BREAKING NEWS! Integrin Binding Motifs In SARS-CoV-2 Spike Protein And MHC Class Alleles Could Be Playing A Key Role In COVID-19 Pathogenesis!
Thailand Medical News Aug 08, 2023 2 years, 9 months, 1 week, 6 days, 7 hours, 4 minutes ago
COVID-19 News: The world has been grappling with the unprecedented COVID-19 pandemic caused by the SARS-CoV-2 virus, a global health crisis reminiscent of the H1N1 influenza outbreak of 1918. Latest
COVID-19 News reports show that the COVID-19 crisis is far from over, with cases rising literally in every geolocation in the world driven by the new SARS-CoV-2 EG.5.2 sub-lineages and other XBB spawns.
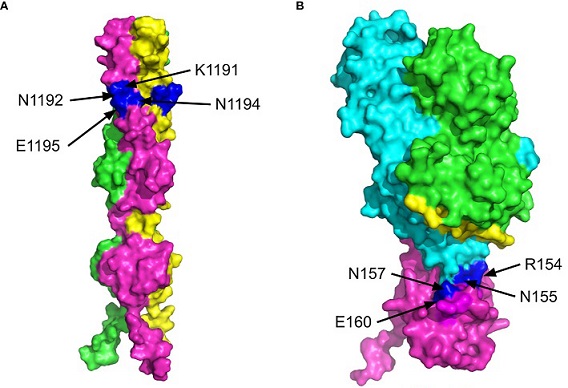 The 3D structures of the C-terminus of the SARS CoV-2 spike protein (P0DTC2, SPIKE_SARS2) and Epstein-Barr Virus (EBV) gp42 protein bound to the MHC class II Receptors HLA-DRB1 (P01911, DRB1_HUMAN) and HLA-DRB3 (P79483, DRB3_HUMAN) β-chains. The surface views are shown with the key residues labelled and highlighted in blue. (A) MultiFOLD model of the SARS CoV-2 spike (P0DTC2 - SPIKE_SARS2) homotrimer complex C-terminus region from residue 1194 onward (plDDT=0.751, pTM=0.599). (B) Crystal structure of EVB gp42 (magenta) bound to the HLA-DR1 complex (PDB ID - 1KG0).
The 3D structures of the C-terminus of the SARS CoV-2 spike protein (P0DTC2, SPIKE_SARS2) and Epstein-Barr Virus (EBV) gp42 protein bound to the MHC class II Receptors HLA-DRB1 (P01911, DRB1_HUMAN) and HLA-DRB3 (P79483, DRB3_HUMAN) β-chains. The surface views are shown with the key residues labelled and highlighted in blue. (A) MultiFOLD model of the SARS CoV-2 spike (P0DTC2 - SPIKE_SARS2) homotrimer complex C-terminus region from residue 1194 onward (plDDT=0.751, pTM=0.599). (B) Crystal structure of EVB gp42 (magenta) bound to the HLA-DR1 complex (PDB ID - 1KG0).
Despite extensive research into the various pathological processes triggered by SARS-CoV-2 infection, such as coagulation irregularities, hyperinflammation, and autoimmune responses, many factors and mechanisms underlying these events remain shrouded in mystery.
Recent scientific investigations have delved into a potentially pivotal role played by integrin binding motifs within the SARS-CoV-2 spike protein and MHC class alleles in the development and progression of COVID-19.
Integrins, essential transmembrane glycoproteins comprising α and β subunits, are renowned for their ability to bind to extracellular matrix molecules, cell-surface components, and soluble ligands.
These versatile receptors facilitate numerous cellular processes, including cell adhesion, migration, immune responses, angiogenesis, and even cancer development. Among these, the RGD (arginine-glycine-aspartic acid) amino acid sequence stands out as a central motif responsible for integrin binding. Other tripeptide motifs, such as KGD, LDV, ECD, and MVD, have also been identified for their roles in integrin interactions.
The significance of integrins in virus infections has been increasingly recognized, with a wide range of viruses exploiting these receptors for cellular entry and infection. Notable examples include coxsackie adenovirus, human cytomegalovirus, foot-and-mouth disease virus, Kaposi’s sarcoma-associated herpes virus, human papillomavirus, and even SARS-CoV-2. In the case of the latter, integrins serve as co-receptors alongside other cellular receptors, potentially augmenting viral infectivity and broadening the spectrum of susceptible cell types and hosts.
Key integrin categories have been delineated based on their specific ligand interactions. These include collagen-binding integrins, RGD motif-recognizing integrins, laminin-binding integrins, and leukocyte-binding integrins. These receptors play indispensable roles in diverse physiological and pathological processes, making them attractive targets for therapeutic interventions.
Within the context of SARS-CoV-2, researchers have homed in on three integrin bindin
g motifs within the spike protein: RGD, LDI, and ECD. These motifs have been hypothesized to bind to integrins present on host platelets and endothelial cells during infection, potentially contributing to the dysregulated coagulation, inflammation, and autoimmune complications observed in COVID-19.
Comprehensive analyses, including the use of advanced protein structure prediction models like MultiFOLD, have shed light on the accessibility of these motifs on the S protein surface. Intriguingly, similarities have been drawn between certain amino acids within the S protein and those pivotal for the binding of Epstein-Barr virus (EBV) gp42 to HLA-DRB1 molecules, a mechanism employed by EBV to evade immune responses.
Remarkably, these analogous amino acids appear to be accessible in the S protein HR2 region, prompting speculation that SARS-CoV-2 could exploit a similar immune evasion mechanism as EBV. However, these conjectures warrant validation through extensive experimental research. If substantiated, these findings could pave the way for novel therapeutic approaches and medications, as well as the development of safer and more effective S protein-based vaccines not only for SARS-CoV-2 but also for potential future coronaviruses.
Recent variant strains of SARS-CoV-2, such as the BA.2, BA.4, and BA.5 variants with the D405N mutation, have provided intriguing insights. These variants, exhibiting milder symptoms, have been linked to a compromised interaction between the S protein's RGD motif and the avβ3 integrin on host cells, reinforcing the pivotal role of integrin binding in COVID-19 pathogenesis.
Moreover, investigations into the prediction of HLA class II binding peptides within the S protein RBD domain have raised the possibility of an antibody response targeting not only the S protein RGD motif but also related motifs within host plasma proteins involved in coagulation processes. This potential cross-reactivity could contribute to the deregulation of these vital physiological functions, further amplifying the complexity of COVID-19.
In light of these emerging insights, further research is imperative to validate and expand upon these hypotheses. Such efforts hold promise not only for a deeper understanding of COVID-19 pathogenesis but also for the potential development of targeted interventions to mitigate the devastating consequences of this global pandemic.
As the scientific community grapples with the ongoing challenges posed by SARS-CoV-2 and prepares for future viral threats, a comprehensive exploration of integrin binding motifs and their multifaceted roles may unlock the doors to innovative treatments, safer vaccines, and a more resilient global health landscape. Additionally, a heightened focus on potential post-vaccination autoimmune manifestations could enhance the safety and efficacy of vaccines against SARS-CoV-2 and other infectious agents, ensuring a healthier and more secure future for all.
The study findings were published in the peer reviewed journal: Frontiers in Immunology.
https://www.frontiersin.org/articles/10.3389/fimmu.2023.1177691/full
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
