various physiological processes, including the immune system. Alteration in gut microbiota composition and diversity is called dysbiosis, which impairs immune response and inflammation.
It has been reported that GI symptoms, including nausea, vomiting, and diarrhea, have been observed in 60% of COVID-19 patients.
Furthermore, an impaired gut microbiota has been observed in COVID-19 patients, irrespective of the presence of GI symptoms. Importantly, it has been observed that dysbiosis persists for up to 6 months after the clinical clearance of SARS-CoV-2 from the respiratory tract.
The type of gut microbiota alterations commonly observed in COVID-19 patients include reduced commensal bacterial populations with immunomodulatory functions that help maintain the integrity of GI barrier and immune homeostasis.
Interestingly, a large proportion of COVID-19 patients develop long-term symptoms, which is commonly termed as long-COVID.
Numerous studies have shown that individuals with long-COVID have reduced commensal bacterial population and altered overall gut microbiota. In addition, the reduced bacterial population correlates with increased serum concentration of proinflammatory mediators in these patients.
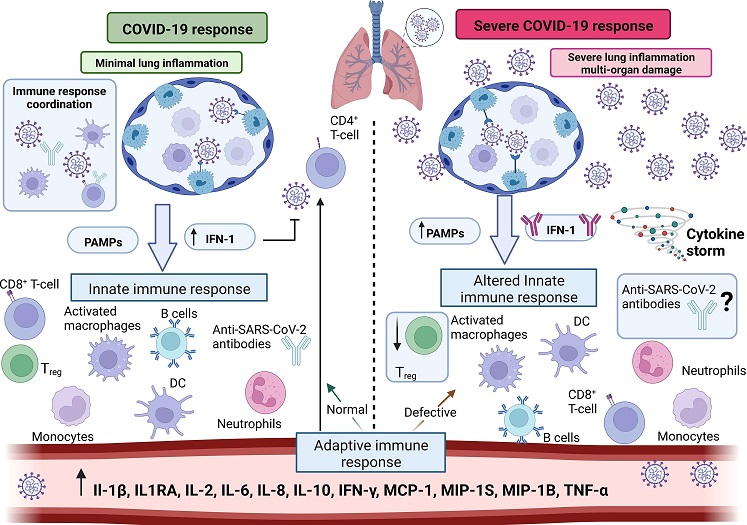 Immune response against SARS-CoV-2 infection. Left: Normal response against COVID-19 infection, with minimal lung inflammation. Right: Defective immune response against severe COVID-19 infection. The cytokine storm might lead to a severe lung inflammation and, eventually, to organ damages.
Immune response against SARS-CoV-2 infection. Left: Normal response against COVID-19 infection, with minimal lung inflammation. Right: Defective immune response against severe COVID-19 infection. The cytokine storm might lead to a severe lung inflammation and, eventually, to organ damages.
Importantly, a reduction in commensal bacterial population is often accompanied by enrichment of pathogenic bacterial population and reduction of gut microbiota diversity.
Research has suggested that these changes in gut microbiota composition and diversity might be associated with increased gut permeability, microbial translocation, hyperinflammation, and poor COVID-19 prognosis.
Aside from the bacterial population, SARS-CoV-2 infection is known to alter the gut fungal population. An enrichment in opportunistic fungal pathogens has been observed in COVID-19 patients. Pathogens such as these are associated with pneumonia and respiratory symptoms and affect the assembly of gut bacteria.
It is already a well-known fact that the spike glycoprotein of SARS-CoV-2 interacts with host cell membrane receptor angiotensin-converting enzyme 2 (ACE2) to initiate the viral entry process. Besides respiratory epithelial cells, ACE2 is expressed at a high level in the stomach, ileum, and colon, highlighting the possibility of direct viral entry into the GI tract.
Significantly, the abundance of certain bacterial species that downregulate ACE2 expression are known to correlate negatively with COVID-19 severity. Patients with diabetes or obesity exhibit a low abundance of these bacteria and are at the highest risk of COVID-19-related mortality.
Also, the gut microbiota is known to stimulate host antiviral immune response by modulating type 1 interferon signaling.
However, in severe COVID-19 patients, an impaired interferon response and a suppressed adaptive immune response are known to cause lung damage. SARS-CoV-2-induced alteration in gut microbiota might be associated with these pathologies.
It is also noted that inflammasome, a cytosolic multiprotein complex, is known to associate with COVID-19 pathogenesis. Recent evidence has suggested that inflammasomes induce the release of neutrophil extracellular traps by neutrophils in severe COVID-19 patients, which in turn is associated with impaired lung function.
The study team says that an altered gut microbiota might contribute to COVID-19 pathogenesis by triggering inflammasome activation. For example, in COVID-19 patients with cardiac disorders, increased activation of inflammasomes and elevated markers of a leaky gut, such as lipopolysaccharide-binding protein, have been observed.
It was also noted that the gut microbiota is vital for the regulation of the adaptive immune system. For example, in response to viral infection, the gut microbiota induces activation of B cells and T cells, thus participating in antibody production and virus-specific memory immune cell production.
Interestingly, the administration of specific commensal bacteria has been found to increase neutralizing antibody levels in the blood in response to viral vaccination. Similar effects have been observed in COVID-19 patients.
The human gut microbiota also plays an essential role in regulating lung health. Immune cells are migrated from the gut to the respiratory tract to destroy invading pathogens. This is called the gut–lung immune axis.
However, alteration in gut microbiota composition increases the risk of respiratory diseases, such as asthma.
Interestingly, in COVID-19 patients, opportunistic upper respiratory tract bacteria have been identified in the gut microbiota. Similarly, an imbalance in lung microbiota has been observed in COVID-19 patients with dysbiosis. These observations highlight the occurrence of bidirectional translocation of microbes between the gut and lung.
Considering the significant association between dysbiosis and anti-SARS-CoV-2 immune response, modulation of gut microbiota has been considered a potential therapeutic intervention for COVID-19.
It is believed that transplantation of fecal microbiota, which represents the whole gut microbiota, from a healthy donor into the GI tract of a recipient is considered a potential strategy to treat bacterial infection. This strategy is currently under clinical investigation in COVID-19 patients.
Certain dietary prebiotics are non-digestible fibers used to increase the proportion of commensal bacteria and reduce the proportion of pathogenic bacteria. There is evidence suggesting the health benefits of prebiotics in COVID-19 patients.
On the other hand, probiotics are live organisms with immunomodulatory effects. In addition, peptides produced by probiotics have shown ACE2 inhibitory effects. Hence, probiotics are considered a potential adjuvant strategy in treating COVID-19 patients.
The study team concluded, “Evidence suggests a key role of the gut commensal microbes in activating the host immune system locally and systemically helping fight viral infections and specifically COVID-19. Similarly, the gut microbiome of the individual might act as an adjuvant of COVID-19 vaccines. Discovery of the precise gut microbiome signatures of subjects with different susceptibility to the infection and response to COVID-19 vaccines is critical to progress in the identification of vulnerable subjects, pathogenic mechanisms and precision anti-viral strategies. Preliminary studies suggest that the oral administration of dietary supplements targeting the gut could help reduce infection severity and viral loads, and aid in the support of COVID-19 outpatients in a cost-effective manner. Although more robust evidence is needed, further research in this direction could positively impact the management of SARS-CoV-2 infection and its long-term consequences, and improve our readiness to face future public health crises.”
For the latest
COVID-19 News, keep on logging to Thailand Medical News.

