COVID-19 News: Scientists From Chile Find That SARS-CoV-2 Spike Protein S1 Activates Cx43 Hemichannels And Disturbs Intracellular Ca2+ Dynamics
Nikhil Prasad Fact checked by:Thailand Medical News Team Oct 27, 2023 2 years, 7 months, 1 week, 4 days, 10 hours, 21 minutes ago
COVID-19 News: The ongoing battle against the coronavirus disease 2019 (COVID-19) pandemic has been characterized by relentless research and discovery. Scientists and researchers worldwide have been working diligently to uncover the mysteries of the virus, SARS-CoV-2, and its various components. Among these, the spike protein S1, a crucial element of the virus, has drawn significant attention. A recent study conducted by scientists from the Pontificia Universidad Católica de Chile, Universidad Autónoma de Chile, and Universidad del Desarrollo-Chile, has shed new light on how the SARS-CoV-2 spike protein S1 affects cellular function, potentially influencing the severity and long-term consequences of COVID-19.
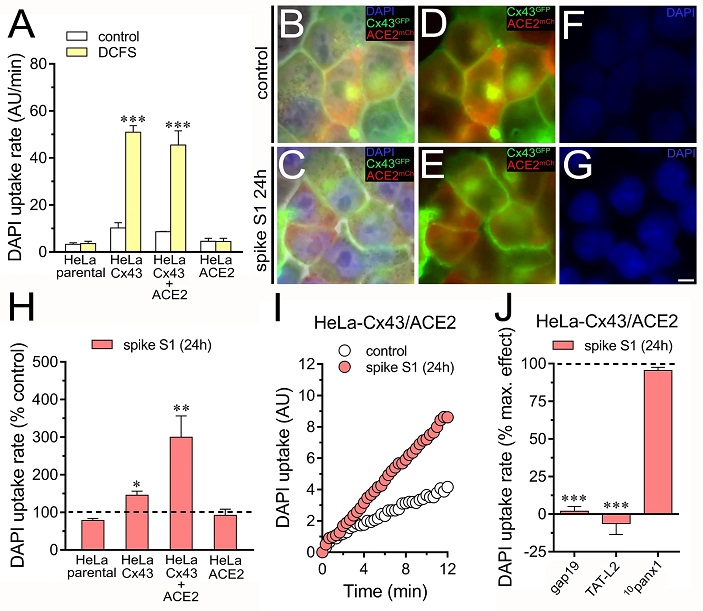 The activation of Cx43 hemichannels induced by spike S1 is potentiated by ACE2. (A) Averaged DAPI uptake rate by HeLa-parental, HeLa-Cx43GFP, HeLa-Cx43GFP/ACE2mCherry or HeLa-ACE2mCherry cells bathed with normal saline (control, white bars) or a divalent cation-free solution (DCFS, yellow bars). ***p < 0.001; effect of DCFS compared to control (two-way ANOVA followed by Tukey’s post-hoc test). (B-C) Representative images depicting the phase view merged with Cx43GFP (green), ACE2mCherry (red), and DAPI (blue, 10 µM and 10 min of exposure) labeling by HeLa-Cx43GFP/ACE2mCherry cells under control conditions (B) or treated with 50 pM spike S1 for 24 h (C). (D-E) Cx43GFP and ACE2mCherry labeling by Cx43GFP/ACE2mCherry cells showed in B and C. (F-G) DAPI staining by Cx43GFP/ACE2mCherry cells shown in B and C. (H) Averaged DAPI uptake rate normalized with the control condition (dashed line) by HeLa-parental, HeLa-Cx43GFP, HeLa-Cx43GFP/ACE2mCherry or HeLa-ACE2mCherry cells treated with 50 pM spike S1 for 24 h. *p < 0.05, **p < 0.01; effect of spike S1 compared to control (one-way ANOVA followed by Tukey’s post-hoc test). (I) Time-lapse measurements of DAPI uptake by HeLa-Cx43GFP/ACE2mCherry cells under control conditions (white circles) or treated with 50 pM spike S1 for 24 h (red circles). (J) Averaged DAPI uptake rate normalized to the maximum effect evoked by spike S1 (dashed line) by HeLa-Cx43GFP cells acutely stimulated with different concentrations of spike S1. (J) Averaged DAPI uptake rate normalized with the control condition (dashed line) by HeLa-Cx43GFP/ACE2mCherry cells treated with 50 pM spike S1 for 24 h plus gap19 (100 µM), TAT-L2 (200 µM) or 10panx1 (100 µM). ***p < 0.001; effect of spike S1 vs. blockers (one-way ANOVA followed by Tukey’s post-hoc test). Data were obtained from at least three independent experiments with three or more repeats each (≥ 20 cells analyzed for each repeat). Calibration bar = 8 μm
The Impact of SARS-CoV-2 on Global Health
