Cancer News: Swiss Study Finds That Arginine Promotes Tumor Growth In Liver Cancer Via Metabolic Reprogramming!
Nikhil Prasad Fact checked by:Thailand Medical News Team Oct 07, 2023 2 years, 7 months, 3 weeks, 4 days, 11 hours, 39 minutes ago
Cancer News: Cancer is a complex and devastating disease that has puzzled scientists for decades. Traditionally, it has been viewed primarily as a disorder of uncontrolled cell proliferation. However, recent research has unveiled a deeper understanding of cancer as a metabolic disease. In other words, cancer occurs when cells undergo metabolic rewiring to fuel their uncontrolled growth. This metabolic shift has opened up new avenues for cancer research and treatment.
Liver cancer, in particular, presents a formidable challenge in the medical field. The liver is a vital organ responsible for various crucial functions, including metabolizing nutrients, storing energy, regulating blood sugar levels, and detoxifying the body. Liver cancer is a lethal disease, often linked to risk factors such as obesity, excessive alcohol consumption, and hepatitis C infection. Early diagnosis and effective therapeutic strategies are imperative for improving the prognosis of liver cancer patients.
 Arginine: The Hidden Promoter of Liver Cancer
Arginine: The Hidden Promoter of Liver Cancer
Researchers at the University of Basel in Switzerland have made a groundbreaking discovery in the realm of liver cancer. They have uncovered a hidden player in the intricate world of cancer metabolism - the amino acid arginine. This revelation has far-reaching implications for our understanding of liver cancer and its potential treatment.
As liver cells transition into cancerous ones, they undergo a remarkable metabolic transformation. Their metabolism is rewired to sustain rapid growth, marked by increased glucose consumption and enhanced nutrient uptake. Dr Dirk Mossmann, the lead author of the study, explained to
Cancer News reporters from TMN, "We investigated liver tumor samples from mice and patients and found elevated levels of arginine, although cancer cells produce less or none of this amino acid. The tumor cells accumulate high levels of arginine by increasing its uptake and suppressing its consumption."
Arginine, a versatile amino acid, plays a pivotal role in this metabolic transformation. While it is traditionally known as a building block for protein synthesis, arginine also acts as a precursor for polyamines, creatine, and nitric oxide. Moreover, it can influence cell growth by activating mTORC1, a key regulator of cell proliferation. Importantly, arginine's impact on metabolism is not limited to mTORC1, suggesting a multifaceted role in cancer progression.
The Role of Arginine in Tumor Growth
High concentrations of arginine have been found to bind to a specific factor within the tumor cells. This binding event triggers a cascade of metabolic reprogramming, fueling the relentless growth of liver cancer. Interestingly, this reprogramming leads the tumor cells to revert back to an undifferentiated embryonic cell state, allowing them to divide indefinitely. The tumor cells also derive another advantage from increased arginine uptake - they evade the immune system's scrutiny.
Dr Mossmann elaborates on this immune escape strategy, "Our immune cells depend on arginine to function properly. Therefore, depleting arginine in the tumor environment helps the tumor cells escape the immune system.&
amp;quot;
Implications for Liver Cancer Diagnosis and Therapy
The discovery of arginine's central role in liver cancer metabolism opens up promising avenues for diagnosis and treatment. Instead of directly depleting arginine, researchers propose targeting the specific arginine-binding factor responsible for metabolic reprogramming. This approach offers several advantages, notably the avoidance of unintended consequences, such as impairing immune cell function, which relies on arginine.
Dr Mossmann highlights this therapeutic potential, stating, "When treating liver tumors with the anticancer drug indisulam, we induce the degradation of this factor and thus prevent metabolic reprogramming. Via this route, one can avoid unwanted side effects of reducing overall arginine levels, like harming immune cells that need arginine to work properly."
Moreover, the metabolic changes associated with increased arginine levels may serve as valuable biomarkers for early cancer detection. Early diagnosis is a critical factor in the successful treatment and improved survival rates of liver cancer patients. Identifying these metabolic changes at an early stage could potentially revolutionize liver cancer screening and prognosis.
Arginine Reprogramming in Liver Cancer: The Molecular Mechanisms
The study provides valuable insights into the molecular mechanisms underlying arginine's role in liver cancer metabolic reprogramming. These findings shed light on the intricate interplay of arginine and RNA-binding motif protein 39 (RBM39) in driving tumorigenicity.
The study observed that, despite the suppression of arginine synthesis genes, arginine levels are elevated in both murine and patient hepatocellular carcinoma (HCC). Tumor cells achieve this by increasing arginine uptake and reducing its conversion into polyamines. The consequences of elevated arginine levels are profound, as they lead to extensive metabolic reprogramming, affecting glucose, amino acid, nucleotide, and fatty acid metabolism.
