COVID-19 News: Study Finds That ORF3a And Spike Proteins Subvert Tetherin - A Host Restriction Factor For SARS-CoV-2, And Promotes Virus Spread
Nikhil Prasad Fact checked by:Thailand Medical News Team Oct 16, 2023 2 years, 6 months, 4 weeks, 1 day, 11 hours, 22 minutes ago
COVID-19 News: In the ongoing battle against COVID-19, researchers from the University of Cambridge in the UK, the US National Institutes of Health in Bethesda, the University of Sheffield in the UK, Imperial College London in the UK, Guy's and St Thomas' NHS Foundation Trust in London, and University College London have made a significant breakthrough. Their study covered in this
COVID-19 News report, sheds light on how the SARS-CoV-2 virus subverts tetherin, a host restriction factor, and promotes virus release. Tetherin is an essential part of the human body's defense against viral infections, and understanding how the virus manipulates it is crucial in the fight against COVID-19.
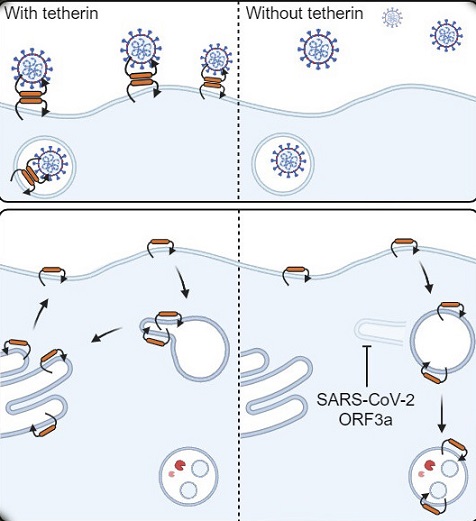 The anti-viral restriction factor Tetherin blocks the egress of SARS-CoV-2. Expression of ORF3a and Spike protein antagonises tetherin to allow SARS-CoV-2 escape.
The anti-viral restriction factor Tetherin blocks the egress of SARS-CoV-2. Expression of ORF3a and Spike protein antagonises tetherin to allow SARS-CoV-2 escape.
-Tetherin loss aids SARS-CoV-2 viral spread.
-SARS-CoV-2 ORF3a alters tetherin localization by impairing retrograde traffic and enhances virus release.
-SARS-CoV-2 Spike causes tetherin downregulation.
Introduction
The COVID-19 pandemic, caused by the SARS-CoV-2 virus, has brought the world to its knees. Understanding the virus's mechanisms of infection and transmission is vital in developing effective countermeasures. SARS-CoV-2, like other coronaviruses, is an enveloped virus, which means it has a lipid envelope derived from the host cell membrane. This envelope shields the viral capsid and helps in evading the host's immune system. However, it also provides an opportunity for the host to integrate antiviral factors within the forming virions.
The entry of SARS-CoV-2 into host cells is mediated by the spike protein's binding to the host receptor angiotensin-converting enzyme-2 (ACE2). Unlike its predecessor, SARS-CoV-1, SARS-CoV-2's spike protein contains a polybasic furin cleavage site, facilitating cleavage into S1 and S2 fragments. This cleavage is essential for the virus's entry and infection. Coronaviruses enter cells through two main mechanisms, direct fusion at the plasma membrane for TMPRSS2-positive cells and endocytosis for TMPRSS2-negative cells. Inside the cell, the viral genome is released and viral replication begins in double-membrane vesicles (DMVs) that act as hubs for viral RNA synthesis. Finally, viral particles are released from infected cells, a process occurring at the ER-to-Golgi intermediate compartments (ERGIC).
Tetherin, also known as Bst2, is an interferon-inducible restriction factor that restricts the egress of various enveloped viruses. It works by linking viral particles to the surface of infected cells, preventing their release and thereby limiting the spread of the virus. Tetherin is a crucial component of the body's innate immune response, and its expression is enhanced by type-I interferon. This protein forms homodimers, linking tetherin molecules on both the virus and the host cell's surface, effectively trapping the viral particles.
Several enveloped viruses, including HIV-1, Kaposi's sarcoma-associated he
rpesvirus, and human coronavirus 229E, are subject to tetherin-dependent restriction. To produce fully released progeny, these viruses have evolved mechanisms to counteract tetherin activity. The specifics of how different viruses downregulate tetherin vary, but they all aim to reduce tetherin levels at the site of viral budding or on the cell's plasma membrane.
Of particular relevance to this study, previous research has shown that some coronaviruses, such as HCoV-229E and SARS-CoV-1, are subject to tetherin restriction. Two SARS-CoV-1 proteins, ORF7a and the spike glycoprotein, have been identified as antagonists of tetherin. The process by which sarbecoviruses, to which SARS-CoV-2 belongs, subvert tetherin's activity remained unclear. This study aims to shed light on how tetherin interacts with SARS-CoV-2 and how the virus manages to counteract it.
