The Dynamic Interplay Between SARS-CoV-2 And Mitochondrial Function: Implications for Long COVID
Long COVID - SARS-CoV-2 - Mitochondria Apr 09, 2023 3 years, 1 month, 3 weeks, 3 hours, 58 minutes ago
Long COVID: Mitochondria, the cellular powerhouses, are crucial for cellular energy production and homeostasis. They form dynamic networks within cells, adapting their morphology to meet fluctuating energy demands. Interestingly, the mitochondrial network is vulnerable to perturbations in its dynamics and function due to interactions between SARS-CoV-2 proteins and certain mitochondrial proteins. These interactions play a pivotal role in the development of
Long COVID, a condition characterized by lingering symptoms following the acute phase of COVID-19.
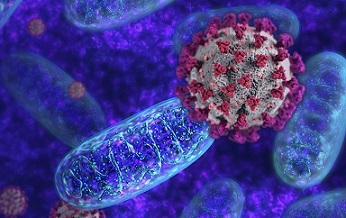
The SARS-CoV-2 M protein interacts with a multitude of mitochondrial proteins, including the beta subunit of mitochondrial processing peptidase (PMPCB), which may disrupt the cleavage of signal peptides from proteins imported into the mitochondrial matrix.
Additionally, the M protein can alter the function of other mitochondrial proteins, such as acyl-CoA medium-chain dehydrogenase (ACADM), coenzyme Q8B (COQ8B), pitrilysin metallopeptidase 1 (PITRM1), and the mitochondrial processing peptidase alpha and beta subunits (PMPCA and PMPCB), among others.
Importantly, the M protein is not the only viral protein capable of modifying mitochondrial function. For instance, the SARS-CoV-2 N protein interacts with eukaryotic translation initiation factor 4E (EIF4E2) family member 2, a repressor of translation initiation. Both M and N proteins of SARS-CoV-2 are known to induce apoptosis in human lung fibroblast cells.
Further, the SARS-CoV-2 nsp4 protein interacts with inner mitochondrial membrane translocases 9,10, and 29 (TIMM 9, 10, and 29), and the viral nsp6 protein associates with the membrane ATP synthase subunit G (ATP5MG), crucial for ATP synthesis during aerobic cellular respiration.
Another non-structural protein, the SARS-CoV-2 nsp7, increases AFM1 gene expression, diminishes both basal and maximal oxygen consumption rates, promotes mitochondrial fission, decreases electron transport chain (ETC) complex I activity, and inhibits oxidative metabolism when introduced into BEAS-2B cells.
These effects result from the interaction between nsp7 and proteins involved in electron transport, such as NADH: Ubiquinone Oxidoreductase Complex Assembly Factors 1 (NDUFAF1) and 2 (NDUFAF2), NADH-cytochrome b5 reductase 3 (CYB5R3), and cytochrome b5 type B (CYB5B).
The SARS-CoV-2 nsp14 has been found to bind with Sirtuin 5 (SIRT5), an NAD-dependent protein deacylase vital for cellular metabolism, which enhances viral replication success. Furthermore, the interaction of nsp14 with Sirtuin 1 (SIRT1) hinders the NRF2/HMOX1 pathway activation, consequently impairing host antioxidant defense.
SARS-CoV-2 also influences mitochondrial protein synthesis and transport. The nsp8 protein may affect the modulation of mitochondrial protein synthesis by interacting with neuroguidine (NGDN), asparaginyl-tRNA synthetase 2 (NARS2), and mitochondrial ribosomal proteins S5, 25, and 27 (MRPS5, 25, and 27) It may also interact with the ETC complex.
Moreover, the nsp10 protein may modulate protein transport from the inner mitochondrial membrane to the mitochondrial matrix through its interaction with the host protein GRPEL1 (GrpE protein homolog
1).
The functional significance of this interaction remains unclear; however, the loss of GRPEL1 has been linked to mitochondrial oxidation of fatty acids, oxidative phosphorylation arrest in musculoskeletal cells, and rapid muscle atrophy.
