COVID-19 News: Study Shows That Airway Basal Stem Cells In COVID-19 Exhibit a Proinflammatory Signature And Impaired Mucocililary Differentiation
Nikhil Prasad Fact checked by:Thailand Medical News Team Sep 25, 2023 2 years, 7 months, 5 days, 1 hour, 51 minutes ago
COVID-19 News: The COVID-19 pandemic, caused by the SARS-CoV-2 virus, has left an indelible mark on the world. Beyond the immediate health crises and overwhelming hospitalizations, the virus has revealed itself to be a formidable adversary, with its profound effects rippling through various aspects of human physiology. Among these, the respiratory system stands as one of the primary battlegrounds, where the virus launches its initial assault, resulting in extensive damage to the airway epithelium and unleashing imbalanced host responses. A crucial player in the complex orchestration of respiratory epithelial regeneration is the airway basal stem cells (BSCs), a fascinating and often overlooked component of the pulmonary microenvironment.
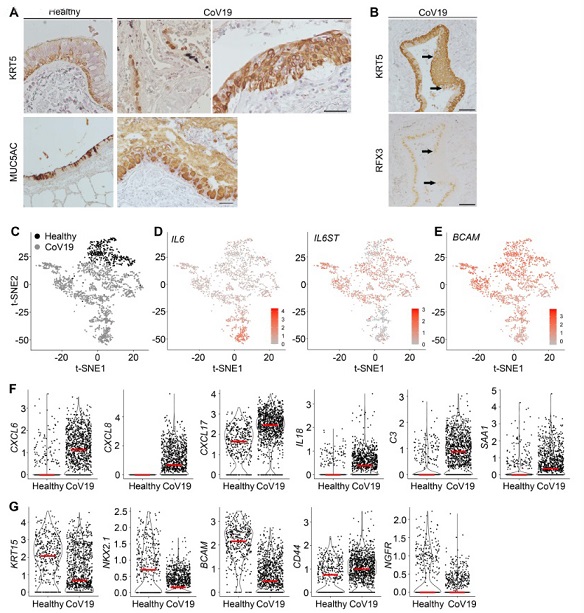 KRT5+ BSCs line the base of pseudostratified airway epithelium of healthy donor lungs (Figure 1A). KRT5 staining of postmortem CoV19 lung samples showed two distinct configurations of BSCs in areas with significant lung injury: KRT5+ BSCs were found as a monolayer above fragmented basement membrane and also in large cell clusters (Figure 1A). Staining for differentiated epithelial cell markers showed a significant increase in MUC5AC in areas with relatively intact pseudostratified epithelium in CoV19 lung samples (Figure 1A), consistent with previous findings of mucus hyperplasia in CoV19 patients. In addition, RFX3+ ciliated cells were found abutting KRT5+ BSC clusters in the conducting airway of postmortem CoV19 lung samples (Figure 1B), suggesting ongoing epithelial differentiation. These findings, together with BSCs being the major proliferating cell type in the conducting airway of CoV19 patients, are consistent with an established role of BSCs in epithelial regeneration following respiratory viral infection. To evaluate changes in BSC gene expression in CoV19, we integrated two already-published datasets of single cell RNA-seq of lung samples from two CoV19 patients (EGAS00001004481) (5) and two healthy donors (EGAS00001004344) (31). We specifically re-analyzed single cell transcriptomes of BSCs using BSC markers established in these two studies. t-distributed stochastic neighbor embedding (t-SNE) showed that compared to BSCs in healthy lungs, BSCs in CoV19 exhibited an increased heterogeneity of gene expression (Figure 1C-E). BSC subtypes with relatively high levels of IL6 and IL6ST expression were uniquely expanded in CoV19 (Figure 1D). Consistently, genes involved in inflammation were among the most upregulated genes in BSCs of CoV19 patients, including chemokine genes (CXCL6/8/17), cytokine and receptor genes (IL6/18, IL6ST), C3, and SAA1 (Figure 1F). BSCs in CoV19 also showed significant changes in expression of lower respiratory epithelial and BSC marker genes including CD44 (upregulated), BCAM, KRT15, NGFR, and NKX2.1 (downregulated) (Figure 1, E and G). Taken together, BSCs in CoV19 display a more diversified transcriptome profile than BSCs in healthy controls. In addition, BSCs with a proinflammatory gene signature are the predominant BSC subpopulation in CoV19
