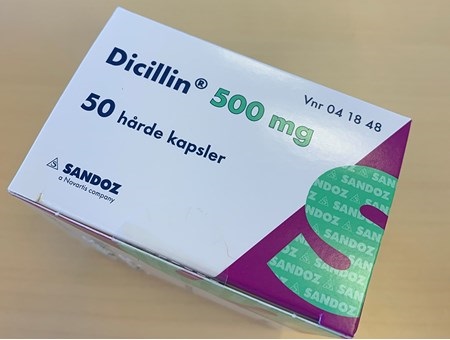
Pharma News: Danish Authorities Initiates Recall Of Sandoz’s Dicillin Antibiotic After Outbreak of Carbapenemase-Producing ST79 Enterobacter Hormaechei!
Pharma News - Sandoz Dicillin Feb 24, 2023 3 years, 2 months, 1 week, 1 day, 17 hours, 50 minutes ago
Pharma News: The Danish Medicines Agency has initiated a recall of Sandoz’s Dicillin antibiotics after it was discovered that the drugs were contaminated and lead to users developing an infection involving Carbapenemase-Producing ST79 Enterobacter Hormaechei.
https://laegemiddelstyrelsen.dk/da/nyheder/2023/antibiotikalaegemidlet-dicillin-fra-firmaet-sandoz-tilbagekaldes/
The recall was initiated after it was found that recently that 11 patients were found positive with a unique epitype of the bacterium Enterobacter hormaechei .
Health authorities found that what the patients have in common is that they have taken the medicine dicloxacillin in capsules from the same manufacturer.
CPE (carbapenemase-producing Enterobacterales) are bacteria (bugs) that live in the gut.
Carbapenemase-producing organisms (CPO) are a group of bacteria that can be difficult to treat because they are resistant to the antibiotics normally used to treat serious infections.
The 11 patients recently found with the infection due to the contaminated antibiotics were not the first to be identified in Denmark.
It was reported that from October 2022 to January 2023, 9 CPO patients with the same unique epitype have been registered: Two cases in October 2022, two cases in December 2022 and 5 cases in January 2023.
Importantly, the cases had no relation to each other, there was no suspicion that the infection had occurred in hospital setting or during travel.
Doctors found the epitype found was so unique and characteristic that leading clinical microbiology departments were contacted to clarify the cause of the outbreak.
Researchers at Odense University Hospital’s clinical microbiology department subsequently noticed that 3 patients who had tested positive in their laboratory had received treatment with Dicillin ® capsules shortly before the outbreak bacteria was detected.
Upon examining unopened packages of Sadoz’s Dicillin medications, they found that the CPO bacteria could be grown from the surface of a capsule of Dicillin 500 mg products.
Detailed investigations by other clinical microbiology departments and the SSI were able to confirm that all the infected patients had been prescribed Dicillin from the same manufacturer prior to the positive test finding of the epitype.
As a result, the Danish health authorities and the Danish Medicines Agency issued an urgent recall of the antibiotics.
It is not known if similar stocks of the medications are also causing problems elsewhere in Europe. Sandoz’s Dicillin antibiotics are extensively distributed all over Europe and also parts of Africa. Some
Pharma News coverages reported oubreaks involving Carbapenemase-Producing ST79 Enterobacter Hormaechei in Poland, Rom
ania and also the United Kingdom but at this stage there is no confirmations if the drugs were involved.
Healthcare professionals are advised to monitor the usage of the drug and any reported cases of Carbapenemase-Producing ST79 Enterobacter Hormaechei infections.
For the latest
Pharma News, keep on logging to Thailand Medical News.
