BREAKING! COVID-19 News: Wisconsin Study Shows That SARS-CoV-2 Causes Cardiac Fibrosis! Many Could Die Suddenly!
COVID-19-News -SARS-CoV-2 Causes Cardiac Fibrosis Feb 18, 2023 3 years, 2 months, 2 weeks, 2 days, 14 hours, 30 minutes ago
COVID-19 News: It is already known that exposure to the SARS-CoV-2 virus via infections or to its spike proteins via ‘prophylactic shots’ can induce a variety of cardio and vascular issues. To date, three years into the COVID-19 pandemic the death rates from heart failures are rising exponentially due to a SARS-CoV-2 induced myocarditis, pericarditis, arrythmia, hypoxic injury, stress (takotsubo) cardiomyopathy, ischemic injury caused by cardiac microvascular damage or epicardial coronary artery disease (with plaque rupture or demand ischemia), right heart strain (cor pulmonale) etc.
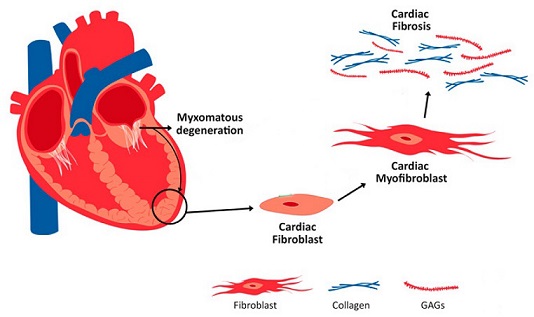
A new study by researchers from Medical College of Wisconsin Milwaukee-USA has found that cardiac fibrosis is yet another causative effect of exposure to the SARS-CoV-2 virus!
Many individuals might not even know or develop symptoms while their heart tissues are undergoing fibrosis and may eventually just suffer a sudden heart failure with a high possibility of a fatal outcome!
Cardiac fibrosis refers to an excess of extracellular matrix (ECM) deposition by cardiac fibroblasts (CFs), which can lead to cardiac dysfunction and heart failure subsequently.
During the early phase of the pandemic, many studies could not claim a direct association between SARS-CoV-2 infection and cardiac fibrosis but it was however determined that existing Cardiac fibrosis conditions were a risk factor for severe COVID-19.
https://www.frontiersin.org/articles/10.3389/fimmu.2021.740260/full
Some past studies and
COVID-19 News coverages did however indicate that SARS-CoV-2 infection could cause cardiac fibrosis or myocardial fibrosis even in non-hospitalized individuals!
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8789662/
https://www.sciencedirect.com/science/article/pii/S2213007121000289
https://www.researchsquare.com/article/rs-105963/v1
https://onlinelibrary.wiley.com/doi/10.1002/ccr3.3972
A recent study by American and Chinese researchers showed that the SARS-CoV-2 spike protein could induce long-term transcriptional suppression of mitochondria metabolic genes and cause cardiac fibrosis.
https://www.biorxiv.org/content/10.1101/2023.01.05.522853v1
Cardiac fibrosis complicates SARS-CoV-2 infections and has been linked to arrhythmic complications in survivors.
The study team sought evidence of increased HSP47 (heat shock protein 47), a stress-inducible chaperone protein that regulates biosynthesis and secretion of procollagen in heart tissue, with the goal of elucidating molecular mechanisms underlying cardiac fibrosis in sub
jects with this viral infection.
Utilizing human autopsy tissue, immunofluorescence, and immunohistochemistry, the study team quantified Hsp47+ cells and collagen α 1(l) in hearts from individuals with SARS-CoV-2 infections.
Since macrophages are also linked to inflammation, the study team measured CD163+ cells in the same tissues.
The study team observed irregular groups of spindle-shaped HSP47+ and CD163+ cells as well as increased collagen α 1(I) deposition, each proximate to one another in "hot spots" of ≈40% of hearts after SARS-CoV-2 infection (HSP47+ P<0.05 versus non-fibrotics and P<0.001 versus controls).
Because HSP47+ cells are consistent with myofibroblasts, subjects with hot spots are termed "profibrotic." The remaining 60% of subjects dying with COVID-19 without hot spots are referred to as "nonfibrotic." No control subject exhibited hot spots.
The study findings showed that colocalization of myofibroblasts, M2(CD163+) macrophages, and collagen α 1(l) may be the first evidence of a COVID-19-related "profibrotic phenotype" in human hearts in situ. The potential public health and diagnostic implications of these observations require follow-up to further define mechanisms of viral-mediated cardiac fibrosis.
The study findings were published in the peer reviewed Journal of the American Heart Association.
https://www.ahajournals.org/doi/full/10.1161/JAHA.122.027990
The study identified increased HSP47+ and CD163+ cells and increased collagen I deposition in hot spots within hearts after SARS‐CoV‐2 infection.
These changes may be the first evidence of a COVID‐19–related profibrotic phenotype in human hearts in situ.
Importantly, profibrotic hot spots could represent paracrine‐mediated effector cell (myofibroblast/M‐CSF and macrophage/PDGF) cross talk and acute fibrogenesis (collagen α 1[I]) within the human myocardium, as proposed in a previous study.
https://pubmed.ncbi.nlm.nih.gov/32058955/
These profibrotic hot spots might also set the stage for enhanced arrhythmogenesis, even sudden cardiac death.
Hence, COVID‐19–induced cardiac fibrosis may have critical long‐term public health implications for health care systems and policy makers.
The study team suggest investigations into mechanisms of viral‐induced integrated stress response pathways, myofibroblast lineage transitional states, and paracrine mediators are priorities. One immediate implication is development of noninvasive approaches (ie, plasma biomarkers and noninvasive imaging) to identify antemortem profibrotic phenotypes. These data may also accelerate clinical trials of antifibrotic therapies and could influence policies addressing cardiovascular health disparities among vulnerable populations.
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
Read Also:
https://www.thailandmedical.news/news/breaking-covid-19-latest-sars-cov-2-induces-immunoparalysis-of-human-host,-infects-monocytes,-macrophages-and-can-cause-fibrosis-in-post-covid-19