The SARS-CoV-2 ORF9b protein interacts with Translocase of Outer Membrane 70 (TOM70), altering responses to type I interferon (IFN-I), which may be beneficial for virus replication.
TOM70 is involved in protein transport into mitochondria and facilitates communication between mitochondria and the nucleus by connecting to the nuclear transcriptional activity of mitochondrial proteins. When bound to ORF9b, TOM70 may not properly bind to HSP90, as TOM70-HSP90 binding is crucial for the development of TOM70-mediated IFN-I activation. Furthermore, TOM70 dysfunctions have been associated with lactic acid production induction, which can inhibit IFN-I responses.
Interestingly, previous studies with SARS-CoV revealed that its ORF9b protein causes morphological changes in mitochondria, such as elongation, by promoting the degradation of dynamin-related protein 1 (DRP1), a GTPase responsible for regulating mitochondrial fission. These morphological alterations have been linked to autophagy.
Consistent with protein-host interactions resulting in mitochondrial dysfunctions, the SARS-CoV-2 nsp12 protein is thought to inhibit branched-chain keto acid dehydrogenase kinase (BCKDK), a regulatory enzyme for branched-chain amino acid kinase. The accumulation of branched-chain amino acids, which can inhibit pyruvate dehydrogenase, mitochondrial respiration chain, and α-ketoglutarate dehydrogenase, can cause apoptosis in glial cells and neurons.
Additionally, the nsp7 and nsp8 proteins, both cofactors of the nsp12 protein, interact with ribosomal proteins in mitochondria.
Various interactions between viral and mitochondrial molecules lead to alterations in the mitochondrial antiviral response, induction of programmed cell death programs, and modulation of amino acid and lipid catabolism, as well as energy production through aerobic cellular respiration.
Consequently, mitochondria lose functionality and accumulate damage, which can be considered a side effect of cellular sequestration by the virus to redirect cellular metabolism towards viral replication. Although this condition typically reverses after viral clearance, its persistence may be associated with mitochondrial-associated Long COVID symptoms, possibly resulting from pre-existing suboptimal mitochondrial function.
Cellular mechanisms responsible for eliminating damaged mitochondria become less effective, leading to their accumulation within cells. Previous studies have reported that the buildup of damaged mitochondria and oxidative stress are common factors in the development of various Long COVID clinical manifestations, such as chronic fatigue syndrome, "brain fog," and cognitive impairment.
In fact, the accumulation of damaged mitochondria in pulmonary artery smooth muscle cells and airway epithelial cells has been hypothesized to result from direct hypoxemia and alveolar injury caused by SARS-CoV-2 infection, subsequently inducing hypoxic pulmonary vasoconstriction, which is altered in some COVID-19 patients.
Thus, some of the aforementioned effects of SARS-CoV-2 proteins on mitochondria may lead to cellular stress, decreased energy production, and increased oxidative damage, which could contribute to the development of Long COVID symptoms such as fatigue, "brain fog," reduced exercise tolerance, and muscle weakness.
In conclusion, the intricate interplay between SARS-CoV-2 proteins and mitochondrial function has significant implications for understanding the pathophysiology of Long COVID.
The various interactions between viral proteins and mitochondrial proteins can lead to a cascade of events that disrupt mitochondrial function, compromise cellular energy production, and increase oxidative stress. These factors may contribute to the persistence of symptoms in Long COVID patients, affecting their quality of life and ability to return to normal activities.
Further research is required to elucidate the exact mechanisms behind these interactions and to develop targeted therapies that address mitochondrial dysfunction in Long COVID patients. By understanding the role of mitochondria in the development and progression of Long COVID, researchers and clinicians can better predict and manage the long-term consequences of SARS-CoV-2 infection.
This knowledge could lead to the development of novel therapeutic strategies that not only alleviate Long COVID symptoms but also prevent the onset of this debilitating condition in COVID-19 patients.