KRT5+ BSCs line the base of pseudostratified airway epithelium of healthy donor lungs (Figure 1A). KRT5 staining of postmortem CoV19 lung samples showed two distinct configurations of BSCs in areas with significant lung injury: KRT5+ BSCs were found as a monolayer above fragmented basement membrane and also in large cell clusters (Figure 1A). Staining for differentiated epithelial cell markers showed a significant increase in MUC5AC in areas with relatively intact pseudostratified epithelium in CoV19 lung samples (Figure 1A), consistent with previous findings of mucus hyperplasia in CoV19 patients. In addition, RFX3+ ciliated cells were found abutting KRT5+ BSC clusters in the conducting airway of postmortem CoV19 lung samples (Figure 1B), suggesting ongoing epithelial differentiation. These findings, together with BSCs being the major proliferating cell type in the conducting airway of CoV19 patients, are consistent with an established role of BSCs in epithelial regeneration following respiratory viral infection. To evaluate changes in BSC gene expression in CoV19, we integrated two already-published datasets of single cell RNA-seq of lung samples from two CoV19 patients (EGAS00001004481) (5) and two healthy donors (EGAS00001004344) (31). We specifically re-analyzed single cell transcriptomes of BSCs using BSC markers established in these two studies. t-distributed stochastic neighbor embedding (t-SNE) showed that compared to BSCs in healthy lungs, BSCs in CoV19 exhibited an increased heterogeneity of gene expression (Figure 1C-E). BSC subtypes with relatively high levels of IL6 and IL6ST expression were uniquely expanded in CoV19 (Figure 1D). Consistently, genes involved in inflammation were among the most upregulated genes in BSCs of CoV19 patients, including chemokine genes (CXCL6/8/17), cytokine and receptor genes (IL6/18, IL6ST), C3, and SAA1 (Figure 1F). BSCs in CoV19 also showed significant changes in expression of lower respiratory epithelial and BSC marker genes including CD44 (upregulated), BCAM, KRT15, NGFR, and NKX2.1 (downregulated) (Figure 1, E and G). Taken together, BSCs in CoV19 display a more diversified transcriptome profile than BSCs in healthy controls. In addition, BSCs with a proinflammatory gene signature are the predominant BSC subpopulation in CoV19
A new study conducted at Massachusetts General Hospital in Boston-USA sheds light on the impact of COVID-19 on airwa
y basal stem cells. This research uncovers a proinflammatory signature and impaired mucociliary differentiation in these vital cells, providing valuable insights into the long-term consequences of the disease. To fully grasp the significance of these findings, we will in this
COVID-19 News coverage explore the role of BSCs in respiratory health, the methods employed to study them, and the implications for future therapeutic strategies.
The Role of Airway Basal Stem Cells in Respiratory Health
To comprehend the significance of this study, it is essential to understand the pivotal role that airway basal stem cells play in maintaining respiratory health. These remarkable cells are situated at the base of the pseudostratified airway epithelium, extending from the trachea to the bronchioles within the human lung. Their identification is facilitated through specific basal cell markers, such as p63 and KRT5. Studies using post-mortem lung samples from COVID-19 patients have demonstrated that BSCs constitute a predominant portion of proliferating cells in the intrapulmonary bronchi, underscoring their indispensable role in epithelial regeneration.
BSCs are known to mediate the repair and regeneration of the airway epithelium following respiratory viral infections. Inflammatory cytokines, such as IL-6, have been shown to modulate the mucociliary differentiation of these cells, and interferons induced by viral infections can compromise the epithelial barrier, increasing susceptibility to secondary infections. Therefore, the state of BSCs is intimately linked to the overall health and function of the respiratory system.
The Challenge of Studying Airway Basal Stem Cells
Traditionally, BSCs have been isolated from surgical resections and bronchial brush samples, which require invasive or post-mortem access to the human lung. However, this approach poses significant biosafety concerns, particularly in the context of COVID-19, where the risk of viral transmission is a paramount concern. Therefore, alternative methods for studying BSCs were necessary to circumvent these challenges.
