University of Manchester Study Reveals Sustained Monocyte Dysfunction As A Key Feature Of Acute COVID-19 Infections And Long COVID
Thailand Medical News Team Aug 14, 2023 2 years, 9 months, 2 weeks, 4 days, 9 hours, 4 minutes ago
COVID-19 News: The world has been grappling with the unprecedented challenges posed by the COVID-19 pandemic, a global health crisis of monumental proportions that has led to over 768 million infections and a staggering 6.9 million deaths worldwide, as of the latest count. As our understanding of the SARS-CoV-2 virus and its impact on human health has evolved, researchers at the University of Manchester - UK, have unveiled a groundbreaking study that sheds light on a previously unexplored facet of COVID-19: the persistent dysfunction of monocytes in both acute infections and the enigmatic phenomenon known as "Long COVID."
Intriguingly, the study suggests that the immune system, particularly the behavior of monocytes, plays a pivotal role not only in the acute phase of COVID-19 but also in the lingering aftermath that characterizes Long COVID. Monocytes, a type of white blood cell, are a crucial component of the body's innate immune response, responsible for patrolling the bloodstream and infiltrating tissues to combat pathogens and promote healing.
The study's origins lie in the growing realization that COVID-19 is more than just a respiratory ailment; it is a disease that can have far-reaching effects on various organs and systems within the body as covered in various studies and
COVID-19 News reports.
While the acute phase of the illness may present with a range of symptoms, from mild flu-like malaise to severe respiratory distress, the aftermath, known as Long COVID, has emerged as a complex syndrome characterized by a constellation of chronic symptoms, including extreme fatigue, breathlessness, myalgia, cognitive impairments ("brain fog"), and mood disturbances like depression.
To unravel the enigma of Long COVID, the study team embarked on an ambitious journey, aiming to dissect the intricate workings of the immune system during and after COVID-19 infection. Their comprehensive study, known as the Coronavirus Immune Response and Clinical Outcomes (CIRCO) study, involved the collection of blood samples from COVID-19 patients at various stages of their illness, as well as from those who had recovered, up to a remarkable nine months post-hospitalization.
The study team’s efforts culminated in a revelation that could transform our understanding of COVID-19 pathophysiology: monocytes, the unsung heroes of the immune system, exhibit sustained dysfunction both during the acute phase of the disease and into the convalescent period. These abnormalities, the study shows, are intricately linked to the development of Long COVID.
Monocytes, typically dispatched from the bone marrow into the bloodstream, play a vital role in responding to respiratory infections by migrating to the lungs. This study builds upon previous research demonstrating heightened systemic levels of monocyte-chemoattractant protein 1 (MCP-1) in COVID-19 patients, indicating their rapid release from the bone marrow. The study team also observed abnormal monocyte migration to peripheral tissues in COVID-19, reinforcing the notion that these immune cells are intimately involved in the disease process.
Central to the
study's findings is the identification of distinct monocyte migration profiles associated with specific features of Long COVID. For patients with unresolved lung injury and persistent shortness of breath, monocytes exhibited heightened expression of certain molecules, such as C-X-C motif chemokine receptor 6 (CXCR6) and adhesion molecule P-selectin glycoprotein ligand 1 (PSGL-1), alongside a preference for migrating towards the CXCR6 ligand C-X-C motif chemokine ligand 16 (CXCL16), which is abundantly expressed in lung tissue.
This aberrant monocyte behavior, the researchers note, mirrors patterns observed in patients with progressive fibrosing interstitial lung disease, providing a crucial link between monocyte dysfunction and ongoing lung injury.
Conversely, Long COVID patients experiencing debilitating fatigue displayed a different set of monocyte abnormalities. These individuals exhibited a sustained reduction in the prostaglandin-generating enzyme cyclooxygenase 2 (COX-2) and CXCR2 expression. These changes, unique to Long COVID patients, were absent in convalescent individuals recovering from respiratory syncytial virus (RSV) or influenza A infections.
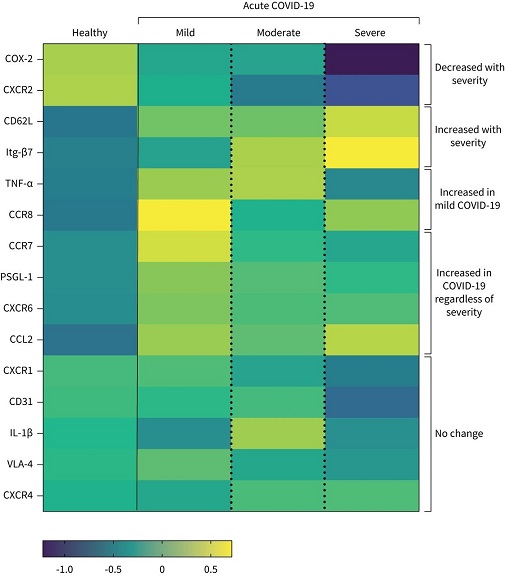 Summary of distinct monocyte profiles in patients with acute COVID-19. Heatmap of indicated immune parameters by row. Each column represents average (mean) Z-scores for each parameter calculated from individual values of expression by monocytes from healthy individuals or patients with mild, moderate or severe COVID-19 as separate groups. Heatmap was generated as a visual guide, using a subset of individuals from each group to include all individuals per group with results for every immune parameter analysed (i.e. individuals where samples were used for surface staining for all migration molecules, and microbial stimulation assays for cytokine and COX-2 readouts). Healthy individuals, n=25; mild COVID-19, n=13; moderate COVID-19, n=14; severe COVID-19, n=7.
Summary of distinct monocyte profiles in patients with acute COVID-19. Heatmap of indicated immune parameters by row. Each column represents average (mean) Z-scores for each parameter calculated from individual values of expression by monocytes from healthy individuals or patients with mild, moderate or severe COVID-19 as separate groups. Heatmap was generated as a visual guide, using a subset of individuals from each group to include all individuals per group with results for every immune parameter analysed (i.e. individuals where samples were used for surface staining for all migration molecules, and microbial stimulation assays for cytokine and COX-2 readouts). Healthy individuals, n=25; mild COVID-19, n=13; moderate COVID-19, n=14; severe COVID-19, n=7.
The implications of this study are profound. It not only underscores the complex interplay between monocytes and the immune response during COVID-19 but also provides a novel lens through which to view Long COVID. By identifying specific monocyte signatures associated with different Long COVID symptoms, the researchers have paved the way for targeted therapeutic strategies aimed at alleviating the chronic and often debilitating effects of the syndrome.
As the world grapples with new variants of the SARS-CoV-2 virus, such as the Omicron variant, the insights from this study remain highly relevant. Despite changes in viral variants, the underlying immunological pathophysiology and the role of monocyte dysfunction appear to persist, making these findings pertinent for future waves of the pandemic.
The study from the University of Manchester and the Manchester Academic Health Science Centre marks a significant step forward in unraveling the mysteries of COVID-19 and Long COVID. By shining a spotlight on the crucial role of monocytes and their sustained dysfunction, researchers have illuminated a path towards targeted interventions that could provide much-needed relief for those grappling with the persistent aftermath of the virus. As the global scientific community continues to work tirelessly to understand and combat the COVID-19 pandemic, studies like this one offer hope for a brighter future, where the enigma of Long COVID can be decoded and its impact mitigated.
The study findings were published in the peer reviewed European Respiratory Journal
https://erj.ersjournals.com/content/61/5/2202226
For the latest
COVID-19 News, keep on logging to Thailand Medical News

