Glaucoma News: German Study Shows That HSP27 Protein Plays A Role In The Development Of Glaucoma!
Nikhil Prasad Fact checked by:Thailand Medical News Oct 16, 2023 2 years, 7 months, 2 weeks, 4 days, 20 hours, 28 minutes ago
Glaucoma News: Glaucoma, a progressive optic neuropathy, is a leading cause of blindness when left untreated. It is often associated with elevated intraocular pressure (IOP) and primarily affects the elderly. However, recent research has shown that factors beyond IOP, including immunological factors such as heat shock proteins (HSPs), may contribute to the development of the disease. This
Glaucoma News report delves into a German study by researchers from University Eye Hospital, Ruhr-University Bochum-Germany that explores the role of the HSP27 protein in the development of glaucoma and the potential age-related effects on this process.
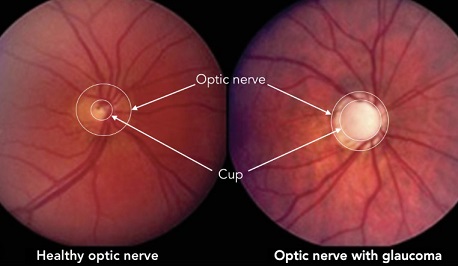
A past study made a significant discovery when they found elevated autoantibody titers against heat shock protein 60 (HSP60) in the serum of individuals with normal-tension glaucoma (NTG), a subtype of glaucoma characterized by typical damage to the optic nerve and RGCs without elevated IOP. This finding led to further investigations into the role of HSPs in glaucoma.
https://www.jneurosci.org/content/28/46/12085.short
https://journals.lww.com/glaucomajournal/citation/2001/10001/serum_autoantibodies_in_patients_with_glaucoma.9.aspx
HSPs are a family of proteins that act as chaperones under normal conditions and have anti-apoptotic effects. They are induced in response to stress and play a crucial role in protecting cells from the harmful effects of various stressors. HSP27, a small HSP, is one member of this family. While HSPs are primarily intracellular, they can also be found extracellularly, acting as stress signals that activate the immune system. Several studies have shown that elevated levels of antibodies against HSPs, including HSP27, are present in glaucoma patients compared to healthy individuals.
The Role of HSP27 in Glaucoma
The effect of HSP27 on retinal cells was investigated by applying antibodies against HSP27 to isolated human retinae. This research revealed that HSP27 induces apoptosis in retinal cells, especially RGCs, which are critical for transmitting visual information from the retina to the brain. Subsequent animal studies, using a rat model, showed glaucoma-like damage after immunization with HSP27. The loss of RGCs and alterations in IgG antibody patterns in the serum were observed.
To better understand the effects of HSP27, a local, intravitreal injection of HSP27 was administered to rats. This approach aimed to mimic the effects of HSP27 in a controlled environment. The results were significant: the rats exhibited glaucoma-like damage, including RGC and amacrine cell degeneration, and optic nerve deterioration.
Moreover, this study analyzed the signaling pathways responsible for these effects. It found the activation of intrinsic and extrinsic apoptosis pathways, an increase in nuclear factor-kappa-light-chain-enhancer of activated B cells (NF-κB), and activation of microglia and T-cells. These findings indicated that extracellular HSP27 had degenerative effects and co
ntributed to the development of glaucoma.
Investigating Age-Related Effects
Glaucoma is an age-related neurodegenerative disease, and inflammation is known to be a part of the disease process. Aging is characterized by a progressive deterioration of cellular and tissue functions and is accompanied by immunological changes. These age-related changes may lead to an increased susceptibility to age-related diseases, including glaucoma. Processes such as oxidative stress, mitochondrial dysfunction, and cellular senescence play a role in making eye cells more susceptible to damage, which can ultimately lead to RGC loss.
In the context of glaucoma, microglia play a pivotal role. These resident immune cells in the central nervous system, including the retina, respond to pathological stimuli and migrate to sites of injury. Their activation can lead to the production of various enzymes and cytokines. Furthermore, microglia interact with macroglia, such as astrocytes and Müller cells, which are crucial for maintaining the structural and functional integrity of the retina.
While glaucoma is primarily an age-related disease, the effects of age on an HSP27 injection had not been previously studied. This research sought to fill that gap by intravitreally injecting HSP27 into both young (1–2 months) and aged (7–8 months) mice. The mice were examined four weeks after injection, and their retinas and optic nerves were evaluated to assess the potential age-dependent effects of HSP27 on glaucoma development.
Findings of the Study
The study found that HSP27 injection resulted in a loss of RGCs and optic nerve degeneration in both young and aged mice. However, the age-dependent effects were less prominent than expected. While the inflammatory response was more pronounced in aged mice, the degenerative effects of HSP27 were comparable between the two age groups. This suggests that HSP27-induced glaucoma is not strongly age-dependent in this model.
The loss of RGCs, a hallmark of glaucomatous degeneration, was confirmed in the study through immunohistological staining and gene expression analysis. Furthermore, neuronal cells, as a whole, were negatively impacted, with a decrease in the expression of Tubb3, a marker for neuronal cells. These findings suggest that HSP27's detrimental effects extend beyond RGCs, affecting various neuronal cells.
The activation of microglia and macroglia was observed in response to HSP27 injection. Microglia, the resident immune cells in the retina, were activated, and this response was more prominent in the early phases of the disease. The study also noted the activation of astrocytes, indicated by increased expression of glial fibrillary acidic protein (GFAP). This reactive gliosis, a response to retinal stress, is a known mechanism in glaucoma patients.
Inflammatory markers, such as interleukin 1 beta (IL-1β) and inducible nitric oxide synthase (iNOS), were upregulated in response to HSP27 injection. IL-1β, an inflammatory cytokine, was more prominently upregulated in aged mice compared to their younger counterparts, indicating age-related effects on the immune response. iNOS, an enzyme involved in immune responses, showed significant changes only when comparing aged HSP27 mice to naive mice, further suggesting an age-related susceptibility to inflammation.
Moreover, the study observed changes in the expression of HSP27 and its related protein, Hspb2. Extracellular HSP27 levels were increased in aged mice, while Hspb2 expression was downregulated in both age groups. The mechanisms by which HSP27 induces glaucoma-like damage require further investigation, particularly regarding its extracellular effects and interactions with immune responses.
Conclusion
The results of this study suggest that HSP27 is involved in the pathogenesis of glaucoma. While there were age-related effects, they were relatively minor, particularly with respect to the degenerative effects of HSP27. The findings indicate that young mice can be used effectively to study the underlying pathomechanisms of HSP27-induced glaucoma and develop potential therapeutic approaches for glaucoma patients in the future.
In conclusion, this German study sheds light on the role of HSP27 in glaucoma, exploring its detrimental effects on retinal cells, the activation of immune responses, and the less prominent age-related effects. By understanding the involvement of HSP27 and related processes, researchers aim to develop new therapeutic strategies that target these pathways and ultimately help prevent or slow the progression of glaucoma.
The study findings were published in the peer reviewed journal: Frontiers in Cellular Science.
https://www.frontiersin.org/articles/10.3389/fncel.2023.1257297/full
For the latest
Glaucoma News, keep on logging to Thailand Medical News.
Disclaimer: This article is a summary and analysis of a scientific study and should not be used as a substitute for professional medical advice as with all articles in this website. If you have concerns about glaucoma or its treatment, please consult a credible licensed healthcare professional who has the relevant knowledge and training.