Mechanistically, arginine binds to RBM39, a critical regulator of metabolic gene expression. RBM39, depending on its interacting partners, functions as a pre-mRNA splicing factor or a transcription coactivator or co-repressor. The study demonstrated that RBM39 primarily controls metabolic gene expression at the transcriptional level. Remarkably, RBM39 promotes the expression of asparagine synthetase (ASNS), which enhances arginine uptake, creating a positive feedback loop to sustain high arginine levels and oncogenic metabolism.
The N-terminal region of RBM39, structurally unresolved and presumed to be disordered, plays a crucial role in arginine binding. This region of RBM39 may be a key regulatory site integrating various inputs, including phosphorylation and arginine binding. Identifying the specific arginine-binding site(s) and elucidating the interaction between RBM39 and transcription regulators remain important areas of future research.
Furthermore, the study reveals that the expression of ASNS and RBM39 is high in developing embryonic liver tissue but low in adult liver tissue. This suggests that the expression of ASNS and RBM39 may mirror an embryonic metabolic state that is reactivated in HCC, supporting the notion that cancer cells exhibit dedifferentiation.
Exploiting Arginine's Role for Cancer Therapy
The newfound understanding of arginine's central role in liver cancer metabolism presents a potential avenue for cancer therapy. Traditional approaches have focused on depleting arginine using enzymes that degrade circulating arginine. However, these strategies have shown limited clinical benefit.
An alternative strategy, proposed by the Swiss researchers, involves targeting the cancer-specific arginine-binding factor, RBM39, instead of broadly reducing arginine levels. This approach has the advantage of avoiding detrimental effects on immune cells that rely on arginine for their activation. Molecular glues, such as the aryl sulfonamide indisulam, have been developed to specifically target RBM39 for ubiquitination and degradation. Preliminary findings suggest that these compounds mimic the effect of arginine depletion and may hold promise for HCC patients with elevated tumoral arginine levels.
Conclusion
In summary, the Swiss study conducted at the University of Basel has unveiled the critical role of arginine in promoting metabolic reprogramming and tumor growth in liver cancer. This discovery has far-reaching implications for the diagnosis and treatment of liver cancer. By targeting the specific arginine-binding factor, RBM39, rather than broadly depleting arginine, researchers may unlock new therapeutic approaches that offer hope to liver cancer patients.
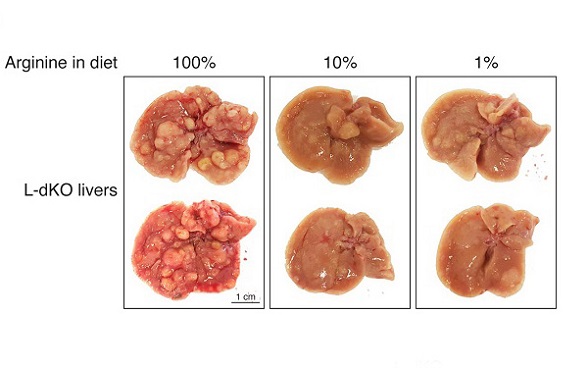 An ex vivo arginine transport assay confirmed that arginine uptake is indeed increased in liver tumors. Thus, L-dKO tumors increase arginine uptake to compensate for downregulation of arginine synthesis. Next, we investigated if high levels of arginine are critical for liver tumor development. L-dKO and control mice were fed diets that contained 10% or 1% of normal levels of arginine found in the standard diet (100% arginine) from 8 to 20 weeks of age. Control mice were not affected by the arginine-restricted diets, as assessed by liver-to-body weight ratio. L-dKO mice displayed characteristic hepatomegaly even upon arginine restriction, again suggesting that the decreased arginine in the diets was not limiting for growth. However, the arginine-restricted diets significantly reduced tumor burden. Thus, high levels of cellular arginine are critical for the development of liver tumors. We note that arginine levels were also higher in presumably non-tumor liver tissue of L-dKO mice compared to normal liver tissue of control mice in normal diet conditions. This is likely due to the fact that at 20 weeks of age, L-dKO mice display multiple macroscopically visible tumors and numerous microscopic tumors in an overall damaged liver, precluding the isolation of “clean” non-tumor tissue. However, arginine levels were significantly lower in non-tumor liver tissue of L-dKO mice upon dietary arginine restriction, but unaffected (i.e., still elevated) in all tumors. This strict correlation of high arginine levels in tumors again suggests that high levels of cellular arginine are critical for tumorigenicity.
