Glaucoma News: Calbindin 2-Specific Deletion Of Arginase 2 Preserves Visual Function After Optic Nerve Crush In Glaucoma Or Optic Neuritis!
Nikhil Prasad Fact checked by:Thailand Medical News Team Oct 13, 2023 2 years, 7 months, 2 weeks, 5 days, 11 hours, 33 minutes ago
Glaucoma News: The loss of vision resulting from damage to the optic nerve is a major concern for individuals suffering from conditions like glaucoma, traumatic optic neuropathy, or optic neuritis. These conditions can lead to the death of retinal ganglion cells (RGCs) and other retinal neurons, ultimately causing irreversible vision impairment and blindness. Understanding the precise mechanisms that underlie this neuronal cell death is crucial for developing effective treatments.
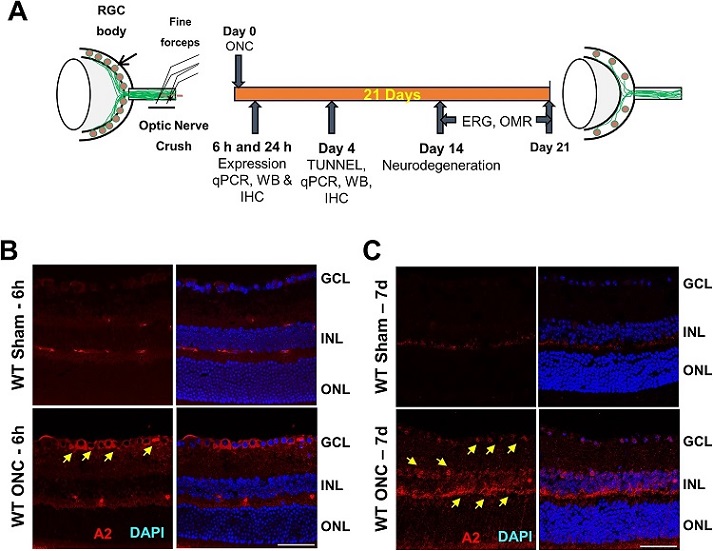 Arginase 2 expression is increased in retinal neurons after optic nerve crush.
Arginase 2 expression is increased in retinal neurons after optic nerve crush.
A Schematic representation of time-points for analyses after ONC. B, C Immunofluorescence imaging of WT retinal sections for A2 immunostaining (in red) showed increases in A2 in response to ONC at 6 h and 7 days after injury (arrows). n = 4. Scale bar = 40 μm.
One common method used to study this process is the optic nerve crush (ONC) mouse model, which mimics the effects of optic nerve injury in humans. Researchers at Augusta University in the United States have been focusing their efforts on investigating the role of the arginase pathway in retinal neurovascular injury. Arginase is an enzyme responsible for converting L-arginine into urea and ornithine. There are two main isoforms of arginase: Arginase 1 (A1) and Arginase 2 (A2), and both have been implicated in various neurological and retinal diseases.
Notably, the mitochondrial isoform, A2, has been linked to pathologies like ischemia-reperfusion (IR) injury and retinopathy of prematurity (ROP).
Previous research from this group has shown that A1 has a neuroprotective role in models of IR injury and ROP. However, A2 has demonstrated a different pattern in these studies. In response to ONC, IR, and ROP injury models, A2 levels were found to increase early on. Deletion of A2 globally has been shown to reduce neurovascular degeneration in these models, suggesting a detrimental role of A2 in retinal neurodegeneration. Still, the specific cell types and mechanisms involved remained unclear.
In this study covered in this
Glaucoma News report, the research team aimed to explore the cell-specific role of A2 in ONC-induced retinal injury by employing a variety of mouse models. Their research primarily focused on the role of A2 in RGCs and other retinal neurons expressing calbindin 2 (Calb2). Calb2 is a marker for a majority of RGCs, as well as amacrine cells and horizontal cells within the mouse retina.
A2 Expression Increases in Retinal Neurons After ONC
The study began by examining the expression of A2 in retinal neurons following ONC. Previous research had already indicated that A2 levels increase in whole retinal extracts after ONC injury within six hours. To understand which specific cell types were responsible for this increase, the researchers used immunofluorescence imaging to locate A2 in the retina. Their findings revealed a substantial rise in A2 expression i
n GCL neurons after six hours of injury, and this increased expression persisted for seven days post-injury. Furthermore, A2 immunoreactivity was also found to be significantly elevated in cells of the INL at the seven-day mark.
Calbindin 2-Specific A2 Deletion Protects Neurons After ONC
Given the strong association between A2 expression and GCL neurons, the researchers decided to investigate the role of A2 in Calb2-expressing neurons during ONC. They generated a transgenic mouse model with A2 specifically deleted in Calb2-expressing neurons, which included RGCs, amacrine cells, and horizontal cells. After subjecting these mice to ONC and analyzing neuronal survival using NeuN staining at day 14 post-injury, the results were quite striking. The A2-deletion mice showed significantly improved neuronal survival compared to the A2 floxed control mice. However, A2 deletion in myeloid or endothelial cells did not impact neuronal cell loss.
