COVID-19 Research: SARS-CoV-2 Infection Causes Alterations In Gut Immunological Barrier!
COVID-19 Research: Alterations In Gut Immunological Barrier Mar 16, 2023 3 years, 1 month, 2 weeks, 5 days, 19 hours, 46 minutes ago
COVID-19 Research: A new study by researchers from University of Patras-Greecee, University Hospital of Patras-Greece and “Agios Andreas” Patras State General Hospital-Greece has found that SARS-CoV-2 infections causes alterations in gut immunological barrier.
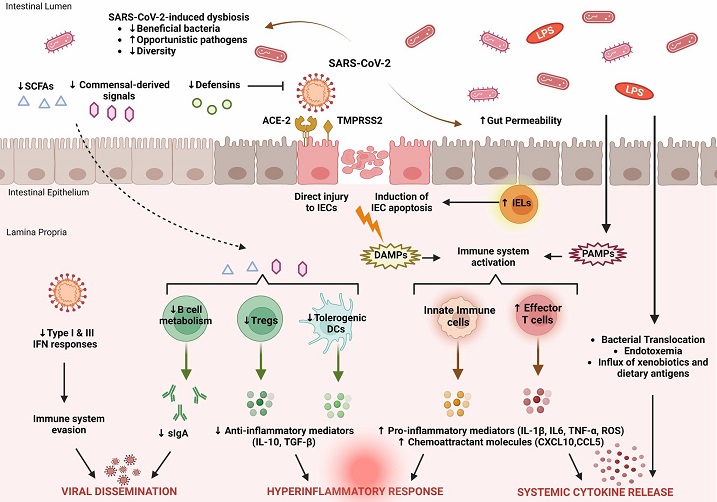 Key features of intestinal immune barrier disruption during SARS-CoV-2 infection. SARS-CoV-2 infection is associated with profound alterations in the intestinal microflora, manifested by decreased species diversity, depletion of symbiotic microorganisms, and prevalence of pathogenic species. Signals and metabolites derived from the intestinal flora, such as short-chain fatty acids (SCFAs), play an important role in controlling mucosal immunity by promoting T regulatory cell (Treg) responses and the activity of tolerogenic dendritic cells (DCs). This immunoregulatory environment, rich in anti-inflammatory mediators (IL-10, TGF-β), is significantly impaired by SARS-CoV-2. As a result, B cell metabolism and maturation are severely impaired, leading to exhaustion of effective plasma cells that produce secretory dimeric immunoglobulin A (sIgA), which is essential for viral containment. The proliferation of SARS-CoV-2 is also facilitated by its ability to evade recognition by the immune system by interfering with type I and type III IFN signaling. SARS-CoV-2 exerts either direct cytopathic effects on intestinal epithelial cells (IECs) expressing ACE2 and TMPRSS2 receptors or indirect immune-mediated injury. During COVID-19, the expression of several antimicrobial peptides, including defensins, is dysregulated, which increases the infectivity of SARS-CoV-2. In addition, recruitment of intraepithelial lymphocytes (IELs) accelerates IEC apoptosis. The release of damage-associated molecular patterns (DAMPs) due to cell injury and the influx of pathogen-associated molecular patterns (PAMPs) as a result of increased gut permeability lead to immune activation. Macrophages/monocytes, neutrophils, and other cells of the innate immune system secrete large amounts of proinflammatory mediators (IL-1β, IL-6, TNF-α, ROS) and chemokines (CCL5, CXCL10) that cause recruitment of additional immune cells and prime effector T cells. In parallel, disruption of the intestinal barrier facilitates bacterial translocation, endotoxemia, and dissemination of other gut-derived stimuli that contribute to systemic hyperinflammatory responses and cytokine release syndrome, leading to severe COVID-19.
