Study Finds That SARS-CoV-2 Infections Can Lead To Lysine Acetylation Of Various Proteins Of The Brain, Possibly Explaining The Various Neurological Issues!
COVID-19 News - SARS-CoV-2 - Lysine Acetylation - Proteins Of The Brain Jun 18, 2023 2 years, 11 months, 3 days, 20 hours, 57 minutes ago
Unraveling the Brain's Acetylome: How SARS-CoV-2 Infections Impact Neurological Manifestations
COVID-19 News: The COVID-19 pandemic has brought the world to its knees, with SARS-CoV-2 wreaking havoc on global health. While respiratory symptoms have been the primary focus, evidence suggests that the virus can also lead to various neurological consequences. Despite the efforts to develop antiviral drugs and vaccines, the battle against the virus persists. Understanding the intricate response of the host's body to SARS-CoV-2 infection is crucial in the pursuit of effective therapies.
A new study conducted by researchers at the Chinese Academy of Medical Sciences and Peking Union Medical College in Beijing-China has shed light on a remarkable finding….the potential involvement of lysine acetylation in the brain's proteins as a consequence of SARS-CoV-2 infections.
A past study covered in a
COVID-19 News report showed that SARS-CoV-2 Nucleocapsid proteins could hijack the human14-3-3 proteins with possible neurological and psychiatric implications. But this new study shows instead that the SARS-CoV-2 virus could be damaging essential brain proteins.
https://www.thailandmedical.news/news/international-study-discovers-that-sars-cov-2-n-proteins-hijacks-human-14-3-3-proteins-with-implications-of-future-rise-in-neurological-and-psychiatri
Unveiling the Acetylome
To unravel the connection between SARS-CoV-2 infection and neurological manifestations, the study team employed a mouse model expressing human ACE2 (K18-hACE2 mice) and utilized advanced LC-MS/MS techniques to comprehensively analyze the acetylomes of brain cortexes in both infected and uninfected mice. This label-free approach enabled the identification of 3829 lysine acetylation (Kac) sites in 1735 proteins, encompassing both histone and nonhistone proteins. Through bioinformatics analysis, the researchers revealed that SARS-CoV-2 infection could potentially impact crucial proteins via acetylation or deacetylation, thereby influencing neurological consequences.
Unraveling the Protein Interactions
In a significant breakthrough, the study identified 26 SARS-CoV-2 proteins that interacted with 61 differentially expressed acetylated proteins. Notably, the researchers discovered acetylated SARS-CoV-2 protein nucleocapsid phosphoprotein, expanding the current knowledge base surrounding the virus's acetylation.
This groundbreaking discovery significantly broadens our understanding of acetylated proteins and provides the first report on the brain cortex's acetylome in the K18-hACE2 mouse model.
These findings lay a solid theoretical foundation for future research into the pathological mechanisms and potential therapies targeting neu
rological consequences following SARS-CoV-2 infection.
Unveiling the Neurological Consequences
Although SARS-CoV-2 primarily affects the respiratory system, it is increasingly evident that the virus can lead to various neurological manifestations. These consequences range from taste and smell disorders to more severe conditions such as encephalitis, meningitis, Parkinson's disease (PD), and Alzheimer's disease (AD). Understanding the intricacies of these neurological consequences is crucial for accurately diagnosing, treating, and predicting the prognosis of COVID-19 patients. The K18-hACE2 mouse model employed in this study provides a valuable platform for studying the pathogenesis of COVID-19 and evaluating potential interventions.
The Intricate Connection: Lysine Acetylation and Neurological Diseases
Lysine acetylation, a protein modification process, plays a vital role in numerous cellular processes and is closely associated with neurological disorders such as AD. Although acetylome analyses have been conducted on various organs and cell lines, investigations into the acetylome of COVID-19 patients or animal models, including K18-hACE2 mice, have been lacking. This study, therefore, aimed to bridge that knowledge gap by exploring the relationship between SARS-CoV-2 infections and acetylation in the brain cortex.
Unprecedented Discoveries
Through their extensive investigation, the study team detected an extraordinary number of acetylated proteins in the mouse brain cortex, significantly expanding the existing acetylome dataset. Furthermore, the study unveiled 473 previously unidentified acetylated proteins. Intriguingly, these proteins were found to be involved in specific pathways, such as the glutamatergic synapse and synaptic vesicle cycle, distinct from acetylated proteins found in other tissues. Noteworthy findings include the upregulation of acetylated microtubule-associated protein tau (Mapt), which plays a pivotal role in PD and AD, and the identification of acetylated sites in α-synuclein (Snca), a protein associated with PD. These discoveries provide further support for the notion that SARS-CoV-2 infection can lead to neurological consequences in the K18-hACE2 mouse model, while also presenting potential targets for therapeutic intervention.