The activation of Cx43 hemichannels induced by spike S1 is potentiated by ACE2. (A) Averaged DAPI uptake rate by HeLa-parental, HeLa-Cx43GFP, HeLa-Cx43GFP/ACE2mCherry or HeLa-ACE2mCherry cells bathed with normal saline (control, white bars) or a divalent cation-free solution (DCFS, yellow bars). ***p < 0.001; effect of DCFS compared to control (two-way ANOVA followed by Tukey’s post-hoc test). (B-C) Representative images depicting the phase view merged with Cx43GFP (green), ACE2mCherry (red), and DAPI (blue, 10 µM and 10 min of exposure) labeling by HeLa-Cx43GFP/ACE2mCherry cells under control conditions (B) or treated with 50 pM spike S1 for 24 h (C). (D-E) Cx43GFP and ACE2mCherry labeling by Cx43GFP/ACE2mCherry cells showed in B and C. (F-G) DAPI staining by Cx43GFP/ACE2mCherry cells shown in B and C. (H) Averaged DAPI uptake rate normalized with the control condition (dashed line) by HeLa-parental, HeLa-Cx43GFP, HeLa-Cx43GFP/ACE2mCherry or HeLa-ACE2mCherry cells treated with 50 pM spike S1 for 24 h. *p < 0.05, **p < 0.01; effect of spike S1 compared to control (one-way ANOVA followed by Tukey’s post-hoc test). (I) Time-lapse measurements of DAPI uptake by HeLa-Cx43GFP/ACE2mCherry cells under control conditions (white circles) or treated with 50 pM spike S1 for 24 h (red circles). (J) Averaged DAPI uptake rate normalized to the maximum effect evoked by spike S1 (dashed line) by HeLa-Cx43GFP cells acutely stimulated with different concentrations of spike S1. (J) Averaged DAPI uptake rate normalized with the control condition (dashed line) by HeLa-Cx43GFP/ACE2mCherry cells treated with 50 pM spike S1 for 24 h plus gap19 (100 µM), TAT-L2 (200 µM) or 10panx1 (100 µM). ***p < 0.001; effect of spike S1 vs. blockers (one-way ANOVA followed by Tukey’s post-hoc test). Data were obtained from at least three independent experiments with three or more repeats each (≥ 20 cells analyzed for each repeat). Calibration bar = 8 μm
The Impact of SARS-CoV-2 on Global Health
As the COVID-19 pandemic continues to unfold, the global community has been confronted with a virus that has already claimed an estimated 18.2 million lives. While many individuals recover fully from COVID-19, there is growing concern about those who experience persistent symptoms beyond three months of infection, commonly referred to as "long-COVID" or "post-acute COVID syndrome." These persistent symptoms encompass a wide range of physical
and neurological issues, such as fatigue, breathlessness, cognitive impairments, and more. Long-COVID is a significant public health concern and has been described as the "next health disaster."
One of the key questions that remain is whether the adverse effects observed in COVID-19 patients are solely a result of the virus's systemic inflammation or if viral proteins themselves directly impact cellular function and tissue health. This study focuses on the latter possibility, specifically examining the role of the spike protein S1 in disrupting cellular function.
Understanding the Spike Protein S1
SARS-CoV-2 is structurally composed of four main proteins, with the spike protein being the most critical for viral entry into host cells. This protein consists of two subunits, S1 and S2, with S1 responsible for binding to the host cell receptor angiotensin-converting enzyme 2 (ACE2). The S2 subunit mediates the fusion of viral and cell membranes, facilitating viral entry. Importantly, the S1 subunit can detach from the virus and enter the bloodstream, where it can interact with various tissues and potentially induce a range of effects.
Studies have shown that the presence of spike S1 in the bloodstream is associated with the severity of COVID-19 and is notably elevated in individuals experiencing long-COVID symptoms. This protein has been implicated in various health issues, including endothelial dysfunction, complement system activation, platelet aggregation, cardiac pericyte dysfunction, and blood-brain barrier disruption.
