Glaucoma News: Unveiling the Mysteries of Glaucoma - Proteomic Insights Into Early Molecular Changes In Response To Ocular Hypertension
Nikhil Prasad Fact checked by:Thailand Medical News Team Oct 08, 2023 2 years, 7 months, 3 weeks, 3 days, 17 hours, 41 minutes ago
Glaucoma News: Glaucoma, a group of optic neuropathies characterized by the progressive degeneration of retinal ganglion cells (RGCs), is a leading cause of irreversible blindness worldwide. Despite its significant impact on vision, the exact pathogenesis of glaucoma remains a subject of ongoing research. The absence of early diagnostic markers and targeted therapeutic approaches further compounds the challenge, making glaucoma a pressing public health concern. Among the known risk factors, elevated intraocular pressure (IOP) stands out as a key contributor to glaucoma onset and progression. However, the intricate molecular events linking elevated IOP to RGC lesions during the early stages of glaucoma remain poorly understood.
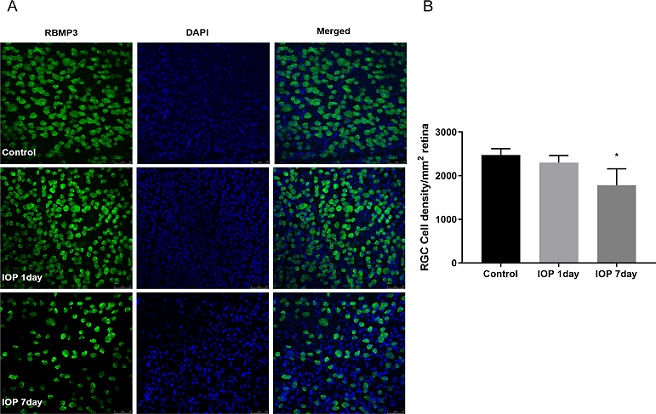 Effect of elevated intraocular pressure (IOP) on retinal morphology. (A) Double-labeling immunohistochemistry of DAPI and RBMP3 in the control on days 1 and 7 of elevated IOP (Scale bar = 50 mm). (B) Retinal ganglion cells decreased on day 7 after elevated IOP (2474 ±84 RGCs/mm2 vs. 1782±216 RGCs/mm2, p<0.05). *, vs. control; *P<0.05.
Effect of elevated intraocular pressure (IOP) on retinal morphology. (A) Double-labeling immunohistochemistry of DAPI and RBMP3 in the control on days 1 and 7 of elevated IOP (Scale bar = 50 mm). (B) Retinal ganglion cells decreased on day 7 after elevated IOP (2474 ±84 RGCs/mm2 vs. 1782±216 RGCs/mm2, p<0.05). *, vs. control; *P<0.05.
Recent studies have illuminated some aspects of the molecular changes associated with glaucoma, albeit with a focus on advanced stages of the disease when RGC damage is already significant. The elusive question is what molecular alterations transpire during the initial phase of elevated IOP to the eventual emergence of RGC lesions. This
Glaucoma News report delves into a comprehensive proteomic study conducted by researchers from Beijing Institute of Ophthalmology, Beijing Tongren Eye Center, Beijing Tongren Hospital, Capital Medical University, Beijing Ophthalmology & Visual Sciences Key Laboratory-China and Beijing Institute of Brain Disorders-China on a rat model of ocular hypertension (OHT) to unravel the intricate time- and tissue-specific molecular changes occurring in the retina, optic nerve head (ONH), and optic nerve (ON) during the early stages of glaucoma pathogenesis.
Understanding these early molecular changes is imperative to decipher the underlying mechanisms driving glaucoma and to identify potential therapeutic targets. The study employed a state-of-the-art quantitative proteomics approach to uncover a myriad of significant findings related to mitochondrial dysfunction, endoplasmic reticulum (ER) stress, heat shock proteins, inflammation, and cytoskeletal dysfunction. In this in-depth analysis, we will explore these findings in detail, shedding light on the molecular intricacies of early glaucomatous progression.
Heat Shock Proteins: Guardians of Retinal Integrity
One of the most intriguing discoveries from the study centers around heat shock proteins, specifically crystallins. Crystallins, belonging to the family of small heat shock proteins, are expressed within RGCs and are known to play pivotal roles in cellular defense mechanisms. These proteins have garnered attention for their involvement in injury response and post-injury repair processes. The study identified a significant upregulation of crystallins, including CRYAA, CRYBA1, CRYBA4, and CRYBB1, on day 1 of OHT, followed by a subsequent downregulation on day 7. This dynamic shift in crystallin expression patt
erns hints at their pivotal role in the early stages of glaucoma, potentially serving as protective factors against stress-induced apoptosis.
Mitochondrial Dysfunction: A Core Culprit
Mitochondrial dysfunction has emerged as a central player in glaucoma pathogenesis. RGCs, with their high oxygen consumption, are particularly susceptible to alterations in mitochondrial function. Mitochondria are renowned as the primary producers of reactive oxygen species (ROS), primarily generated through electron leakage from complexes I and III of the oxidative phosphorylation pathway. Furthermore, the inhibition of electron transport along this pathway exacerbates ROS production. The study revealed a downregulation of mitochondria-related proteins such as Ndufa4, Atp5o, and Atp5mj in all three tissues on day 1 of OHT. This observation signifies early mitochondrial impairment.