Calbindin 2-Specific A2 Deletion Preserves Visual Function
Improving neuronal survival is a significant step, but the researchers also wanted to assess the functional benefits of A2 deletion in Calb2-expressing neurons. They monitored visual function at day 14 post-ONC using optokinetic response tracking techniques. The A2-deletion mice exhibited significantly better visual acuity and contrast sensitivity compared to their A2 floxed counterparts. Additionally, the study involved pattern electroretinography (PERG) to assess ganglion cell function, and the A2-deletion mice displayed improved results.
Calbindin 2-Specific A2 Deletion Promotes Survival Signaling After ONC
Further investigation revealed that A2 deletion in Calb2-expressing neurons led to increased expression of neurotrophic factors, such as brain-derived neurotrophic factor (BDNF) and fibroblast growth factor 2 (FGF2). These factors are crucial for neuronal survival, and their increased presence suggested enhanced survival signaling in the A2-deletion mice. Indeed, the researchers found that the Akt and ERK1/2 signaling pathways, which are associated with cell survival, were significantly activated in the A2-deletion mice after ONC.
Calbindin 2-Specific A2 Deletion Limits ONC-Induced Cell Death
Cell death after ONC injury often involves DNA damage and apoptotic processes. To investigate this aspect, the study performed TUNEL staining, a method for identifying apoptotic cells, at four days post-injury. The A2-deletion mice displayed fewer TUNEL-positive GCL neurons compared to the A2 floxed controls. Moreover, they exhibited reduced cleavage of caspase 3 and poly ADP-ribose polymerase (PARP), both of which are markers of programmed cell death.
Calbindin 2-Specific A2 Deletion Reduces Activation of Müller Cells and Microglia/Macrophages and Prevents Inflammation After ONC
Following ONC, a common response in the retina is the activation of glial cells and microglia/macrophages. This can lead to inflammation and retinal neurodegeneration. The researchers found that A2-deletion mice displayed reduced activation of Müller cells and microglia/macrophages, as indicated by decreased immunoreactivity for GFAP and Iba1. Furthermore, they conducted gene expression analysis and observed reduced levels of pro-inflammatory markers, including interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and iNOS, in the A2-deletion mice.
A2 Overexpression in Neuronal Cells Induces Mitochondrial Dysfunction
Mitochondrial dysfunction is a significant factor in cell death after ONC. The study examined the role of A2 in this context by assessing the expression of the mitochondrial fission protein, dynamin-related protein 1 (Drp1). A2 deletion reduced Drp1 expression and improved mitochondrial respiration, indicating its involvement in mitochondrial dysfunction.
In vitro experiments using R28 retinal neuronal cells revealed that A2 overexpression increased Drp1 expression and decreased mitochondrial respiration. Treatment with the arginase inhibitor 2(S)-amino-6-boronohexanoic Acid (ABH) improved mitochondrial function. Additionally, the study utilized glutamate treatment to mimic excitotoxicity, a key contributor to neuronal injury. A2 overexpression or glutamate insult both induced comparable alterations in mitochondrial function.
Conclusion
In summary, this research sheds light on the detrimental role of A2 in neuronal survival and visual function after ONC injury. The study discovered that A2 deletion specifically in Calb2-expressing neurons resulted in improved neuronal survival, visual function, and multiple signaling pathways that support cell survival. A2 deletion also reduced DNA damage, apoptosis, inflammation, and mitochondrial dysfunction. The results emphasize the importance of A2's role in retinal neurodegeneration and propose it as a potential therapeutic target for optic nerve injuries and related conditions. The findings are a significant step towards understanding the precise mechanisms underlying retinal cell death and developing targeted therapies to preserve vision in cases of optic nerve injury.
The study findings were published in the peer reviewed journal: Cell Death & Disease.
https://www.nature.com/articles/s41419-023-06180-6
For the latest
Glaucoma News, keep on logging to Thailand Medical News.
Read Also:
https://www.thailandmedical.news/news/glaucoma-news-study-show-that-long-term-statin-use-increases-risk-of-onset-and-progression-of-glaucoma
https://www.thailandmedical.news/news/glaucoma-news-texas-study-alarmingly-finds-that-glucocorticoids-and-steroids-can-cause-glaucoma-and-also-accelerate-glaucoma-progression
https://www.thailandmedical.news/news/glaucoma-news-breakthrough-discovery-chinese-scientists-uncover-crucial-circrnas-implicated-in-acute-angle-closure-glaucoma
https://www.thailandmedical.news/news/glaucoma-new:-u-s-study-finds-that-complement-system-proteins-in-human-aqueous-humor-plays-a-role-in-primary-open-angle-glaucoma
https://www.thailandmedical.news/news/glaucoma-news-unveiling-the-mysteries-of-glaucoma-proteomic-insights-into-early-molecular-changes-in-response-to-ocular-hypertension
https://www.thailandmedical.news/news/glaucoma-news-german-study-shows-that-citicoline-does-not-slow-the-progression-of-glaucoma-beware-of-misleading-claims-online
https://www.thailandmedical.news/news/glaucoma-news-ohio-state-university-study-identifies-novel-rare-variants-and-genes-associated-with-intraocular-pressure-and-glaucoma
https://www.thailandmedical.news/news/breaking-glaucoma-news-scientists-at-university-of-california-regenerate-human-retinal-ganglion-cells-by-manipulating-gene-regulation
