BREAKING! Study Finds That Diabetes Can Also Contribute To The Development Of Lung Conditions Such As Idiopathic Pulmonary Fibrosis (IPF)!
Diabetes - Idiopathic Pulmonary Fibrosis (IPF) Apr 08, 2023 3 years, 6 days, 8 hours, 12 minutes ago
Medical News: A new study by researchers from Sichuan University-China, Sun Yat-sen University-China and the Aviation Industry Corporation of China Hospital-China has shockingly found that diabetes can also contribute to the development of a variety of lung conditions including asthma, chronic obstructive pulmonary disease (COPD) and more so Idiopathic Pulmonary Fibrosis (IPF) due to its effects in causing endothelial dysfunction and vasculopathy.
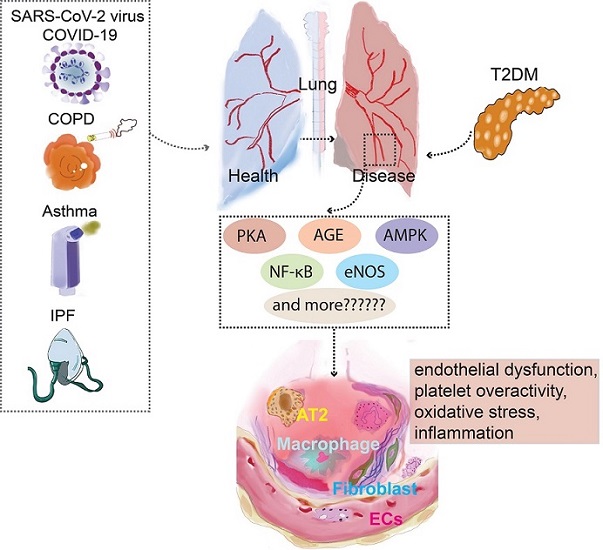 PKA,AGE,AMPK, NF-κB,eNOS are presented as an example for a list of prospective therapeutic targets that diabetes and pulmonary diseases potentially explore through vascular endothelial cells. NF-κB, Nuclear factor kappa B; AT2, Alveolar Type II Cells; COPD, Chronic obstructive pulmonary disease; PKC, Protein kinase C; COVID-19, Coronavirus disease 2019; eNOS, Endothelial NO synthase; AMPK, AMP-activated protein kinase.
PKA,AGE,AMPK, NF-κB,eNOS are presented as an example for a list of prospective therapeutic targets that diabetes and pulmonary diseases potentially explore through vascular endothelial cells. NF-κB, Nuclear factor kappa B; AT2, Alveolar Type II Cells; COPD, Chronic obstructive pulmonary disease; PKC, Protein kinase C; COVID-19, Coronavirus disease 2019; eNOS, Endothelial NO synthase; AMPK, AMP-activated protein kinase.
Monitoring comorbid diseases is crucial for patients with Type 2 diabetes mellitus (T2DM). Vascular diseases, including coronary artery disease, cerebrovascular accidents, retinopathy, nephropathy, and neuropathy, contribute significantly to morbidity and mortality in diabetic patients as covered in numerous past studies and also
Medical News coverages.
Furthermore, diabetes increases the risk of asthma, chronic obstructive pulmonary disease (COPD), obstructive sleep apnea syndrome (OSA), acute lung injury, and respiratory infections.
Many studies have shown that various metabolic pathologies accompany respiratory diseases, and the pathophysiological mechanisms that determine the major degenerative complications of diabetes may also contribute to lung function deficits.
Emerging evidence suggests that endothelial dysfunction plays a significant role in the pathophysiology of diabetic vasculopathy. Endothelial dysfunction is already present in the early stages of diabetic microangiopathy. Although it is unclear whether endothelial dysfunction is a feature of diabetes itself or whether other factors are necessary to cause endothelial dysfunction, given the presence of diabetes, it is believed that endothelial dysfunction is the hallmark stage of diabetes.