Key features of intestinal immune barrier disruption during SARS-CoV-2 infection. SARS-CoV-2 infection is associated with profound alterations in the intestinal microflora, manifested by decreased species diversity, depletion of symbiotic microorganisms, and prevalence of pathogenic species. Signals and metabolites derived from the intestinal flora, such as short-chain fatty acids (SCFAs), play an important role in controlling mucosal immunity by promoting T regulatory cell (Treg) responses and the activity of tolerogenic dendritic cells (DCs). This immunoregulatory environment, rich in anti-inflammatory mediators (IL-10, TGF-β), is significantly impaired by SARS-CoV-2. As a result, B cell metabolism and maturation are severely impaired, leading to exhaustion of effective plasma cells that produce secretory dimeric immunoglobulin A (sIgA), which is essential for viral containment. The proliferation of SARS-CoV-2 is also facilitated by its ability to evade recognition by the immune system by interfering with type I and type III IFN signaling. SARS-CoV-2 exerts either direct cytopathic effects on intestinal epithelial cells (IECs) expressing ACE2 and TMPRSS2 receptors or indirect immune-mediated injury. During COVID-19, the expression of several antimicrobial peptides, including defensins, is dysregulated, which increases the infectivity of SARS-CoV-2. In addition, recruitment of intraepithelial lymphocytes (IELs) accelerates IEC apoptosis. The release of damage-associated molecular patterns (DAMPs) due to cell injury and the influx of pathogen-associated molecular patterns (PAMPs) as a result of increased gut permeability lead to immune activation. Macrophages/monocytes, neutrophils, and other cells of the innate immune system secrete large amounts of proinflammatory mediators (IL-1β, IL-6, TNF-α, ROS) and chemokines (CCL5, CXCL10) that cause recruitment of additional immune cells and prime effector T cells. In parallel, disruption of the intestinal barrier facilitates bacterial translocation, endotoxemia, and dissemination of other gut-derived stimuli that contribute to systemic hyperinflammatory responses and cytokine release syndrome, leading to severe COVID-19.
SARS-CoV-2 can directly infect the gastrointestinal tract or cause indirect damage through the release of inflammatory mediators. A key factor in the severity of the disease is the impaired intestinal barrier function, which allows excessive microbial and endotoxin translocation, leading to a strong systemic immune response and the development of sepsis syndrome.
The gut immune system is negatively impacted by the virus, resulting in a decreased or dysfunctional gut immunological barrier.
SARS-CoV-2 infection leads to impairment of the gut barrier in several ways, including dysregulation of the gut microbiota, damage to the intestinal epithelial cells, and disruption of tight junction homeostasis. This multifactorial impairment of the gut barrier can result in bacterial and fungal translocation and subsequent systemic inflammation, which can worsen COVID-19 severity.
Antiviral peptides, imm
une cell chemotaxis, and secretory immunoglobulins are among the important parameters that are affected. The activation of immune cells such as CD4+ and CD8+ T cells, Th17 cells, neutrophils, dendritic cells, and macrophages, as well as the decrease in regulatory T cells, leads to an overactive immune response and increased expression of proinflammatory cytokines.
The changes in the gut's immunological barrier can be further exacerbated by a dysbiotic gut microbiota, which can promote proinflammatory signals and metabolites. Conversely, the proinflammatory intestinal environment can also disrupt the intestinal epithelium and promote enterocyte apoptosis and tight junction disruption.
The
COVID-19 Research findings were published in the peer reviewed journal:
Frontiers In Immunogy.
https://www.frontiersin.org/articles/10.3389/fimmu.2023.1129190/full
The Alterations To The Immunological Barrier In The Intestines Caused By SARS-CoV-2.
Intestinal Inflammation And Fecal Calprotectin
Fecal calprotectin, a marker of bowel inflammation produced by activated neutrophils in the intestine, has been extensively studied in COVID-19 patients. It is a reliable marker for detecting inflammation in conditions such as inflammatory bowel disease. High levels of fecal calprotectin in SARS-CoV-2 infected individuals are associated with an increased risk of COVID-19 pneumonia and are positively correlated with other markers of disease severity. There is no correlation between gastrointestinal symptoms and fecal calprotectin, but it has been found to be a better predictor of disease progression than C-reactive protein. The increase in fecal calprotectin in COVID-19 patients may be due to immune cell migration and hypoxic intestinal damage, rather than intestinal inflammation and enterocyte destruction. Serum calprotectin has also been found to be a strong predictor of poor clinical outcomes in COVID-19 patients, highlighting its potential value as a diagnostic and prognostic marker.
Alterations In Immune Cells
Research has shown changes in immune cells in the gastrointestinal mucosa of patients with COVID-19, with leukocytic infiltration of various cells including monocytes, macrophages, dendritic cells, natural killer cells, and B cells. Similar results were obtained from lung tissue, indicating that these organs are the primary location of the immune response during SARS-CoV-2 infection. In addition, an increase in certain immune cells was observed in intestinal biopsies, even a month after infection. Dysregulation of CD4+ T cells in the gut, leading to intestinal barrier dysfunction and leaky gut that promotes systemic inflammation, is also observed in COVID-19, similar to HIV infection. This dysregulation results in overactivation of IL-17 producing Th17 cells.