Ultimately, the complex relationship between SARS-CoV-2 and mitochondrial function serves as a critical reminder of the importance of maintaining mitochondrial health and integrity in the face of viral infections. As researchers continue to uncover the secrets behind this intricate dance between virus and host, they will pave the way for improved clinical care and patient outcomes, helping to mitigate the long-lasting impacts of COVID-19 on individuals and communities worldwide.
References:
Zhao G, Shi SQ, Yang Y, Peng JP. M and N proteins of SARS coronavirus induce apoptosis in HPF cells. Cell Biol Toxicol. 2006 Sep;22(5):313–22.
https://pubmed.ncbi.nlm.nih.gov/16845612/
Gao S, Zhang L. ACE2 partially dictates the host range and tropism of SARS-CoV-2. Comput Struct Biotechnol J. 2020;18:4040–7.
https://pubmed.ncbi.nlm.nih.gov/33282147/
Archer SL, Dasgupta A, Chen KH, Wu D, Baid K, Mamatis JE, et al. SARS-CoV-2 mitochondriopathy in COVID19 pneumonia exacerbates hypoxemia. Redox Biol. 2022 Dec;58:102508.
https://pubmed.ncbi.nlm.nih.gov/36334378/
Du J, Zhou Y, Su X, Yu JJ, Khan S, Jiang H, et al. Sirt5 is a NAD-dependent protein lysine demalonylase and desuccinylase. Science. 2011 Nov 11;334(6057):806–9.
https://pubmed.ncbi.nlm.nih.gov/22076378/
Walter M, Chen IP, Vallejo-Gracia A, Kim IJ, Bielska O, Lam VL, et al. SIRT5 is a proviral factor that interacts with SARS-CoV-2 Nsp14 protein. Dittmann M, editor. PLoS Pathog. 2022 Sep 12;18(9):e1010811
https://pubmed.ncbi.nlm.nih.gov/36095012/
Batra N, De Souza C, Batra J, Raetz AG, Yu AM. The HMOX1 Pathway as a Promising Target for the Treatment and Prevention of SARS-CoV-2 of 2019 (COVID-19). Int J Mol Sci. 2020 Sep 3;21(17):6412
https://pubmed.ncbi.nlm.nih.gov/32899231/
Zhang S, Wang J, Wang L, Aliyari S, Cheng G. SARS-CoV-2 virus NSP14 Impairs NRF2/HMOX1 activation by targeting Sirtuin 1. Cell Mol Immunol. 2022 Jun 23;19(8):872–82.
https://pubmed.ncbi.nlm.nih.gov/35732914/
Feng Y, Tang K, Lai Q, Liang J, Feng M, Zhou ZW, et al. The Landscape of Aminoacyl-tRNA Synthetases Involved in Severe Acute Respiratory Syndrome Coronavirus 2 Infection. Front Physiol. 2021;12:818297
https://pubmed.ncbi.nlm.nih.gov/35153822/
Ghosh N, Saha I, Sharma N. Interactome of human and SARS-CoV-2 proteins to identify human hub proteins associated with comorbidities. Comput Biol Med. 2021 Nov;138:104889.
https://pubmed.ncbi.nlm.nih.gov/34655901/
Wang T, Cao Y, Zhang H, Wang Z, Man CH, Yang Y, et al. COVID-19 metabolism: Mechanisms and therapeutic targets. MedComm [Internet]. 2022 Sep ;3(3).
https://pubmed.ncbi.nlm.nih.gov/35958432/
Neupane N, Rajendran J, Kvist J, Harjuhaahto S, Hu B, Kinnunen V, et al. Inter-organellar and systemic responses to impaired mitochondrial matrix protein import in skeletal muscle. Commun Biol. 2022 Oct 5;5(1):1060.
