U.S. Study Shows That Upper-Room Germicidal Ultraviolet-C Irradiation (UR GUV) Devices Effective in Eradicating Aerosolized SARS-CoV-2 In Enclosed Areas
Medical News - Upper-Room Germicidal Ultraviolet-C Irradiation (UR GUV) Devices Oct 29, 2022 3 years, 6 months, 1 week, 3 days, 17 hours, 10 minutes ago
A new study by a private American biosafety technology company called R-Zero Systems in Utah has found that using Upper-Room Germicidal Ultraviolet-C Irradiation (UR GUV) devices are effective in eradicating aerosolized SARS-CoV-2 in enclosed areas.

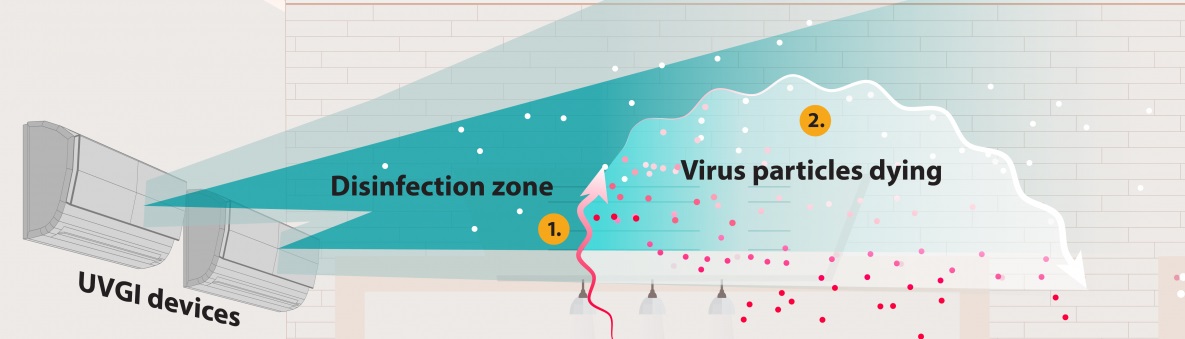
Ultraviolet germicidal irradiation, or UVGI, is the use of ultraviolet (UV) energy to kill viral, bacterial, and fungal organisms. UVGI fixtures produce UV-C energy, which has shorter wavelengths than more penetrating UV-A and UV-B rays and pose less risk to human health. Upper-room UVGI refers to a disinfection zone of UV energy that is located above people in the rooms they occupy. This kills airborne pathogens in the room where they are released. Fixtures are installed to prevent direct UV exposures to people in the room.
Air passes through the disinfection zone from air flow through HVAC system, fans, and/or open windows.
The airborne pathogens are killed once they receive an appropriate amount of UV energy. The particles remain in the air, but they are no longer infectious.
Note: For airborne viral particles, upper-room UVGI systems provide air changes per hour that are similar to the introduction of clean air into the space.
The COVID-19 disease can cause life-threatening respiratory infections that have had a profound impact on indoor air quality awareness. Ultraviolet-C (UV-C) is a physical disinfection process that triggers microbial inactivation through creating irreversible genetic material damage.
In this study, an upper room device equipped with germicidal UV-C (UR GUV) was evaluated against airborne SARS-CoV-2 for antimicrobial efficacy using a robust aerosol testing protocol. In 30 minutes, it led to a virucidal efficacy of 99.994 % in a large, room-sized chamber. UR GUV is a promising mitigation strategy for airborne pathogens.
The study findings were published on a pre-print server and are currently being peer reviewed.
https://www.biorxiv.org/content/10.1101/2022.10.04.510919v1
The upper-room germicidal ultraviolet-C (UV-C) irradiation (also known as UR GUV) is an infection prevention tool first indicated for the inactivation of airborne Mycobacterium tuberculosis and has subsequently seen broader adoption during COVID-19.
Basically, UR GUV fixtures create a field of non-ionizing radiation above the heads of indoor room occupants. The irradiation field acts as a disinfection zone leading to the inactivation of airborne microorganisms that move from the breathing zone to the disinfection zone through natural vertical air movement.
It is already known that UV-C light induces damage to the genomes of bacteria, viruses, and other microorganisms by breaking bonds and forming photodimeric lesions in nucleic acids. These lesions prevent both transcription and replication and ultimately lead to microbial inactivation.
