Nikhil Prasad Fact checked by:Thailand Medical News Team Jan 08, 2026 4 months, 2 weeks, 6 days, 11 hours, 1 minute ago
Medical News: A Tiny Trio Inside the Spike Protein
Scientists from IHU Méditerranée Infection, Microbes Evolution Phylogeny and Infections (MEPHI) at Aix-Marseille University, and INSERM UA16 Faculty of Medicine at Aix-Marseille University have uncovered an astonishing secret buried inside the SARS-CoV-2 spike.
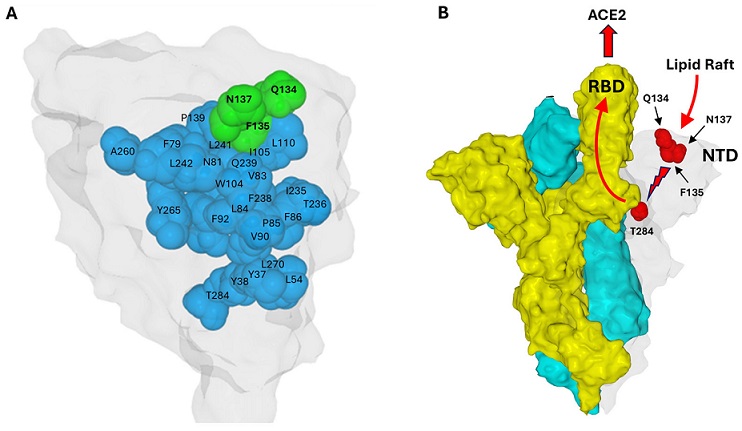 Structural Continuum of the Conformational Wave Initiated by the Q134-F135-N137 Triad Extending to Position 284 in the Wuhan Spike Protein. (A). The NTD surface is colored light gray. The Q134-F135-N137 triad is colored green, the amino acids involved in the conformational wave are blue. (B). Proposed mechanism: A conformational wave initiated by the NTD–lipid raft interaction (light gray protomer) propagates from residue F135 to T284. In the trimeric structure, T284 makes direct contact with the adjacent protomer (colored yellow), triggering the unmasking of its RBD to enable ACE2 binding. The third protomer is colored cyan. In this respect, T284 acts as a conformational transducer that connects the NTD of one protomer and the RBD of the neighbor protomer. Residues Q134, F135, and N137 are solvent-exposed (surface-accessible), whereas the other residues discussed are buried in the protein interior (core/internal residues). Protein structures were generated with MMV.
Structural Continuum of the Conformational Wave Initiated by the Q134-F135-N137 Triad Extending to Position 284 in the Wuhan Spike Protein. (A). The NTD surface is colored light gray. The Q134-F135-N137 triad is colored green, the amino acids involved in the conformational wave are blue. (B). Proposed mechanism: A conformational wave initiated by the NTD–lipid raft interaction (light gray protomer) propagates from residue F135 to T284. In the trimeric structure, T284 makes direct contact with the adjacent protomer (colored yellow), triggering the unmasking of its RBD to enable ACE2 binding. The third protomer is colored cyan. In this respect, T284 acts as a conformational transducer that connects the NTD of one protomer and the RBD of the neighbor protomer. Residues Q134, F135, and N137 are solvent-exposed (surface-accessible), whereas the other residues discussed are buried in the protein interior (core/internal residues). Protein structures were generated with MMV.
Their new review shows that just three amino acids deep in the spike’s N-terminal domain—named Q134, F135 and N137—help trigger the dramatic shape shift that lets the virus invade human cells.
Why the Trio Matters
When the virus first touches a human cell, it does not latch onto ACE2 immediately. Instead, the spike protein lands on lipid rafts, small greasy “landing pads” on the cell surface rich in gangliosides.
The researchers describe how this first contact causes the spike to twist, bend and open, revealing the hidden receptor-binding domain (RBD) that grabs ACE2. Without this flip from closed to open, infection cannot proceed.
What shocked researchers is that despite six years of evolution—from Wuhan through Delta, early Omicron and today’s JN.1—the Q134-F135-N137 triad never mutates. Almost every other nearby region has mutated heavily.
A Domino Effect Through the Spike
Using molecular models and prior lab data, the team found that the triad seems to act like a switch.
When gangliosides touch Q134-F135-N137, a chain reaction spreads through the spike to another amino acid called T284, which sits next to a RBD from the neighboring spike subunit.
The review explains that this push travels like a domino wave moving more than 40 angstroms inside the protein and ends by popping the RBD upright so it can bind ACE2.
Maybe Not Just Mechanical
The central amino acid, F135, is special. It is aromatic, meaning it carries a ring-shaped structure often involved in electronic “hopping” inside proteins.
The team notes that a line of aromatic residues forms a potential electron transfer pathway, raising the possibility of a fast quantum wave spreadi
ng information before the slower mechanical shape change happens.
How the Virus Outfoxes Immunity
The study points out that SARS-CoV-2 constantly mutates to dodge antibodies, especially on outer parts of the N-terminal domain.
But it never alters the triad, likely because breaking it would stop the shape-shift that makes infection possible. In simple terms, the virus can swap decorations on the outside but cannot afford to touch its internal gears.
Closing Thoughts
This
Medical News report highlights how evolution has preserved a tiny but vital three-amino-acid gear in the spike machine. The review suggests that blocking this triad directly—or breaking the “wave” linking it to the RBD—could stop infection at step one. In conclusion, understanding and exploiting this invariant molecular switch may finally hand researchers a universal weak point shared across all major variants. If the triad can be disrupted, the spike may never fully activate, potentially halting viral entry regardless of future mutations. Such a strategy could form the basis for next generation drugs or vaccines built to stay effective long after the virus evolves again.
The study findings were published in the peer reviewed journal: Biomolecules.
https://www.mdpi.com/2218-273X/16/1/111
For the latest COVID-19 news, keep on logging to Thailand
Medical News.
Read Also:
https://www.thailandmedical.news/articles/coronavirus