Unraveling the Mechanisms
The process of lysine acetylation involves the transfer of acetyl groups to lysine residues, regulated by lysine acetyltransferases (KATs) and lysine deacetylases (KDACs). The study identified changes in acetylated sites within KATs, with most sites downregulated following SARS-CoV-2 infection. Understanding these mechanisms will be instrumental in deciphering the link between acetylation and the neurological consequences of COVID-19.
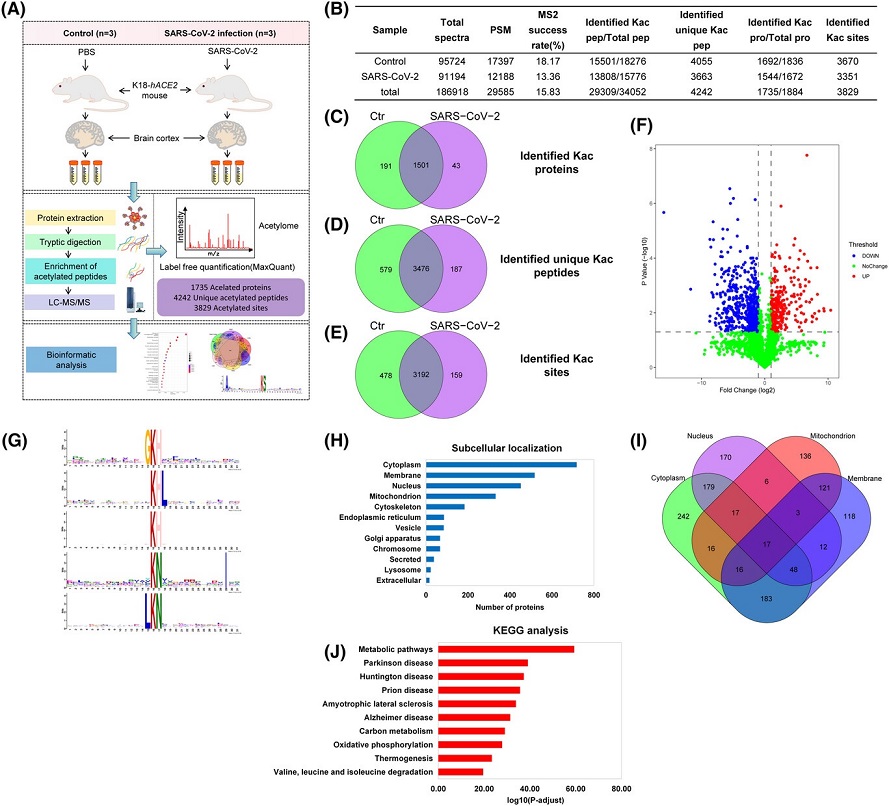 Overview of acetylome analysis in the brain cortex. (A) Workflow for the K18-hACE2 mouse brain cortex acetylated proteome analysis study. (B) Identified spectra, peptides, proteins, and lysine acetylation (Kac) sites for the acetylome. The control and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection were used to analyze the label-free acetylome (each group has three biological replicates.). Venn diagram of (C) identified acetylated proteins, (D) identified unique acetylated peptides, and (E) identified Kac sites in the two groups. (F) Volcano plot displaying the log2 fold changes of quantifiable Class I Kac site intensities in the SARS-CoV-2 infection versus control Kac sites (fold change > 2 or < 0.5, and t-test significance, p value < 0.05). Red dots represent 297 significantly upregulated quantifiable ClassIKac sites in 201 proteins and blue dots represent the 635 downregulated quantifiable ClassIKac sites in 439 proteins. (G) Sequence motifs of acetylated sites ±15 amino acids from the targeted lysine residue. Motifs were compiled from all acetylated peptides containing quantifiable Class I Kac sites. (H) The subcellular localization of Kac proteins. (I) Venn diagram of the overlaps of Kac proteins in cytoplasm, nucleus, mitochondrion, and membrane. (J) Top 10 enriched items in the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis of Kac proteins.
