SARS-CoV-2, Bacterial Co-Infections And The Onset Of COVID-19 Autoimmune Cardiopathies
COVID-19 News - SARS-CoV-2 - Bacterial Co-Infections - Autoimmune Cardiopathies Jul 30, 2023 2 years, 9 months, 3 weeks, 1 day, 14 hours, 43 minutes ago
Research revealed that cardiac proteins and their mimics, whether from the virus or bacteria, often had complementary relationships.
COVID-19 News: As the COVID-19 pandemic swept the globe, medical researchers raced to understand the mysterious complications arising from SARS-CoV-2 infections and vaccinations. Among these perplexing phenomena were autoimmune cardiopathies (AC)- a rare but severe condition affecting the heart.
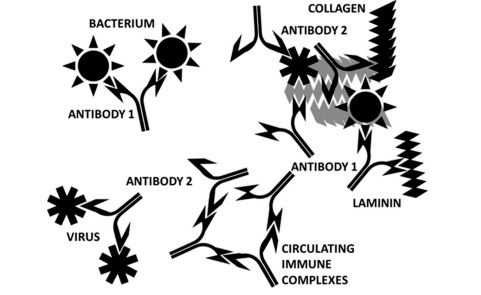 The initiation of autoimmune cardiopathies. A virus that expresses antigens that mimic human cardiac proteins (SARS-CoV-2 and adenoviruses, in this case) elicits a set of antibodies (“antibody 2”). A bacterial co- or super-infection that mimics complementary cardiac proteins induces another set of antibodies (“antibody 1”). Some of these antibodies are complementary to each other (in the sense of idiotype-anti-idiotype) and bind to each other to form circulating immune complexes. The binding of viruses and bacteria to complementary antigens on cardiomyocytes results in the release of cardiac proteins, some of which are recognized by the microbeinduced antibodies because of molecular mimicry. The immune system is now unable to distinguish “self” from “non-self” because each microbe-induced antibody not only cross-reacts with a selfprotein because of mimicry but also recognizes the other set of antibodies as targets as well.
The initiation of autoimmune cardiopathies. A virus that expresses antigens that mimic human cardiac proteins (SARS-CoV-2 and adenoviruses, in this case) elicits a set of antibodies (“antibody 2”). A bacterial co- or super-infection that mimics complementary cardiac proteins induces another set of antibodies (“antibody 1”). Some of these antibodies are complementary to each other (in the sense of idiotype-anti-idiotype) and bind to each other to form circulating immune complexes. The binding of viruses and bacteria to complementary antigens on cardiomyocytes results in the release of cardiac proteins, some of which are recognized by the microbeinduced antibodies because of molecular mimicry. The immune system is now unable to distinguish “self” from “non-self” because each microbe-induced antibody not only cross-reacts with a selfprotein because of mimicry but also recognizes the other set of antibodies as targets as well.
Researchers at Michigan State University embarked on a groundbreaking study to uncover the enigmatic triggers of these conditions. Their findings, based on extensive proteomic analyses and immunoadsorption assays, provided a fascinating insight into the interplay between viral and bacterial infections, shedding light on the possible mechanisms behind autoimmune cardiopathies (AC).
The Prevalence and Mysteries of Autoimmune Cardiopathies
Autoimmune complications have been observed following COVID-19 infections and vaccinations, with myocarditis and pericarditis being the most common among them as reported in various studies and past
COVID-19 News reports. Hospitalized COVID-19 patients showed a high likelihood of developing myocarditis, and even post-recovery patients displayed signs of cardiac damage. Strikingly, a significant proportion of myocarditis cases occurred in individuals who had cleared the viral infection, suggesting a delayed autoimmune response.
Puzzles to Solve - Why Not Everyone and the Role of Bacterial Co-Infections
The study faced intriguing questions: why do only some individuals develop autoimmune cardiopathies? And why are these complications more likely in severe cases or post-vaccination? The researchers noticed a difference in bacterial co-infections between COVID-19 patients and vaccine recipients, with the latter group having a lower probability of bacterial involvement. Bacteria such as Streptococci, Staphylococci, Klebsiella, and Enterococcus faecium were frequently associated with SARS-CoV-2 co-infections. Notably, the
se same bacteria were also linked to triggering endocarditis, suggesting a possible role in cardiac autoimmunity.
The Proteomic Clues: Viral and Bacterial Mimicry
Through proteomic analysis, the research team found that SARS-CoV-2 proteins exhibited a striking resemblance to human cardiac proteins, more so than other viruses. Similarly, certain bacteria, particularly Streptococci, displayed significant similarities to cardiac proteins but in a different set compared to SARS-CoV-2.
Furthermore, antibodies against SARS-CoV-2 and COVID-19-associated bacteria showed a strong affinity for cardiac proteins, confirming the mimicry.
Unraveling the Web of Autoimmunity -Complementarity and Idiotype-Anti-Idiotype Relationships
The study revealed that cardiac proteins and their mimics, whether from the virus or bacteria, often had complementary relationships. Actin and myosin, troponin and myosin, laminin and collagen, and many others bind together as part of normal physiological functions. These complementarities extended to the antibodies produced in response to the infections, creating idiotype-anti-idiotype pairs that could also bind to each other, forming circulating immune complexes (CIC).
The Proposed Model of Autoimmune Cardiopathy Induction
The study team proposed a comprehensive model to explain the induction of autoimmune cardiopathies. In this model, both SARS-CoV-2 and bacterial antigens cooperate to elicit antibodies targeting complementary cardiac antigens, leading to an autoimmune response. The binding complementarities between the antigens and their mimics, along with the formation of CIC, play pivotal roles in destabilizing cardiomyocyte integrity. The subsequent release of hidden antigens further exacerbates the autoimmune activity, confusing the immune system and promoting an ongoing autoimmune disease process.
Implications for COVID-19 Vaccination and Beyond
The study highlighted the low probability of bacterial co-infections among vaccinees, suggesting a potential means to decrease autoimmune cardiopathy risk. Screening individuals for bacterial infections before vaccination and considering vaccination against streptococcal infections could mitigate the risk of autoimmune complications. Furthermore, the findings shed light on the risks associated with adenoviruses and coxsackieviruses, suggesting that certain bacteria might interact with these viruses to trigger autoimmune responses.
Conclusion
Michigan State University's pioneering study has brought us closer to understanding the intricate mechanisms behind COVID-19 autoimmune cardiopathies. The interactions between SARS-CoV-2, bacterial co-infections, and cardiac proteins create a complex web of autoimmunity that demands further exploration. These insights not only contribute to managing COVID-19-related complications but also offer new perspectives on understanding autoimmune diseases triggered by viral and bacterial infections.
The study findings were published in the peer reviewed International Journal of Molecular Sciences.
https://www.mdpi.com/1422-0067/24/15/12177
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