Numerous factors contribute to endothelial dysfunction in diabetes, such as activation of protein kinase C (PKC), increased expression of transforming growth factor-beta (TGF-β) and vascular endothelial growth factor (VEGF), non-enzymatic glycation, oxidative stress, activation of the coagulation cascade, increased expression of tumor necrosis factor-alpha (TNF-α), high levels of insulin and insulin precursor molecules, and hyperglycemic pseudohypoxia. These factors may all contribute to endothelial dysfunction. However, the importance of these proposed pathways has not been evaluated in the context of diabetes and lung disease.
Considering that the lungs are anatomically covered in a significant number of vascular endothelial cells, this increases the likelihood that endothelial cell damage caused by diabetic lesions will result in lung disease.
Endothelial cells, therefore, play a special role in "c
onnecting" diabetes and lung disease. The discovery of abnormal pulmonary function in some diabetic patients raises the possibility that the lung should be viewed as a "target organ" for diabetes.
Endothelial dysfunction in chronic lung diseases has been gradually increasing in recent years, especially in Coronavirus disease 2019 (COVID-19) and idiopathic pulmonary fibrosis (IPF), COPD, and asthma.
In contrast, endothelial dysfunction in diabetes leading to acute or chronic airway diseases of the respiratory system is rarely studied. The primary purpose of this study review was to examine the mechanisms of action of the vascular endothelial cells as a shared target in the co-morbidity of T2DM and respiratory disease (COVID-19, COPD, asthma, and IPF) as well as the effects of T2DM treatment on respiratory disease via the vascular endothelial cells.
Potential Mechanisms of Diabetes-Related Lung Disease
The traditional role of blood vessels is to carry oxygen and vital nutrients to other tissues. Endothelial cells, the primary core lesion cells in most vascular disorders, were long believed to be controlled only by angiogenic growth factors like VEGF and other signals like Notch.
However, recent research indicates that the metabolic switch in endothelial cells also has an impact on the angiogenic switch. The vessel wall is impacted by the metabolic environment of T2DM, which includes endothelial dysfunction, platelet overactivity, oxidative stress, and inflammation, as well as insulin resistance, hyperglycemia, and the formation of excess free fatty acids and other metabolic abnormalities.
Vasoconstriction is further intensified, and thrombosis is expedited when these mechanisms are active. The endothelial dysfunction driven on by diabetes is a critical early stage that results in microvascular and macrovascular complications.
Hyperglycemia-induced oxidative stress and inflammation
Hyperglycemia, a characteristic feature of diabetes, is known to induce oxidative stress, leading to the generation of reactive oxygen species (ROS) in endothelial cells. ROS overproduction can damage cellular components, such as proteins, lipids, and DNA, resulting in cellular dysfunction, injury, and eventually, cell death.
Excessive ROS levels can also activate several pro-inflammatory transcription factors, including nuclear factor-kappa B (NF-κB), which in turn, upregulates the expression of various pro-inflammatory cytokines, adhesion molecules, and chemokines. This inflammatory cascade can further exacerbate endothelial dysfunction and damage, contributing to the development and progression of diabetic microangiopathy and associated pulmonary dysfunction.
Non-enzymatic glycation and advanced glycation end products (AGEs)
In diabetes, persistent hyperglycemia leads to the formation of AGEs through non-enzymatic glycation of proteins, lipids, and nucleic acids. AGEs accumulate in the extracellular matrix and bind to their specific receptor (RAGE) on the cell surface. This binding activates intracellular signaling pathways, promoting the production of ROS and the release of pro-inflammatory cytokines, which can further exacerbate endothelial dysfunction and inflammation.
AGEs can also directly alter the structure and function of the extracellular matrix, impairing the mechanical properties of the lung tissue and leading to pulmonary dysfunction.
Endothelial progenitor cells (EPCs) and angiogenesis
EPCs are essential for vascular repair and regeneration, as they can differentiate into mature endothelial cells and contribute to new blood vessel formation (angiogenesis). In diabetes, the number and function of EPCs are impaired, leading to reduced angiogenic capacity and impaired vascular repair. This EPC dysfunction can contribute to the development of diabetic microangiopathy and associated pulmonary complications.
