Glaucoma News: U.S. Study Finds That Complement System Proteins In Human Aqueous Humor Plays A Role In Primary Open-Angle Glaucoma!
Nikhil Prasad Fact checked by:Thailand Medical News Team Oct 09, 2023 2 years, 7 months, 1 week, 5 days, 15 hours, 29 minutes ago
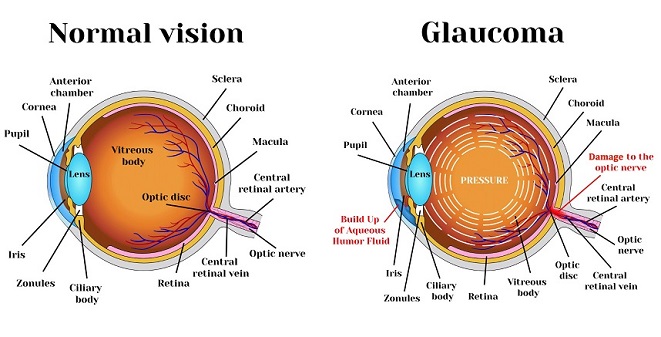
Glaucoma News: Primary open-angle glaucoma (POAG) is a progressive eye disease characterized by optic nerve damage and visual field loss, often leading to irreversible blindness if left untreated. Understanding the intricate mechanisms underlying this condition is crucial for its early diagnosis and effective management. Recent research conducted by the Morsani College of Medicine at the University of South Florida and the Medical College of Georgia at Augusta University in the United States has shed light on the role of complement system proteins in the aqueous humor (AH) and their association with POAG pathogenesis.
Thailand Medical News has started this new daily segment of
Glaucoma News as the current COVID-19 surges are expected to cause millions globally to develop new onset diabetes and also new onset hypertension which are contributory factors to the increasing incidences of glaucoma.
The Complement System and Ocular Immune Privilege
The eye possesses a unique immunological environment known as ocular immune privilege, which is essential for maintaining the delicate balance between immune defense and prevention of harmful inflammation within the eye. This privilege is upheld through various mechanisms, including mucosal immunity, blood-ocular barriers, ocular parenchymal cells, and the secretion of regulatory molecules in the vitreous and aqueous humor. Any disruption in these pathways can disturb ocular immune privilege and contribute to the development of ocular pathophysiological conditions.
The aqueous humor (AH), a clear fluid present in the anterior chamber of the eye, plays a vital role in maintaining intraocular pressure (IOP), removing metabolic waste, and supplying nutrients and oxygen to non-vascular ocular tissues. Moreover, the AH contains a substantial concentration of immunomodulatory molecules, including members of the complement family.
Complement Proteins in Ocular Health
Complement proteins are key players in the body's immune and inflammatory responses, making them essential for immune modulation within the eye. Previous studies have established a connection between alterations in complement proteins within the AH and the pathogenesis of various ocular disorders, including uveitis, macular degeneration, and primary open-angle glaucoma. Additionally, inhibiting components of the complement cascade has shown promise in preventing synaptic degeneration in experimental models of glaucoma.
The Objective of the Study
The primary objective of the study was to investigate the presence of complement proteins in the AH of human subjects and their potential association with the development and progression of primary open-angle glaucoma (POAG). The study involved quantifying the levels of complement proteins in AH samples collected from 258 subjects who had undergone either glaucoma or cataract surgeries. The research also aimed to examine how these proteins might differ based on factors such as sex and race.
Revealing Complement Protein Alterations in POAG
The study successfully identified 32 complement proteins within the AH samples collecte
d from both POAG and cataract patients. Among these proteins, 22 were notably abundant, appearing in more than 50% of the AH samples. Proteomic analysis revealed significant alterations in complement protein levels among POAG patients compared to those with cataracts.
Intriguingly, complement protein C3 was found to be the most abundant in the AH samples, indicating its pivotal role in the ocular environment. However, the levels of C3 exhibited distinct patterns based on race, with an increase observed in African American POAG patients and a decrease in Caucasian POAG patients.
Race-Specific Differences in Complement Protein Profiles
The study also unveiled race-specific differences in complement protein profiles associated with POAG. In the African American population with glaucoma, four complement proteins, including C4A, C4B, F2, and C7, were significantly elevated compared to those with cataracts. These findings suggest a potential involvement of the complement cascade in the accelerated onset and progression of glaucoma within this demographic group.
In contrast, in the Caucasian cohort, eight complement proteins, namely C6, C8G, CFH, C3, CFHR1, CFI, CLU, and SERPING1, were significantly downregulated in POAG subjects. These differences in complement protein profiles based on race highlight the complexity of glaucoma's underlying pathogenesis and immune response.
Sex-Specific Differences in Complement Protein Profiles
Sex-specific differences in complement protein profiles were also identified in the study. In males with POAG, the levels of CLU, C6, and CFH proteins were significantly decreased compared to the controls. Reduced levels of CLU may render retinal neurons more vulnerable to complement-mediated inflammation and cell damage. Additionally, decreased CFH levels may suggest compromised regulatory control over the alternative complement pathway in males with POAG.
In females with POAG, the AH exhibited increased levels of complement proteins C4B and F2, along with a decrease in C8G. These findings suggest potential implications for inflammation and blood flow changes in the eye's vasculature, contributing to retinal cell damage in female POAG patients.
Limitations and Future Directions
While this study provides valuable insights into complement proteins in the AH and their relation to POAG, there are several limitations to consider. The study's statistical power was limited, preventing a detailed comparison between African American and Caucasian males/females with POAG and cataracts. Additionally, the influence of anti-glaucoma therapy on AH complement profiles requires further investigation.
Furthermore, future research should focus on the interplay between anti-glaucoma therapies and complement protein expression within the AH. Moreover, studying the complement proteins in ocular tissues, such as the optic nerve and retina, could provide a more comprehensive understanding of the molecular mechanisms regulating POAG.
Conclusion
In conclusion, this groundbreaking study utilizes advanced liquid chromatography–tandem mass spectrometry (LC-MS/MS) analyses to uncover the role of complement system proteins in the aqueous humor and their connection to primary open-angle glaucoma. The findings not only expand our understanding of complement proteins in the AH but also highlight their differential expression within different racial, sexual, and glaucoma subgroups.
While further research is needed to elucidate the precise contribution of the complement system to glaucoma pathology, these insights have the potential to advance personalized treatment strategies for glaucoma management. By targeting complement proteins in the ocular environment, future therapies may offer improved outcomes for individuals with primary open-angle glaucoma, ultimately preserving their vision and quality of life.
The study findings were published in the peer reviewed Journal of Personalized Medicine.
https://www.mdpi.com/2075-4426/13/9/1400
For the latest
Glaucoma News, keep on logging to Thailand Medical News.
