Study Uncovers Role Of Gas6 Protein And TAM Tyrosine Kinase Receptors In COVID-19! Axl Has Been Identified As A Possible SARS-CoV-2 Receptor!
Thailand Medical News Team Aug 13, 2023 2 years, 9 months, 1 week, 1 day, 13 hours, 54 minutes ago
COVID-19 News: In the midst of the global COVID-19 pandemic, the scientific community's relentless pursuit of understanding the intricate mechanisms underlying the disease has unveiled fascinating insights into the role of Growth Arrest-Specific 6 (Gas6) protein and TAM Tyrosine Kinase Receptors. These molecular players, often associated with regulating innate immunity and inflammatory responses, have emerged as intriguing candidates in the complex puzzle of COVID-19 pathogenesis. Furthermore, recent investigations and
COVID-19 News reports have highlighted Axl, a member of the TAM family, as a possible receptor for the SARS-CoV-2 virus, shedding new light on the cellular entry and infection mechanisms.
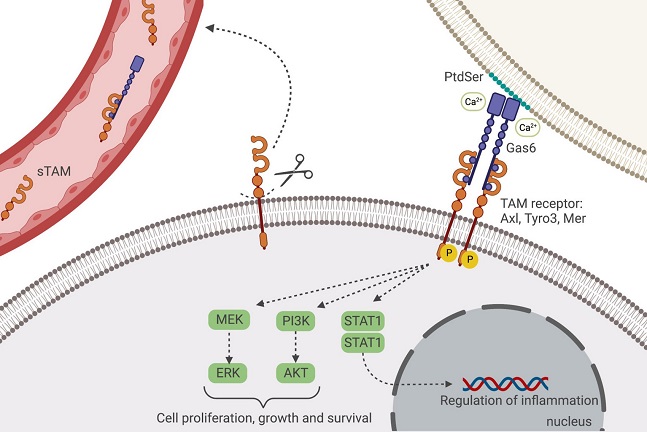 Schematic representation of Gas6/TAM binding and pathways. Gas6 binds to all the three receptors (Axl, Tyro3 and Mer) in the absence or in the presence of phosphatidylserine (PtdSer). In the latter, Gas6 interacts, through a Ca2+-dependent binding, with the PtdSer displayed on the extracellular surface of a plasma membrane and with the TAM-expressing cell. Upon formation of the Gas6/TAM tetrameric complex, the TAM receptor autophosphorylates its tyrosine residues in the kinase domain which activates the downstream signaling pathways, including PI3K/Akt, MEK/ERK and STAT1 pathways. The extracellular domains of TAM receptors can undergo proteolytic cleavage and be released into the bloodstream, acting as decoy receptors for Gas6.
Schematic representation of Gas6/TAM binding and pathways. Gas6 binds to all the three receptors (Axl, Tyro3 and Mer) in the absence or in the presence of phosphatidylserine (PtdSer). In the latter, Gas6 interacts, through a Ca2+-dependent binding, with the PtdSer displayed on the extracellular surface of a plasma membrane and with the TAM-expressing cell. Upon formation of the Gas6/TAM tetrameric complex, the TAM receptor autophosphorylates its tyrosine residues in the kinase domain which activates the downstream signaling pathways, including PI3K/Akt, MEK/ERK and STAT1 pathways. The extracellular domains of TAM receptors can undergo proteolytic cleavage and be released into the bloodstream, acting as decoy receptors for Gas6.
Collaborative research efforts across esteemed institutions such as the University of Piemonte Orientale (UPO) in Italy, AOU Maggiore della Carità Hospital in Italy, and the University of Trieste in Italy, have paved the way for a deeper understanding of these critical interactions.
The TAM family comprises three tyrosine kinase receptors -Tyro3, Axl, and Mer, each with a distinctive role in cellular processes. These receptors, characterized by their structural domains, including extracellular fibronectin type III and immunoglobulin-like domains, play a crucial role in recognizing apoptotic debris by immune cells.
