Glaucoma News: Study Finds That Long Non-Coding RNA KCNQ1OT1 Accelerates Glaucoma Progression Via microRNA-93-5p/ Homeobox Box 3 Axis
Nikhil Prasad Fact checked by:Thailand Medical News Team Nov 01, 2023 2 years, 7 months, 4 days, 12 hours, 55 minutes ago
Glaucoma News: Glaucoma, a neurodegenerative eye disease characterized by the gradual loss of retinal ganglion cells (RGCs) and their axons, is a leading cause of irreversible vision loss worldwide. The rising global incidence of glaucoma has prompted an urgent need for better diagnostic biomarkers and a deeper understanding of the disease's molecular mechanisms. In recent years, research has increasingly focused on long non-coding RNAs (lncRNAs), which have been implicated in various biological processes and shown to play a role in diseases such as cancer and diabetes. This
Glaucoma News report covers a study by researchers from The First Affiliated Hospital of Gannan Medical College, Ganzhou City-China, which delves into the role of the lncRNA KCNQ1 opposite strand/antisense transcript 1 (KCNQ1OT1) in glaucoma, aiming to shed light on its potential as a diagnostic biomarker and its involvement in the progression of this debilitating eye condition.
 Graphical Abstract
The Prevalence and Challenges of Glaucoma
Graphical Abstract
The Prevalence and Challenges of Glaucoma
Glaucoma poses a significant public health challenge as it often remains undetected until vision loss becomes irreversible. This lack of early symptoms and reliable diagnostic markers has led to a situation where glaucoma is commonly diagnosed at advanced stages, making treatment less effective. Intraocular pressure (IOP) is currently the only modifiable risk factor for glaucoma, and treatments primarily focus on reducing IOP through medications, laser therapy, and, when necessary, surgical interventions. While these approaches have seen significant advancements, there are currently no foolproof methods for preventing the onset of glaucoma or reversing its progression. Furthermore, diagnosing glaucoma primarily relies on subjective judgments through optic nerve examination and ultrasonic biological microscopy, both of which are costly and operator-dependent.
Emerging Role of Long Non-Coding RNAs in Glaucoma
Long non-coding RNAs (lncRNAs) are a class of endogenous non-coding RNAs that are over 200 nucleotides in length. While they do not code for proteins, lncRNAs have been shown to modulate gene expression through various mechanisms, including epigenetic, translational, and transcriptional regulation. Research has revealed their involvement in processes such as cell proliferation, apoptosis, immune responses, and oxidative stress. Specifically, the lncRNA MALAT1 has been implicated in retinal ganglion cell apoptosis in glaucoma via the phosphatidylinositol 3-kinase (PI3K)/Akt signaling pathway. However, the role of KCNQ1OT1 in glaucoma remains largely unexplored.
KCNQ1OT1: A Potential Biomarker for Glaucoma
KCNQ1OT1, located on huma
n chromosome 11p15.5, has previously been associated with various diseases, including eye conditions. Initial research linked KCNQ1OT1 to diabetic complications such as diabetic retinopathy and diabetic cardiomyopathy.
Subsequent studies demonstrated its role in promoting pathological mechanisms in diabetic endothelial cornea and stimulating lens epithelial cell apoptosis and oxidative stress. Additionally, KCNQ1OT1 has been shown to enhance the migration of corneal epithelial cells and expedite corneal wound healing. However, its expression and function in glaucoma have not been thoroughly investigated.
This study set out to examine KCNQ1OT1 levels in glaucoma patients and explore its potential as a diagnostic marker. The results revealed that KCNQ1OT1 expression was elevated in glaucoma patients and correlated with clinicopathological indicators. Furthermore, elevated KCNQ1OT1 was observed in a rat model of glaucoma induced by N-methyl-D-aspartate (NMDA) and in an in vitro model of retinal ganglion cells (RGCs). These findings suggest that KCNQ1OT1 could serve as a promising biomarker for glaucoma.
