Study Discovers That Proteins c-Src Kinase And ZNRF1 Controls TLR3 Signaling And Interferon Production In Viral Infections Including COVID-19
COVID-19-News - c-Src Kinase - ZNRF1 - TLR3 Signaling - Interferon Jun 09, 2023 2 years, 11 months, 3 weeks, 5 days, 23 hours, 49 minutes ago
New Breakthrough in Antiviral Immunity: The Key to Protecting the Lungs During COVID-19 Infections.
COVID-19 News: A recent study conducted by Taiwanese researchers from the National Taiwan University, Kaohsiung Veterans General Hospital, National Health Research Institutes and Institute of Molecular Biology-Taiwan has revealed a crucial mechanism that controls the immune response against viral infections in the lungs. The study findings provide exciting insights into the body's defense system and offers potential strategies to prevent lung damage caused by viral infections including infections by the SARS-CoV-2 coronavirus that causes the COVID-19 disease.
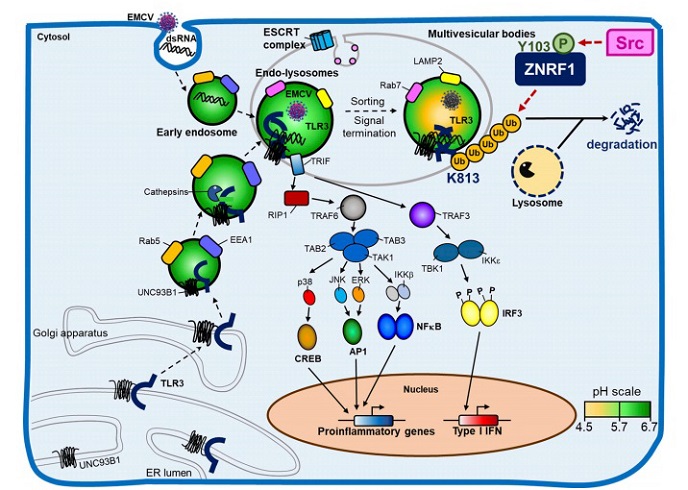 Graphical model summarizing regulation of TLR3 trafficking and type I IFN production by ZNRF1. Upon TLR3 activation by poly(I:C) or an invading RNA virus, ZNRF1 is activated by c-Src kinase by phosphorylation at tyrosine 103. ZNRF1 then catalyzes K63-linked polyubiquitination of TLR3 at lysine 813, which subsequently promotes TLR3 trafficking from endolysosomes to MVBs/lysosomes for degradation, thereby terminating TLR3-triggered innate immune responses. When ZNRF1 is depleted, TLR3 ubiquitination is decreased, thereby increasing endosomal accumulation of TLR3, which leads to prolonged activation of its downstream signaling and enhanced production of type I IFNs and inflammatory mediators.
Graphical model summarizing regulation of TLR3 trafficking and type I IFN production by ZNRF1. Upon TLR3 activation by poly(I:C) or an invading RNA virus, ZNRF1 is activated by c-Src kinase by phosphorylation at tyrosine 103. ZNRF1 then catalyzes K63-linked polyubiquitination of TLR3 at lysine 813, which subsequently promotes TLR3 trafficking from endolysosomes to MVBs/lysosomes for degradation, thereby terminating TLR3-triggered innate immune responses. When ZNRF1 is depleted, TLR3 ubiquitination is decreased, thereby increasing endosomal accumulation of TLR3, which leads to prolonged activation of its downstream signaling and enhanced production of type I IFNs and inflammatory mediators.
The human body relies on a powerful antiviral cytokine called type I interferon to combat invading viruses. However, prolonged interferon production can have detrimental effects on the host. Understanding the regulation of this process is essential for maintaining a balanced immune response. The study team focused on a specific immune receptor called TLR3, which plays a crucial role in antiviral immunity.
TLR3 is responsible for recognizing viral RNA and initiating an immune response. The localization of TLR3 within cells determines the production of type I interferons. However, the mechanism that terminates TLR3 signaling and interferon production has remained a mystery.
This new study uncovered a previously unknown axis involving two proteins, c-Src kinase and ZNRF1, that controls TLR3 trafficking and the termination of its signaling.
The study team discovered that when TLR3 engages with viral RNA, it activates c-Src kinase, which, in turn, phosphorylates ZNRF1. This phosphorylation event leads to the ubiquitination of TLR3 and promotes its trafficking to cellular compartments called multivesicular bodies/lysosomes, where it is degraded. This degradation effectively terminates TLR3 signaling and prevents excessive interferon production.
To investigate the role of ZNRF1 in antiviral immunity, the study team used ZNRF1-deficient cells and observed their response to viral infections.
Surprisingly, these cells showed enhanced production of type I interferons and increased resistance to infection by encephalomyocarditis virus (EMCV) and SARS-CoV-2, the virus responsible for COVID-19. These findings suggest that the c-Src–ZNRF1 axis acts as a negative fee
dback mechanism to control TLR3 signaling and limit viral replication.
However, the study also uncovered an unexpected consequence of ZNRF1 deficiency. While the ZNRF1-deficient mice exhibited enhanced antiviral immunity, they were more susceptible to respiratory bacterial superinfections due to exacerbated lung barrier damage. This highlights the delicate balance required in the immune response and underscores the importance of properly regulating interferon production.
The study findings of this study have significant implications for our understanding of antiviral immunity and the development of therapies for respiratory viral infections.
By targeting the Src–ZNRF1 axis, it may be possible to modulate interferon responses and prevent lung damage caused by viral infections. This could have important implications for the treatment of diseases like COVID-19, where an overactive immune response can contribute to severe lung complications as covered in various case reports, studies and
COVID-19 News coverages.
The study team also highlighted the clinical relevance of their findings. Recent studies have shown that patients with genetic mutations affecting TLR3 signaling experience more severe outcomes from COVID-19. Additionally, the expression of ZNRF1 in peripheral blood mononuclear cells was found to be positively correlated with the severity of the disease. These observations further emphasize the importance of TLR3 and the Src–ZNRF1 axis in controlling viral infections.
In conclusion, the discovery of the Src–ZNRF1 axis and its role in regulating TLR3 signaling represents a significant breakthrough in our understanding of antiviral immunity. This study provides valuable insights into the mechanisms that control interferon production and highlights the delicate balance needed to protect the lungs from viral infections. Further research in this field may lead to novel therapeutic approaches that can modulate the Src–ZNRF1 axis to enhance antiviral immune responses or suppress excessive inflammation in the lungs.
The study findings were published in the peer reviewed Journal of Experimental Medicine.
https://rupress.org/jem/article/220/8/e20220727/214096/The-Src-ZNRF1-axis-controls-TLR3-trafficking-and
https://rupress.org/jem/article-pdf/220/8/e20220727/1452180/jem_20220727.pdf
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