Conclusion
Overview of acetylome analysis in the brain cortex. (A) Workflow for the K18-hACE2 mouse brain cortex acetylated proteome analysis study. (B) Identified spectra, peptides, proteins, and lysine acetylation (Kac) sites for the acetylome. The control and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection were used to analyze the label-free acetylome (each group has three biological replicates.). Venn diagram of (C) identified acetylated proteins, (D) identified unique acetylated peptides, and (E) identified Kac sites in the two groups. (F) Volcano plot displaying the log2 fold changes of quantifiable Class I Kac site intensities in the SARS-CoV-2 infection versus control Kac sites (fold change > 2 or < 0.5, and t-test significance, p value < 0.05). Red dots represent 297 significantly upregulated quantifiable ClassIKac sites in 201 proteins and blue dots represent the 635 downregulated quantifiable ClassIKac sites in 439 proteins. (G) Sequence motifs of acetylated sites ±15 amino acids from the targeted lysine residue. Motifs were compiled from all acetylated peptides containing quantifiable Class I Kac sites. (H) The subcellular localization of Kac proteins. (I) Venn diagram of the overlaps of Kac proteins in cytoplasm, nucleus, mitochondrion, and membrane. (J) Top 10 enriched items in the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis of Kac proteins.
Conclusion
This groundbreaking study has provided comprehensive insights into the brain cortex's acetylome following SARS-CoV-2 infection in the K18-hACE2 mouse model. The identification of numerous acetylated proteins expands our understanding of their potential involvement in neurological consequences, including PD and AD. By shedding light on the intricate relationship between lysine acetylation and SARS-CoV-2 infections, this research opens up new avenues for investigating the pathogenesis of and developing treatments for the neurological manifestations of COVID-19. Future studies will delve deeper into the underlying mechanisms and strive to uncover effective therapeutic interventions to combat these neurological consequences.
The study findings were published in the peer reviewed journal: Proteomics.
https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/10.1002/pmic.202300096
For the latest
COVID-19 News, keep on Logging to Thailand Medical News.
Read Also:
https://www.thailandmedical.news/news/american-scientists-discover-lower-white-matter-cerebral-blood-flow-in-those-who-have-recovered-from-mild-covid-19
https://www.thailandmedical.news/news/breaking-italian-study-discovers-that-brain-fog-in-long-covid-is-due-to-impaired-glutamate-and-gabab-brain-pathways
https://www.thailandmedical.news/news/breaking-news-study-reveals-link-between-sars-cov-2-and-brain-inflammation-a-potential-breakthrough-for-long-covid-treatment
https://www.thailandmedical.news/news/persistent-brain-injury-in-post-covid:-unraveling-the-mysteries-of-neurological-complications
https://www.thailandmedical.news/news/chinese-researchers-warn-that-that-some-post-covid-neurological-issues-could-be-due-to-increasing-incidences-of-autoimmune-encephalitis
https://www.thailandmedical.news/news/breaking-news-u-s-nih-study-finds-that-those-with-long-covid-neurological-symptoms-have-lower-levels-of-cd4-and-cd8-t-cells
https://www.thailandmedical.news/news/deadly-brain-invader-is-covid-19-unleashing-a-prion-plague-on-humanity
https://www.thailandmedical.news/news/sars-cov-2-infections-literally-causes-your-brain-cells-to-die-more-emerging-cases-of-post-covid-acute-necrotizing-encephalopathy-in-young-adults
https://www.thailandmedical.news/news/study-finds-sars-cov-2-spike-protein-accumulating-in-skull-marrow,-brain-meninges-and-brain-parenchyma-while-killing-brain-cells
https://www.thailandmedical.news/news/study-proposes-that-sars-cov-2-induced-neuroinflammation-causes-dopamine-dysfunction-and-transmission-imbalance,-contributing-to-long-covid
https://www.thailandmedical.news/news/must-read-covid-19-news-sars-cov-2-altered-blood-brain-barrier-opens-questions-about-drugs-used-during-treatments-and-their-effects
https://www.thailandmedical.news/news/mice-study-shows-that-sars-cov-2-omicron-variant-can-still-cause-brain-infections-and-lymphoid-depletion-despite-being-milder
https://www.thailandmedical.news/news/mit-study-shows-that-long-covid-neurological-issues-is-due-to-delayed-clearance-of-sar-cov-2-caused-by-immune-imprinting