KCNQ1OT1's Role in Glaucoma Progression
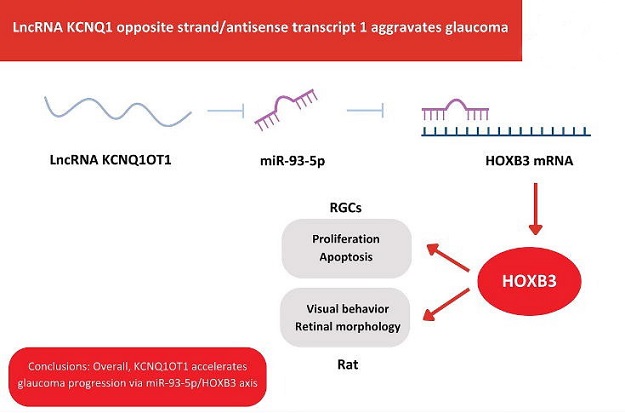
In the in vivo model, rats injected with NMDA experienced visual impairment, increased intraocular pressure (IOP), accelerated RGC apoptosis, and upregulated KCNQ1OT1 expression over time. Silencing KCNQ1OT1 resulted in improved visual function in these glaucoma rats. In the in vitro model, NMDA treatment led to a gradual increase in KCNQ1OT1 expression in RGCs. Silencing KCNQ1OT1 mitigated the negative effects of NMDA on RGC proliferation and altered the expression of key apoptosis-related genes, such as Bcl-2 and Bax.
MicroRNAs (miRNAs) are known to play crucial roles in glaucoma, with several studies highlighting their involvement in the condition. An earlier study identified a panel of differentially expressed miRNAs in glaucoma optic neuropathy, including miR-93-5p. MiR-93-5p has been found to modulate NMDA-induced RGC autophagy via the Akt/mammalian target of rapamycin (mTOR) pathway and inhibit retinal neuronal apoptosis in acute ocular hypertension models. In the present research, miR-93-5p was downregulated in glaucoma, and its elevation was shown to alleviate the inhibitory effects of NMDA on RGC growth.
Interestingly, miR-93-5p was found to interact with homeobox box 3 (HOXB3).
HOXB3, a transcription factor belonging to the HOX gene family, is well-known for its role in embryonic development. Research on HOXB3 has primarily focused on its implications in cancer, such as cervical squamous cell carcinoma, colon cancer, and pancreatic cancer. Recent studies have also linked HOXB3 to eye diseases. In this study, HOXB3 expression was found to be elevated in NMDA-treated RGCs. Additionally, increasing HOXB3 levels reversed the effects of silenced KCNQ1OT1 on RGCs. These findings highlight the existence of a novel regulatory pathway involving KCNQ1OT1, miR-93-5p, and HOXB3 in the progression of glaucoma.
Limitations and Future Directions
While this study provides valuable insights into the potential role of KCNQ1OT1 as a diagnostic biomarker and its involvement in glaucoma progression, it is not without limitations. One notable limitation is the relatively small sample size of clinical specimens used in the study. To further validate these findings and strengthen their clinical relevance, larger-scale studies are necessary. Additionally, future research should explore the clinical applications of KCNQ1OT1 in diagnosing and treating glaucoma.
Conclusion
In summary, this study presents compelling evidence for the elevation of KCNQ1OT1 in glaucoma patients, suggesting its potential as a diagnostic biomarker. The research also elucidates the role of KCNQ1OT1 in glaucoma progression, both in vivo and in vitro, by modulating the miR-93-5p/HOXB3 axis. These findings open new avenues for the development of diagnostic tools and therapeutic targets in the fight against glaucoma, a condition that continues to challenge the field of ophthalmology. Future research endeavors may uncover additional facets of this complex disease and further validate the clinical utility of KCNQ1OT1.
The study findings were published in the peer reviewed journal: Electronic Journal of Biotechnology.
https://www.sciencedirect.com/science/article/pii/S0717345823000349
For the latest
Glaucoma News, keep on logging to Thailand Medical News.