However, despite significant technological advancements in the field, questions remain about the real world efficacy of UR GUV fixtures as a standard Environmental Protection Agency (EPA) method does not currently exist for evaluating the efficacy of UV-C against airborne pathogens.
Hence the study team decided to assess the real-time inactivation of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) using
ultraviolet-C.
With many waves and surges involving various emerging SARS-CoV-2 variants and sub-lineages, the ongoing COVID-19 pandemic has severely disrupted human health, societies, and healthcare systems worldwide. The introduction of more contagious SARS-CoV-2 variants and COVID-19-related long-term health issues continue to have a negative impact on the global economy and health despite the fact that vaccinations and public health precautions have significantly reduced the risk of serious COVID-19 outcomes.
The study team strongly believes that of UR GUV fixtures could help societies revert back to a pre-COVID normal if deployed extensively.
For the study, a patient room-sized chamber was used to test the effectiveness of UR GUV devices against large quantities of live viral aerosols. SARS-CoV-2 was used as the challenge organism.
Conforming to biosafety Level 3 (BSL3) requirements, testing was performed in a sealed 20'x8'x8' chamber. The test chamber's overall dimensions produced an air displacement volume of roughly 36,245.56 liters, exceeding the EPA's recommended room size and simulating real-world testing of disinfection systems against aerosolized bacteria. The center of the 20-foot wall included a nebulizing port that protruded 24 inches from the wall and was coupled with a programmable compressor system.
A total of four air sampling probes were installed in the room; each placed six feet off the chamber floor and in the middle of the space. At the center of one of the eight-foot walls, roughly 6.5 feet off the ground, was mounted a UR GUV device with 48 light-emitting diodes emitting from the light source.
In order to produce bioaerosol, a Blaustein Atomizing Module (BLAM) having preset pounds per square inch (PSI) along with a computer-controlled liquid delivery system was evaluated for average particle size distribution.
The study team placed 7.04 x 106 median tissue culture infectious dose (TCID50)/mL of SARS-CoV-2 in a viral suspension media in the nebulizer, and untreated local atmospheric air was nebulized at a flow rate of 1 mL/min. The residual viral stock volume in the nebulizer was weighed after nebulization to ensure that roughly the same volume was nebulized for each of the three replicate runs.
The four air sampling probes, each linked to a calibrated Gilian 10i vacuum device and set to a standard flow of 5.02 L/min with a tolerance of 0.20%, were used to collect bioaerosol samples at zero, five, 15, and 30 minutes.
At each time point, sample collection volumes were adjusted to ten-minute draws, allowing for roughly 50 liters of air collection per collection port. The collection of air samples was restricted to ten-minute intervals. To calculate a mean across the four sampling sites, all sample discs were combined into a single collection tube. The UR GUV device was then operated in the same manner with appropriate time points and collection rates following the control runs. Utilizing the TCID50 approach, the viral suspensions from treated and control discs were measured.
The study findings showed demonstrated a meaningful recovery after a natural viability reduction of aerosolized SARS-CoV-2 for 30 minutes inside the chamber under controlled conditions.
It was found that after five minutes of device operation, the initial concentration of 7.04 x 106 TCID50/mL for three tests against SARS-CoV-2 was lowered to 4.55 x 106, 4.72 x 106, and 4.25 x 106 TCID50/mL, averaging to about 4.51 x 106 TCID50/mL.
The UR GUV device decreased the collected SARS-CoV-2 after 15 minutes to 1.25 x 106, 9.86 x 105, and 7.47 x 105, with an average of about 9.93 x 105 TCID50/mL. Approximately 30 minutes after, the collected SARS-CoV-2 reduced to 4.80 x 102, 1.20 x 102, and 1.20 x 102, average 2.40 x 102 TCID50/mL.
The study findings demonstrated a robust aerosol testing protocol which showed that the tested UR GUV device was highly effective against airborne SARS-CoV-2.
The study team also believe that this study protocol can be used as a reference by manufacturers and infection preventionists to assess the effectiveness of other UR GUV technologies.
Companies or individuals interested to procur UR GUV devices for either residential, office, retail or other settings can contact Thailand
Medical News as we have a variety of Ur GUR device suppliers and also contractors who can do the installation for you anywhere in Thailand, Vietnam, Malaysia, Australia, Japan, United States, Canada, Spain and Germany.