Tetherin Downregulation by SARS-CoV-2
The study conducted by these researchers demonstrates the crucial role that tetherin plays in restricting the release of enveloped SARS-CoV-2 virions from infected cells. The researchers used a range of cell types, including primary human airway epithelial (HAE) cells, HeLa cells stably expressing ACE2, A549 cells stably expressing ACE2, and T84 cells expressing endogenous ACE2. They found that SARS-CoV-2 infection resulted in a significant downregulation of tetherin in all cell types, regardless of whether tetherin was constitutively expressed or induced by interferon. This downregulation of tetherin was observed both at the cell surface and within intracellular compartments.
The researchers used immunofluorescence microscopy and electron microscopy to visualize these changes. Infected cells displayed lower levels of tetherin compared to neighboring uninfected cells, indicating that infected cells may produce interferon, which in turn enhances tetherin expression in uninfected neighboring cells.
The study's findings reveal that tetherin plays a significant role in tethering newly formed enveloped SARS-CoV-2 virions to the surface of infected cells, effectively preventing their release. This restriction was observed across various cell types, highlighting the broad antiviral potential of tetherin. Moreover, the study showed that tetherin's downregulation by SARS-CoV-2 is not solely dependent on the virus's ability to antagonize interferon responses, indicating that other mechanisms are at play in reducing tetherin levels.
Tetherin Loss Facilitates SARS-CoV-2 Spread
To further understand the functional role of tetherin in SARS-CoV-2 infection, the researchers conducted growth curve experiments using wild-type HeLa cells and HeLa cells lacking tetherin (Bst2KO). The results demonstrated that intracellular virions accumulated primarily within the first 24 hours post-infection. Wild-type HeLa cells exhibited significantly higher intracellular virus titers compared to Bst2KO cells when infected with a low multiplicity of infection (MOI). This indicates that tetherin restricts the release of virions from infected cells, as virions accumulate in wild-type cells.
However, at higher MOIs, the intracellular viral titers were similar between wild-type and Bst2KO cells, while Bst2KO cells consistently released more virions, indicating that the inability to tether nascent virions resulted in higher virion release. This finding confirms that tetherin functions as a restriction factor against SARS-CoV-2, limiting viral egress from infected cells.
In essence, tetherin acts as a barrier to the release of SARS-CoV-2, and its loss enhances the virus's ability to spread. This is a significant finding that provides insights into the mechanisms underlying the spread of the virus in the human body.
SARS-CoV-2 Proteins Responsible for Tetherin Downregulation
The researchers were interested in identifying which specific SARS-CoV-2 proteins are responsible for downregulating tetherin. To address this question, they conducted a screening of various SARS-CoV-2 open reading frames (ORFs) to identify potential tetherin antagonists. They transfected HeLa cells with different ORFs and examined their effects on tetherin expression.
Interestingly, the study revealed that ORF3a, a protein encoded by SARS-CoV-2, plays a significant role in tetherin downregulation. Expression of ORF3a resulted in the redistribution of tetherin away from biosynthetic organelles and towards late endosomes and lysosomes. This effect was not seen with the other SARS-CoV-2 ORFs.
Furthermore, the researchers found that the mechanism by which ORF3a downregulates tetherin is distinct from ORF7a of SARS-CoV-1, which also antagonizes tetherin but acts through endocytosis and lysosomal degradation. ORF3a is unique to SARS-CoV-2 and does not have a counterpart in SARS-CoV-1. Therefore, this study identified a novel mechanism by which SARS-CoV-2 subverts tetherin activity through ORF3a.
Conclusions and Implications
The study conducted has provided valuable insights into how SARS-CoV-2 subverts tetherin, a host restriction factor that inhibits the release of viral particles from infected cells. The study revealed that SARS-CoV-2 infection leads to the downregulation of tetherin, which enhances the virus's ability to spread.
This downregulation is not solely dependent on the virus's ability to antagonize interferon responses, indicating that other mechanisms are at play.
The researchers identified the SARS-CoV-2 protein ORF3a as a significant factor responsible for tetherin downregulation. ORF3a's ability to redistribute tetherin away from biosynthetic organelles and towards late endosomes and lysosomes represents a novel mechanism distinct from other known tetherin antagonists.
This research has important implications for our understanding of SARS-CoV-2 infection and its interactions with the host immune response. By identifying specific viral proteins responsible for counteracting tetherin and understanding the mechanisms involved, scientists may be able to develop targeted therapies or interventions to enhance the host's ability to restrict viral egress. Additionally, this knowledge may contribute to the development of antiviral strategies that can be applied to combat not only SARS-CoV-2 but other enveloped viruses as well.
As the COVID-19 pandemic continues to evolve, ongoing research into the virus's interactions with host factors and immune responses is essential to inform public health measures and treatment strategies. Studies like this one provide a deeper understanding of the complex relationship between the virus and the host, which is crucial in the global effort to control and ultimately overcome COVID-19.
The study findings were published in the peer reviewed journal: EMBO Reports.
https://www.embopress.org/doi/full/10.15252/embr.202357224
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