Endothelial-to-mesenchymal transition (EndMT)
EndMT is a process in which endothelial cells lose their characteristic features and acquire a mesenchymal phenotype. This process has been implicated in the development of diabetic microangiopathy and fibrosis. In the context of diabetes-related lung disease, EndMT may contribute to the remodeling and fibrotic changes in the lung parenchyma, leading to impaired lung function.
Therapeutic Approaches Targeting Endothelial Dysfunction in Diabetes-Related Lung Disease
Given the crucial role of endothelial dysfunction in the development of diabetes-related lung disease, several therapeutic strategies have been explored to target and ameliorate this dysfunction. Some of these approaches include:
-Antioxidants: Antioxidant therapies, such as N-acetylcysteine, vitamin C, and vitamin E, have been shown to reduce oxidative stress and improve endothelial function in diabetic patients.
-Anti-inflammatory agents: Medications with anti-inflammatory properties, such as statins, angiotensin-converting enzyme inhibitors (ACEIs), and angiotensin II receptor blockers (ARBs), have been shown to exert beneficial effects on endothelial function in diabetes.
-Glycemic control: Optimal glycemic control is essential to prevent the formation of AGEs and reduce oxidative stress, thus improving endothelial function.
Lifestyle interventions: Lifestyle modifications, including a healthy diet, regular exercise, and smoking cessation, can improve endothelial function and reduce the risk of diabetes-related lung disease.
Type 2 Diabetes Mellitus (T2DM) And Interstitial Pulmonary Fibrosis (IPF)
Interstitial Pulmonary Fibrosis (IPF) and Type 2 Diabetes Mellitus (T2DM) share a connection due to the lung's dense network of connective tissue and alveolar capillaries, making it a potential target for diabetic microvascular damage. High blood sugar levels can cause fibrotic changes and alveolar microangiopathy. This process involves three main stages: oxidative stress injury, alveolar inflammation, and pro-fibrotic cytokine secretion.
Diabetes leads to endothelium damage-related lung injury through several pathways. These include increased expression of AGEs, RAGE, and SIRT, all of which are linked to the onset of the disease. Diabetes causes AGEs to accumulate in the lungs, leading to pulmonary fibroblast aggregation. RAGE stimulates TGF-β production and inhibits Smad2, ERK, and JNK signaling. TGF-β is a key player in tissue remodeling and fibrosis. SIRT is involved in insulin secretion, cell cycle regulation, and apoptosis, and plays a crucial role in diabetic microangiopathy.
SIRT blocks oxidative stress, pro-inflammatory cytokines, mitochondrial DNA damage, and fibronectin. Additionally, pro-inflammatory and pro-fibrotic substances, such as fibronectin and angiotensin II (Ang II), are elevated in diabetic lung tissue. High-glucose environments trigger multiple pathways, leading to intracellular stress and abnormal cytokine production. This results in damage to lung structures and pathological pulmonary fibrosis.
Metformin has potent antifibrotic effects in IPF patients' lungs through various mechanisms. It inhibits TGF-1 activity, collagen synthesis, activates PPAR signaling, and induces lipogenic differentiation of fibroblasts. Metformin also inactivates and triggers apoptosis of myofibroblasts, reversing pulmonary fibrosis progression. GLP-1 receptor agonists, like exendin-4, significantly reduce lung damage and fibrosis by deactivating NF-κB. DPP-4 inhibitors and PPAR-γ agonists have shown promise in treating pulmonary fibrosis. Vildagliptin inhibits endothelial to mesenchymal transition (EndMT), which may be helpful in treating pulmonary fibrosis. PPAR-γ agonists, like rosiglitazone and selegiline, have demonstrated effectiveness in treating BLM-induced lung fibrosis.
Conclusion
Endothelial dysfunction plays a significant role in the pathophysiology of diabetic microangiopathy and associated pulmonary dysfunction. A better understanding of the underlying mechanisms and the development of targeted therapeutic approaches may help to reduce the morbidity and mortality associated with diabetes-related lung disease.
The study findings were published in the peer reviewed journal: Frontiers in Endocrinology.
https://www.frontiersin.org/articles/10.3389/fendo.2023.1073878/full
For the latest
Medical News, keep on logging to Thailand Medical News.