Furthermore, spike S1 can lead to cognitive deficits and anxiety-like behaviors. These findings raise the possibility that the acute and long-lasting symptoms observed in COVID-19 patients may stem from the direct actions of spike S1, although the exact mechanisms remain unclear.
The Role of Connexin Hemichannels
This study covered in this
COVID-19 News report, delves into the potential involvement of connexin hemichannels in the disruption of cellular function by spike S1. Connexin hemichannels are large-pore channels formed by transmembrane proteins called connexins. These channels play a crucial role in intercellular communication, allowing for the exchange of ions and molecules between the cytoplasm and the extracellular space. Among connexins, connexin 43 (Cx43) is the most widespread and essential in various organs and systems, contributing to processes like cell migration, cardiac excitability, and synaptic transmission.
Under normal conditions, these channels facilitate vital processes by allowing the diffusion of signaling molecules such as Ca2+, ATP, and more. However, excessive and prolonged opening of connexin hemichannels can result in the release of harmful molecules, causing problems like calcium overload and disrupted ionic balance. This study builds upon previous research that has linked the activation of connexin hemichannels to virus infections, particularly in the context of the human immunodeficiency virus (HIV) and its envelope protein (gp120).
Key Findings
The study demonstrates that spike S1 has a direct impact on the activity of Cx43 hemichannels. Importantly, the presence of ACE2 receptors, which serve as the binding sites for SARS-CoV-2, enhances the effect of spike S1 on these channels. The researchers conducted experiments involving dye uptake and patch clamp experiments to measure the activity of Cx43 hemichannels. They found that spike S1 not only increased the activity of these channels but also triggered the release of ATP and induced changes in intracellular calcium dynamics mediated by ATP.
The experiments also showed that blocking connexin hemichannels effectively counteracted the activation induced by spike S1, underscoring the specific role of these channels in the process. Furthermore, the study revealed that the spike S1-induced activation of Cx43 hemichannels coincided with a reduction in cell-cell coupling, emphasizing the potential impact on cellular function.
ATP, a key molecule in cellular energy transfer, has been previously associated with danger signaling in response to viral infections. This study demonstrates that ATP release is promoted by the spike S1 protein and is further potentiated by the presence of both Cx43 and ACE2 receptors. Importantly, the researchers noted that while spike S1 did not directly lead to cell death, it impaired cell function, suggesting that its impact is primarily on cellular processes rather than survival.
Mechanisms Behind Activation
The study suggests multiple mechanisms behind the activation of Cx43 hemichannels by spike S1. One possibility is the increase in intracellular calcium levels, as even a moderate rise in calcium concentration can enhance Cx43 hemichannel activity. Additionally, the action of nitric oxide (NO) through S-nitrosylation of Cx43 hemichannels could be another contributing factor. Spike S1 has been shown to increase the release of NO, and this molecule is known to play a role in the activation of Cx43 hemichannels during inflammatory conditions.
Significance and Implications
One intriguing aspect of this research is its potential relevance to the side effects observed in SARS-CoV-2 vaccines, particularly those utilizing mRNA technology. These vaccines have been effective in generating robust immunity against COVID-19, but there have been rare cases of adverse effects, including myocardial infarction, Bell's palsy, and myocarditis. While these side effects are not directly linked to spike S1, it is possible that the activation of Cx43 hemichannels and the downstream signaling induced by the spike protein could contribute to adverse cellular function, and further investigation is needed to clarify this potential connection.
In conclusion, this study highlights the novel finding that spike S1 from SARS-CoV-2 rapidly activates Cx43 hemichannels, leading to increased ATP release and alterations in intracellular calcium dynamics. This effect is especially potent in cells expressing high levels of ACE2 receptors, which are prominent in the cardiorespiratory system. This research unveils a previously unrecognized mechanism through which SARS-CoV-2 disrupts cellular function and sheds light on potential pharmacological targets for the development of therapies to combat SARS-CoV-2 infection and its long-term consequences, including long-COVID.
The ongoing pandemic continues to challenge our understanding of viral infections and their impact on the human body. Studies like this provide crucial insights into the intricate mechanisms underlying the disease, offering hope for improved treatments and strategies for managing its consequences. As the scientific community unravels more about the virus, it becomes increasingly important to remain vigilant and informed to protect public health.
The study findings were published in the peer reviewed journal: Biological Research.
https://biolres.biomedcentral.com/articles/10.1186/s40659-023-00468-9
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