https://pubmed.ncbi.nlm.nih.gov/36198903/
Jiang HW, Zhang HN, Meng QF, Xie J, Li Y, Chen H, et al. SARS-CoV-2 Orf9b suppresses type I interferon responses by targeting TOM70. Cell Mol Immunol. 2020 Sep;17(9):998–1000
https://pubmed.ncbi.nlm.nih.gov/32728199/
Liu Q, Chang CE, Wooldredge AC, Fong B, Kennedy BK, Zhou C. Tom70-based transcriptional regulation of mitochondrial biogenesis and aging. eLife. 2022 Mar 2;11:e75658.
https://pubmed.ncbi.nlm.nih.gov/35234609/
Miller K, McGrath ME, Hu Z, Ariannejad S, Weston S, Frieman M, et al. Coronavirus interactions with the cellular autophagy machinery. Autophagy. 2020 Dec;16(12):2131–9.
https://pubmed.ncbi.nlm.nih.gov/32964796/
Shi CS, Qi HY, Boularan C, Huang NN, Abu-Asab M, Shelhamer JH, et al. SARS-coronavirus open reading frame9b suppresses innate immunity by targeting mitochondria and the MAVS/TRAF3/TRAF6 signalosome. J Immunol. 2014 Sep 15;193(6):3080–9.
https://pubmed.ncbi.nlm.nih.gov/25135833/
Wang T, Cao Y, Zhang H, Wang Z, Man CH, Yang Y, et al. COVID-19 metabolism: Mechanisms and therapeutic targets. MedComm [Internet]. 2022 Sep [cited 2023 Mar 2];3(3).
https://pubmed.ncbi.nlm.nih.gov/35958432/
Du C, Liu WJ, Yang J, Zhao SS, Liu HX. The Role of Branched-Chain Amino Acids and Branched-Chain α-Keto Acid Dehydrogenase Kinase in Metabolic Disorders. Front Nutr. 2022 Jul 18;9:932670.
https://pubmed.ncbi.nlm.nih.gov/35923208/
Nunn AVW, Guy GW, Brysch W, Bell JD. Understanding Long COVID; Mitochondrial Health and Adaptation— Old Pathways, New Problems. Biomedicines. 2022 Dec 2;10(12):3113.
https://pubmed.ncbi.nlm.nih.gov/36551869/
Stefano GB, Büttiker P, Weissenberger S, Martin A, Ptacek R, Kream RM. Editorial: The Pathogenesis of LongTerm Neuropsychiatric COVID-19 and the Role of Microglia, Mitochondria, and Persistent Neuroinflammation: A Hypothesis. Med Sci Monit. 2021 May 10;27:e933015.
https://pubmed.ncbi.nlm.nih.gov/34016942/
Paul BD, Lemle MD, Komaroff AL, Snyder SH. Redox imbalance links COVID-19 and myalgic encephalomyelitis/chronic fatigue syndrome. Proc Natl Acad Sci U S A. 2021 Aug 24;118(34):e2024358118
https://pubmed.ncbi.nlm.nih.gov/34400495/
Wood E, Hall KH, Tate W. Role of mitochondria, oxidative stress and the response to antioxidants in myalgic encephalomyelitis/chronic fatigue syndrome: A possible approach to SARS-CoV-2 ‘long-haulers’? Chronic Diseases and Translational Medicine. 2021 Mar;7(1):14–26.
https://pubmed.ncbi.nlm.nih.gov/33251031/
Javadov S, Kozlov AV, Camara AKS. Mitochondria in Health and Diseases. Cells. 2020 May 9;9(5):1177.
https://pubmed.ncbi.nlm.nih.gov/32397376/
Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, White KM, et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020 Jul;583(7816):459–68.
https://pubmed.ncbi.nlm.nih.gov/32353859/
Morita M, Ler LW, Fabian MR, Siddiqui N, Mullin M, Henderson VC, et al. A novel 4EHP-GIGYF2 translational repressor complex is essential for mammalian development. Mol Cell Biol. 2012 Sep;32(17):3585–93.
https://pubmed.ncbi.nlm.nih.gov/22751931/
For the latest
Long COVID News, keep on logging to Thailand Medical News.