In this new study, the research team employed a novel technique, using tracheal aspirates (TA) as a source of functional BSCs. These aspirates, collected during routine care of intubated patients, contain rare BSCs that slough off due to injuries sustained during intubation and suctioning, in addition to epithelial damage related to specific lung diseases. BSCs derived from TA of control patients, intubated due to non-respiratory conditions, were shown to differentiate into functional airway epithelium, establishing the viability of this approach.
The COVID-19 Impact on Airway Basal Stem Cells
To assess the effect of COVID-19 on BSCs, the researchers derived BSC lines from tracheal aspirates of COVID-19 patients who tested negative for SARS-CoV-2 viral RNA. These cells were termed "CoV19-exposed BSCs." Comparing the transcriptome and mucociliary differentiation of these CoV19-exposed BSCs with control BSCs yielded critical insights into the long-term consequences of acute inflammation in COVID-19.
Results of the Study
The study revealed several noteworthy findings:
-
Proinflammatory Gene Signature: CoV19-exposed BSCs exhibited a proinflammatory gene signature, indicating a sustained state of inflammation within these cells.
-Bona Fide Bronchial BSCs: BSCs derived from tracheal aspirates were confirmed to be bona fide bronchial BSCs, capable of differentiating into airway epithelial cells.
-STAT3 Hyperactivation: CoV19-exposed BSCs displayed hyperactivation of STAT3, a signaling molecule associated with inflammation and cell differentiation.
-Goblet Cell Hyperplasia: These cells also exhibited goblet cell hyperplasia, as evidenced by overexpression of MUC5AC, a mucin associated with goblet cells.
-Induction of CoV19-Exposure Phenotypes in Control BSCs: Remarkably, the study demonstrated that control BSCs, when pretreated with a cytokine cocktail, developed phenotypes resembling CoV19-exposed BSCs, suggesting that the inflammatory milieu in COVID-19 may play a critical role in driving these changes.
-Partial Blockade of STAT3 Hyperactivation: Partial blockade of STAT3 hyperactivation normalized the mucociliary differentiation of CoV19-exposed BSCs, hinting at potential therapeutic interventions.
Implications and Future Directions
The study's findings hold significant implications for our understanding of COVID-19's long-term effects on respiratory health. CoV19-exposed BSCs provide a valuable translational model for investigating the inflammatory mediators that impair the mucociliary differentiation of BSCs. This knowledge is crucial for the development of therapeutic strategies aimed at facilitating the recovery of patients with severe COVID-19.
However, it's important to acknowledge the study's limitations. The BSCs used were exclusively derived from COVID-19 patients with severe infections requiring intubation. Whether patients with milder to moderate COVID-19 develop similar proinflammatory signatures in their BSCs remains an open question. Additionally, the study does not address the duration of the observed phenotypes in CoV19-exposed BSCs, an important consideration given the persistence of post-COVID respiratory symptoms in some patients.
Furthermore, animal studies are lacking, primarily due to differences in BSC distribution between rodents and humans. Nevertheless, the study's findings provide a strong foundation for future research, highlighting the need for a deeper understanding of the mechanisms underlying prolonged BSC phenotypes following inflammatory exposure in COVID-19 and potentially other respiratory infections.
Conclusion
In conclusion, the impact of COVID-19 extends beyond its acute phase, leaving lasting effects on the respiratory system. The study conducted at Massachusetts General Hospital underscores the importance of airway basal stem cells in maintaining respiratory health and reveals a proinflammatory signature and impaired mucociliary differentiation in CoV19-exposed BSCs. This research not only enhances our comprehension of COVID-19's long-term consequences but also offers a potential avenue for therapeutic intervention, ultimately improving the recovery prospects of patients affected by this devastating disease. As we continue to navigate the ever-evolving landscape of COVID-19 research, the role of airway basal stem cells stands as a critical piece of the puzzle in our quest for effective treatments and interventions.
The study findings were published in the peer reviewed American Journal of Respiratory Cell and Molecular Biology.
https://www.atsjournals.org/doi/abs/10.1165/rcmb.2023-0104OC
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