Dysregulation Of Interferon Responses
When SARS-CoV-2 infects intestinal cells, it triggers an increase in proinflammatory cytokines, but the virus can inhibit the formation of a complex responsible for activating IFN transcription and synthesis. Pre-treating human intestinal cells and colon organoids with IFN-β and type III IFN provides protection against SARS-CoV-2 infection, with the latter being more effective. Depletion of the type III IFN receptor increases SARS-CoV-2 infectivity, viral genome replication, and virion production. Pre-existing neutralizing autoantibodies to type I IFN increase the risk of severe COVID-19, and loss-of-function variants of certain genes can also lead to severe symptoms.
Changes In Other Inflammatory Mediators
When SARS-CoV-2 infects the nasal cavity, several cytokines and inflammatory mediators are initially produced in the gastrointestinal tissues, including IL-4, IL-1β, TNF-α, and IL-17A. In addition, infection in the digestive tract can lead to upregulation of the anti-inflammatory IL-10 and inhibition of the pro-inflammatory IL-1β and IFN-γ. Evidence suggests that several cytokines, including IL-6, CXCL10, and CCL5, are increased in gut-on-a-chip models and pluripotent stem cells derived from small intestinal epithelial cells or intestinal organoids. Stool samples from hospitalized COVID-19 patients show higher levels of IL-8 and IL-18 and lower levels of IL-10. Treatment with short-chain fatty acids can reduce virus-enterocyte fusion, and remdesivir administration to infected intestinal epithelial cells can reduce cytokine gene induction. Soluble mucosal addressin cell adhesion molecule (sMAdCAM) is inversely correlated with serum IL-6 levels during SARS-CoV-2 infection and may be a useful marker for monitoring mucosal homeostasis and guiding therapy.
Disruption Of Antimicrobial Peptide Production
The gastrointestinal tract has an antiviral response involving Paneth cells and neutrophils, which produce defensins. Defensins have antimicrobial and antiviral effects, serve as chemo-attractants and activators for immune cells. α-defensin 5 protects the ACE2 receptor and prevents SARS-CoV-2 binding, and α-defensin 5 levels are elevated before infection. Intestinal hypoxia increases β-defensin 1 production in later stages of SARS-CoV-2 infection.
Dysregulation Of Secretory IgA Production
The gut's immune system is strengthened by sIgA, which is produced by mucosal lymphoid tissues like GALT. Commensal microorganisms play a crucial role in regulating the production of effective antibodies and class switching of IgA. The intestine's sIgA predominance is likely why there is less inflammation compared to lung tissue. Dimeric IgA was found to be more effective than IgA monomers or IgG in neutralizing SARS-CoV-2 in an in vitro study of COVID-19 convalescent patients.
SARS-Cov-2-Mediated Gut Microbiome And Immunological Changes
The balance between beneficial and pathogenic bacteria in the gut is important for immune function. There are significant alterations in the gut microbiome observed in patients with COVID-19, but the underlying mechanisms are still unclear. Dysbiosis in the gut microbiota during COVID-19 includes a reduction in the ratio of Firmicutes to Bacteroidetes and fewer butyrate-producing bacterial species. Several beneficial genera are also reduced, and alterations in the gut microbiome are closely associated with the clinical severity of COVID-19. However, emerging evidence suggests that the regulatory functions of the gut microbiota effectively support recovery from SARS-CoV-2 infection.
Conclusion
SARS-CoV-2 infection disrupts intestinal immunological homeostasis, impairing mucosal immune cell function and production of signaling molecules. Gut dysbiosis caused by SARS-CoV-2 could drive these changes through commensal-derived signals and metabolites. Dysregulation of intestinal immune cells and proinflammatory cytokine production can compromise intestinal epithelial cell integrity and their connections, leading to gut barrier dysfunction. This can lead to the translocation of microbes and endotoxins into the systemic circulation, causing a hyperinflammatory response and distant organ dysfunction. Gut immunologic barrier failure occurs early in the course of COVID-19 and correlates with disease severity, making it a potential prognostic marker. Further studies are needed to explore the role of immunologic therapies in improving gut barrier function and expanding therapeutic options against COVID-19.
For the latest
COVID-19 Research, keep on logging to Thailand Medical News.