Intriguingly, the study also detected an upregulation of proteins linked to glutathione metabolism and respiratory electron transport, notably Gstm1, Gstp1, Gstk1, and Gsta1, after 7 days of OHT in the retina. These findings suggest the activation of a protective response against oxidative stress. Glaucoma is recognized for inducing oxidative stress in RGCs due to elevated IOP. To counteract the detrimental effects of reactive oxygen species, cells rely on antioxidant enzymes like superoxide dismutase, catalase, and glutathione transferase, as well as small molecule antioxidants such as glutathione.
ER Stress: The Unfolded Response
Endoplasmic reticulum (ER) stress is increasingly being implicated in RGC death in chronic glaucoma models. Cells mount an unfolded protein response (UPR) to alleviate ER stress and restore ER homeostasis. The three primary molecular players in this response are pancreatic ER kinase (PERK), activating transcription factor 6, and inositol-requiring transmembrane kinase/endonuclease 1. The study indicated a downregulation of the EIF2 signaling pathway, associated with UPR, in all three tissues on day 1 of OHT. This suggests an early cellular response aimed at mitigating ER stress.
Intriguingly, the study also observed an upregulation of LXR/RXR and estrogen receptor signaling pathways, both known to protect RGCs against ER stress. These findings underscore the complex nature of cellular responses to ER stress and suggest the existence of multiple pathways involved in disease pathogenesis, beyond the ER stress-induced cell death pathway.
Inflammation: The Immune Connection
Inflammation is a pivotal factor in the pathogenesis of glaucoma. The study identified an upregulation of alpha-2 macroglobulin (A2M) in all three tissues on both days 1 and 7 after OHT. A2M is a multifaceted protein known for its roles in the immune response, protein conformation stabilization, and acute-phase response in immune-mediated inflammatory reactions within the central nervous system. Interestingly, a previous study associated serum A2M concentrations with preclinical Alzheimer's disease, suggesting its potential as an early biomarker for neuronal damage in glaucoma.
Additionally, the study highlighted the activation of the complement and coagulation cascades after 1 day of OHT, particularly in the retina. This activation indicates immune responses occurring in different tissues during the early stages of glaucoma. The complement system, represented by proteins such as C3, Gc, Fga, Hpx, Plg, Itih4, Pzp, and Fetub, becomes involved in the response to elevated IOP. Fragments of the complement system attract and activate phagocytes within the retina, including microglia and macrophages.
Cytoskeleton: The Scaffold of RGC Function
The cytoskeleton plays a pivotal role in RGC function. Interestingly, axonal dysfunction and degeneration in RGCs often precede the loss of cell bodies. The study identified changes in microtubule-associated proteins in ONH tissue, including Tubb4a, Tubb2a, and Tuba4a, which were downregulated on day 1 and recovered on day 7 of OHT.
Furthermore, inositol signaling pathways, including INPP5B, INPP4A, INPP4B, and PTEN, appeared to be upregulated on day 1 in the ONH and ON tissues and significantly upregulated by day 7. These findings suggest the involvement of these pathways in early cellular responses to external stimuli.
The study also noted changes in synaptogenic signaling pathways, integrin signaling, and actin cytoskeletal signaling pathways, particularly after 7 days of OHT. These observations point toward potential mechanisms contributing to alterations in extracellular matrix, cell adhesion, and actin dynamics.
Conclusion
In summary, the proteomic study conducted on a rat model of OHT offers critical insights into the early molecular changes that occur within the retina, ONH, and ON during the initial stages of glaucoma pathogenesis. These findings emphasize the significance of mitochondrial dysfunction, ER stress, heat shock proteins, inflammation, and cytoskeletal alterations as key contributors to the early phases of glaucomatous progression.
Crystallins, known for their protective roles in cellular defense mechanisms, demonstrate dynamic expression patterns that may serve as a protective response against stress-induced apoptosis. Meanwhile, mitochondrial dysfunction, characterized by the downregulation of key proteins and the activation of protective pathways, underscores the importance of mitochondrial health in RGC survival.
ER stress, a cellular response to unfolded proteins, is implicated in glaucoma pathogenesis, with potential protective pathways identified. Inflammation emerges as a crucial factor in the disease, with A2M and complement activation suggesting the involvement of immune responses. Lastly, cytoskeletal changes hint at early alterations in RGC function and integrity.
These early molecular events offer a starting point for further investigation and therapeutic development in the quest to unravel the mysteries of glaucoma. Crystallins, mitochondrial function, ER stress pathways, A2M, and cytoskeletal dynamics are among the key areas that warrant in-depth exploration for potential interventions in this debilitating eye disease. Understanding these early molecular changes may pave the way for the development of novel diagnostic tools and targeted therapies, ultimately improving the management of glaucoma and preserving vision in affected individuals.
Th study findings were published in the peer reviewed journal: Molecular and Cellular Proteomics.
https://www.sciencedirect.com/science/article/pii/S1535947623001652
For the latest
Glaucoma News, keep on logging to Thailand Medical News.