Their interactions with Gas6, a vital ligand, trigger a cascade of events that contribute to immune response regulation, inflammation modulation, and even cancer progression. A noteworthy feature is the formation of a tetrameric complex between Gas6 and TAM receptors, demonstrating the complexity and precision of these molecular interactions.
The recognition of phosphatidylserine (PtdSer), a phospholipid exposed during apoptosis or cellular stress, adds a layer of sophistication to the Gas6/TAM system. This intricate recognition mechanism prompts a phagocytic response by TAM-expressing cells, ultimately influencing downstream signaling pathways such as the phosphoinositide 3 kinase (PI3K)/AKT and mitogen-activated protein kinase (MAPK) pathways. Furthermore, the shedding of soluble TAM receptors by metalloproteases provides an additional layer of regulation, influencing the availability of ligands for interaction. The significance of these interactions is underscored by the detection of increased levels of circulating receptors and Gas6 in various human diseases.
Against the backdrop of COVID-19, the role of
the Gas6/TAM axis takes on a heightened importance. The disease, primarily impacting the respiratory system, has been linked to persistent lung damage and fibrotic abnormalities among survivors. The elusive nature of COVID-related fibrosis has prompted a quest for understanding the underlying mechanisms. The Gas6/TAM axis, with its multifaceted involvement in immune response modulation, has emerged as a potential key player in unraveling the mysteries of COVID-19 pathogenesis.
One noteworthy revelation is the identification of Axl as a potential receptor for the SARS-CoV-2 virus.
https://www.nature.com/articles/s41422-020-00460-y
While ACE2 remains a well-established receptor, its low expression in the respiratory tract has spurred investigations into alternative entry routes. Research findings indicate that Axl, in particular, interacts with the N-terminal domain of the SARS-CoV-2 spike protein, facilitating viral entry into pulmonary and bronchial epithelial cells. The correlation between Axl expression and SARS-CoV-2 infection levels further highlights the potential significance of this receptor in viral pathogenesis. Therapeutic strategies targeting Axl have even been explored as potential avenues for COVID-19 treatment.
Excitingly, recent studies have illuminated the intricate interactions between the Gas6/TAM axis and the innate immune response. The axis is implicated in anti-inflammatory signaling, potentially serving a protective role in response to pathogenic invasion. Elevated levels of plasma Gas6 and soluble TAM receptors have been correlated with disease severity in COVID-19 patients, suggesting their potential as early biomarkers for disease prognosis. These findings emphasize the delicate balance between inflammatory responses and disease outcomes.
The Gas6/TAM system's involvement in lung fibrosis, a hallmark of severe COVID-19 cases, holds significant promise. While much of the focus has been on tumor development and cancer therapy resistance, emerging studies are delving into its role in pulmonary fibrosis. Axl's contribution to alveolar epithelium integrity and the activation of myofibroblasts, along with the role of Gas6 in fibroblast invasion and differentiation, highlight the potential interplay between the Gas6/TAM axis and the development of fibrotic lung diseases. These insights have far-reaching implications, not only for COVID-19 but also for other fibrotic conditions.
In conclusion, the unraveling of the complex interactions between Gas6, TAM receptors, and COVID-19 presents an exciting frontier in biomedical research. As our understanding of these molecular mechanisms deepens, new avenues for therapeutic intervention may emerge, offering hope for improved treatments for COVID-19 patients and others grappling with fibrotic lung diseases. Collaborative efforts across international institutions continue to shed light on these intricate pathways, promising a brighter future in the battle against devastating diseases like COVID-19.
The study findings were published in the peer reviewed journal: Microorganisms.
https://www.mdpi.com/2076-2607/11/8/2038#B130-microorganisms-11-02038
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
Read Also:
https://www.thailandmedical.news/news/covid-19-news-decreased-growth-arrest-specific-6-gas6-and-saxl-plasma-levels-linked-to-hair-loss-in-post-covid-individuals
