Study Shows SARS-CoV-2 Impair Genes Associated With Pain-Related Pathways Resulting In Somatosensory Abnormalities For Exposed Individuals
COVID-19 News - SARS-CoV-2 Impair Genes Associated With Pain-Related Pathways May 14, 2023 3 years, 1 week, 14 hours, 20 minutes ago
COVID-19 News: The COVID-19 disease caused by SARS-CoV-2 poses a myriad of health challenges since its advent. With a spectrum of clinical outcomes from asymptomatic infection to mortality, the scientific world has been grappling to comprehend its complex nature. The disease has a broad range of symptoms, varying from common respiratory infection manifestations like cough, congestion, and fever to sensory-related issues like headaches and anosmia. In severe scenarios, the virus affects nearly every organ, leading to strokes, cardiovascular damage, and acute renal failure. Notably, a considerable number of patients, regardless of the severity of their infection, experience persistent sensory-related symptoms, including headaches, visceral pain, and nerve inflammation.

In a groundbreaking study, researchers from Boston University Chobanian & Avedisian School of Medicine, Icahn School of Medicine at Mount Sinai, and New York University have discovered that SARS-CoV-2 impacts thousands of genes even after the viral infection has been cleared. These affected genes are linked to neurodegeneration and pain-related pathways, implying lasting damage to the dorsal root ganglia - the spinal nerves responsible for conveying sensory messages. This damage could possibly be the underlying cause of the Post-COVID Conditions.
Lead researcher, Dr Venetia Zachariou, chair of pharmacology, physiology & biophysics at BU Chobanian & Avedisian School of Medicine explained to
COVID-19 News reporters at TMN, "Post COVID patients frequently report abnormal perception of touch, pressure, temperature, pain, or tingling. Our research suggests that SARS-CoV-2 may induce lasting pain in a unique way, highlighting the need for treatments that target virus-specific molecular pathways."
The study team employed experimental models involving hamsters infected with SARS-CoV-2 to study the virus's impact on sensitivity to touch, both during active infection and long after the infection had been cleared. They then compared these effects to those prompted by influenza A virus infection. The results revealed a slow but progressive increase in sensory sensitivity over time in the SARS-CoV-2 model, different from the influenza A virus, which induced rapid hypersensitivity during active infection that normalized once the infection was over.
RNA sequencing analysis of thoracic DRGs (Dorsal Root Ganglias) 1 to 4 days after infection suggested perturbations in predominantly neuronal signaling in SARS-CoV-2–infected animals as opposed to type I interferon signaling in IAV-infected animals. Later, 31 days after infection, a neuropathic transcriptome emerged in thoracic DRGs from SARS-CoV-2–infected hamsters, which coincided with SARS-CoV-2–specific mechanical hypersensitivity. These data revealed potential targets for pain management, including the RNA binding protein ILF3, which was validated in murine pain models. This work elucidates transcriptomic signatures in the DRGs triggered by SARS-CoV-2 that may underlie both short- and long-term sensory abnormalities.
th:662px" />
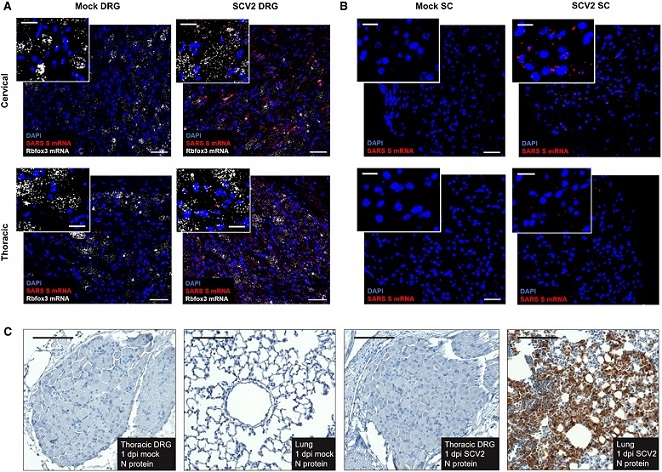
Viral mRNA is visibly detectable in DRGs and SCs by in situ hybridization, but mature virus is not detectable by immunohistochemistry.
(A) Expression pattern of Spike (S) RNA (red) around DAPI (blue) and Rbfox3 (white) in cervical and thoracic DRGs of SARS-CoV-2–infected and mock male hamsters at 1 dpi. Data are representative of n = 2 animals per group. (B) Expression patterns of Spike RNA (red) around DAPI (blue) in cervical and thoracic SCs from SARS-CoV-2–infected and mock-control male hamsters at 1 dpi. Data are representative of n = 2 per group. (C) Nucleocapsid protein expression pattern in thoracic DRGs of SARS-CoV-2–infected and mock-control male hamsters as well as lung tissue of male SARS-CoV-2–infected animals at 1 dpi. Data are representative of n = 2 animals per group. Scale bars, 50 μm; insets, 20 μm.
This study has provided a new lens to view the impact of SARS-CoV-2 on human host genes, offering valuable insights into the gene expression changes in sensory ganglia several weeks post-infection. "We hope our findings will pave the way for new strategies to address the somatosensory symptoms of Post COVID and ME/CFS (myalgic encephalomyelitis/chronic fatigue syndrome), which are only just beginning to be acknowledged by mainstream medicine. Our data, now publicly available, can yield insights into new therapeutic strategies," adds Dr Zachariou.
The study importantly identified the cytokine-associated factor ILF3, among other potential therapeutic targets, to treat pain associated with Post COVID and other neuropathies.
The study findings are a stepping stone in understanding the neurologic symptoms of COVID-19 and provide a roadmap for future research in this area. The discovery of SARS-CoV-2's unique impact on the nervous system can potentially guide the development of novel therapeutics for this and various other pain conditions.
While the scientific community has made significant strides in understanding and managing COVID-19, these new insights underline the fact that we are only just beginning to comprehend the virus's long-term effects. The unexplored landscape of Post COVID presents a new frontier for research, one that holds the promise of providing relief to countless individuals suffering from lingering and often debilitating symptoms including various forms of pain.
The study findings were published in the peer reviewed journal: Science Signaling.
https://www.science.org/doi/10.1126/scisignal.ade4984
For the latest
COVID-19 News, keep on logging to Thailand Medical News.

 th:662px" />
th:662px" />