An ex vivo arginine transport assay confirmed that arginine uptake is indeed increased in liver tumors. Thus, L-dKO tumors increase arginine uptake to compensate for downregulation of arginine synthesis. Next, we investigated if high levels of arginine are critical for liver tumor development. L-dKO and control mice were fed diets that contained 10% or 1% of normal levels of arginine found in the standard diet (100% arginine) from 8 to 20 weeks of age. Control mice were not affected by the arginine-restricted diets, as assessed by liver-to-body weight ratio. L-dKO mice displayed characteristic hepatomegaly even upon arginine restriction, again suggesting that the decreased arginine in the diets was not limiting for growth. However, the arginine-restricted diets significantly reduced tumor burden. Thus, high levels of cellular arginine are critical for the development of liver tumors. We note that arginine levels were also higher in presumably non-tumor liver tissue of L-dKO mice compared to normal liver tissue of control mice in normal diet conditions. This is likely due to the fact that at 20 weeks of age, L-dKO mice display multiple macroscopically visible tumors and numerous microscopic tumors in an overall damaged liver, precluding the isolation of “clean” non-tumor tissue. However, arginine levels were significantly lower in non-tumor liver tissue of L-dKO mice upon dietary arginine restriction, but unaffected (i.e., still elevated) in all tumors. This strict correlation of high arginine levels in tumors again suggests that high levels of cellular arginine are critical for tumorigenicity.
Furthermore, the study highlights the potential of metabolic changes associated with increased arginine levels as early biomarkers for liver cancer detection. Early diagnosis is paramount in improving the prognosis and survival rates of liver cancer patients. The intricate molecular mechanisms uncovered in this research offer a deeper understanding of liver cancer biology and open up exciting possibilities for future studies and innovative therapies.
The study findings were published in the peer reviewed journal: Cell.
https://www.cell.com/cell/fulltext/S0092-8674(23)01032-2
Thailand
Medical News would like add that with exposure to SARS-CoV-2 also being able to lead to a variety of liver complications including liver cancer, it is advisable that intake of foods containg high levels of arginine also be restricted to moderate levels in these current times.Although arginine is required by immune cells for proper functioning, excess dietary arginine can lead to issues.Further SARS-CoV-2 also leads to reactivation of dormant herpes viruses which also feeds on arginine to aid in their growth and spread!
https://www.thailandmedical.news/news/breaking-sars-cov-2-infection-induces-increase-of-gp73-that-causes-dysglycaemia-increased-gp73-could-also-imply-future-liver-disease-and-liver-cancer
https://www.thailandmedical.news/news/breaking-study-finds-that-upregulated-interleukin-6-drives-development-of-rare-liver-cancers-covid-19-infections-causes-elevated-il-6-levels-in-many
https://www.thailandmedical.news/news/coronavirus-news-german-study-confirms-that-sars-cov-2-coronavirus-can-invade-and-damage-the-liver
https://www.thailandmedical.news/news/covid-19-research-university-of-bristol-researchers-warn-that-liver-complications-are-common-in-covid-19-patients
https://www.thailandmedical.news/news/breaking-covid-19-research-studies-warn-of-high-prevalence-of-liver-injury-in-covid-19-patients
https://www.thailandmedical.news/news/breaking-more-emerging-chinese-research-studies-shows-that-the-sars-cov-2-coronavirus-also-attacks-the-kidneys,-pancreas-and-liver
https://www.thailandmedical.news/news/belgium-researchers-warn-that-more-hospitalized-covid-19-patients-are-manifesting-a-rare-chronic-liver-disease-known-as-sclerosing-cholangitis-could-b
https://www.thailandmedical.news/news/research-news-scripps-scientist-uncover-important-role-of-liver-cells-in-defense-against-certain-viruses-including-potentially-against-sars-cov-2
https://www.thailandmedical.news/news/researchers-alarmingly-find-that-metabolic-associated-fatty-liver-disease-mafld-may-be-a-prevalent-long-covid-manifestation
https://www.thailandmedical.news/news/researchers-alarmingly-find-that-metabolic-associated-fatty-liver-disease-mafld-may-be-a-prevalent-long-covid-manifestation
https://www.thailandmedical.news/news/are-the-new-sars-cov-2-variants-circulating-in-europe-more-neuropathogenic-with-a-modified-pathogenesis-brain-and-liver-issues-on-the-rise
https://www.thailandmedical.news/news/breaking-covid-19-news-sars-cov-2-that-causes-liver-impairment-in-many-also-cause-hypoalbuminemia
https://www.thailandmedical.news/news/breaking-covid-19-news-brazil-study-shows-that-most-long-covid-individuals-continue-to-sustain-damaged-liver-functions-up-to-20-months
https://www.thailandmedical.news/news/covid-19-research-the-detrimental-effects-of-tnf%CE%B1-on-liver-during-sars-cov-2-infection-and-its-impact-on-patient-survival
https://www.thailandmedical.news/news/new-york-clinicians-sound-the-alarm-covid-19-asymptomatic-individuals-can-suffer-acute-liver-injury
For the latest
Cancer News, keep on logging to Thailand Medical News.

