Swedish Study Shows That Non-Neutralizing Antibodies Can Also Confer Protection To SARS-Cov-2 Infection By Mediating Phagocytosis
Source: Medical News - Non-Neutralizing Antibodies Jan 17, 2022 4 years, 3 months, 3 weeks, 18 hours ago
A new study by researchers from Lund University-Sweden, Skåne University Hospital-Sweden and the Public Health Agency of Sweden has found that contrary to previous notions, non-neutralizing antibodies can also play an important role in providing protection to SARS-CoV-2 infections as they help to mediate phagocytosis.
As a result of spike-specific antibodies being critical and central to effective COVID-19 immunity, many research efforts have only focused on antibodies that neutralize the ACE2-Spike interaction but not on non-neutralizing antibodies.
Unknown to many, antibody-dependent phagocytosis is an immune mechanism enhanced by opsonization, where typically, more bound antibodies trigger a stronger phagocyte response.
The Swedish study team that were also assisted by experts from Ecole Normale Supérieure Paris-Saclay-France, Institut National de la Santé et de la Recherche Médicale (INSERM)-France, Institute for Computational Science-Switzerland, Tanea Medical Ab-Sweden and Adlego Biomedical AB-Sweden, show that Spike-specific antibodies, dependent on concentration, can either enhance or reduce Spike-bead phagocytosis by monocytes independently of the antibody neutralization potential.
Interestingly, the study team found that both convalescent patient plasma and patient-derived monoclonal antibodies lead to maximum opsonization already at low levels of bound antibodies and is reduced as antibody binding to Spike protein increases.
Furthermore, the team also showed that this Spike-dependent modulation of opsonization correlate with the outcome in an experimental SARS-CoV-2 infection model.
These study findings suggest that the levels of anti-Spike antibodies could influence monocyte-mediated immune functions and propose that non-neutralizing antibodies could confer protection to SARS-CoV-2 infection by mediating phagocytosis.
The study findings were published in the peer reviewed journal: Frontiers in Immunology.
https://www.frontiersin.org/articles/10.3389/fimmu.2021.808932/full
Typically, when most people talk about antibodies against COVID-19, they tend to mean the so-called neutralizing antibodies, that offer protection by blocking the virus from invading our cells.
However, this new study has revealed that non-neutralizing antibodies may also be important in providing protection against COVID.
Lead researcher, Dr Pontus Nordenfelt from Lund University told Thailand
Medical News, "Our study findings indicate that non-neutralizing antibodies could also provide protection. This would mean that we have broader protection from antibodies than previously thought, making us less vulnerable to mutations of the virus. It warrants further investigation."
To date, in its efforts to produce vaccines and treatments, the research community has only focused on neutralizing antibodies, which prevent the spike protein of the virus from binding to the surface protein on our cells.
However, in this large project led by the study team from Lund University, a more detailed study has been carried out
on how the immune system also fights off the virus by means of phagocytosis ie the ability to ingest foreign particles, in COVID-19 patients.
In immunology, phagocytosis is a type of cleansing mechanism during which the immune system's players target potential threats, such as bacteria or viruses, and ingests them, and this mechanism is facilitated if the harmful substance is marked by an antibody.
The study findings indicate that non-neutralizing antibodies are also important in the fight against COVID through this mechanism.
Dr Pontus Nordenfelt added, "We observed that many of the antibodies that develop in response to COVID-19 are opsonizing, i.e. they signal to the immune system's phagocytes to ingest the virus. Our study findings indicate that non-neutralizing antibodies, so long as they are able to opsonize, are also able to provide an effective response to the virus."
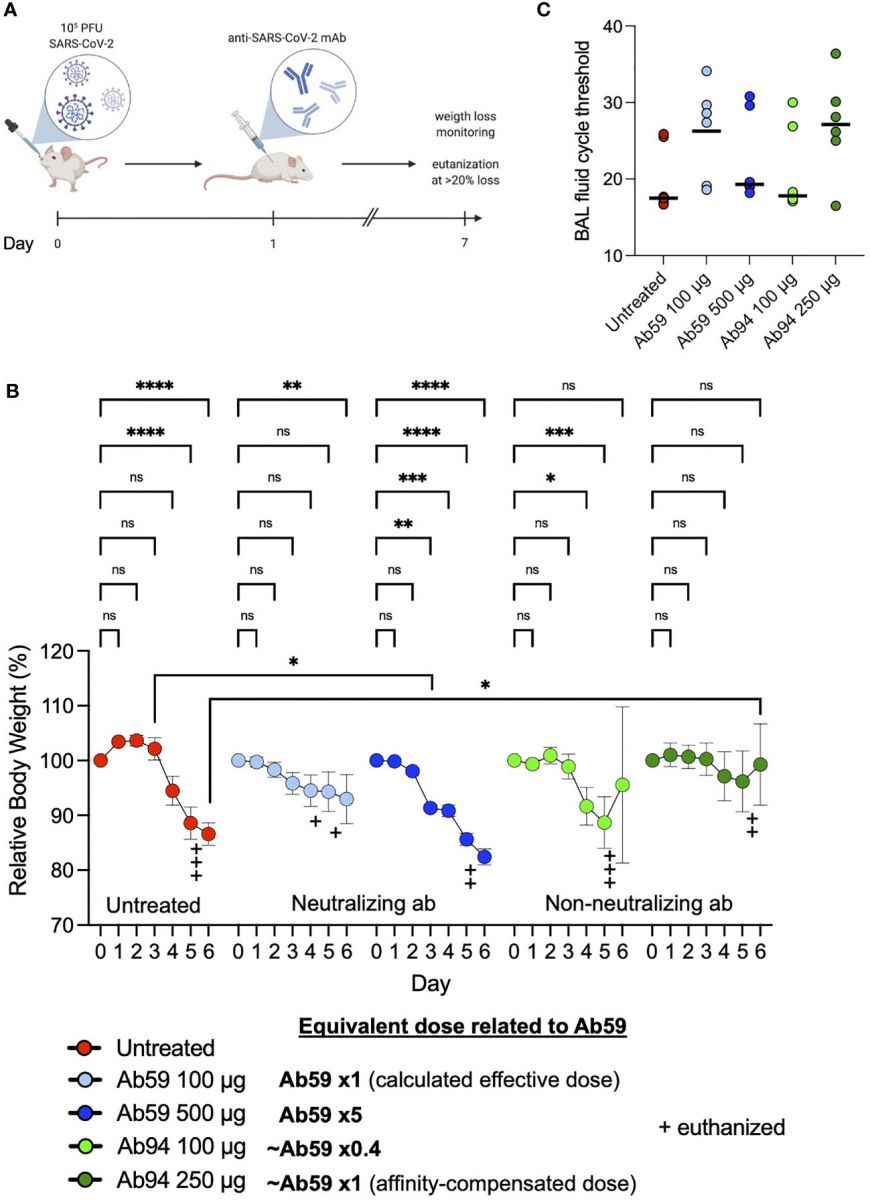 Non-neutralizing antibodies can protect against SARS-CoV-2 infection. (A) Humanized ACE2 mice were infected intranasally with SARS-CoV-2 (Wuhan strain). One day after infection, the animals (N=7 per group) were treated intraperitoneally with antibodies. Relative body weights were recorded and tabulated. (B) Body weights relative to each individual mouse over the time course of viral infection and treatment. (C) Quantitative PCR analysis was performed on the BAL fluids obtained from the infected mice at the experiment termination. The cycle threshold (Ct) values were detected and graphed. Error bars represent the SD. Statistical significance was assessed using two-way ANOVA with Dunnett’s multiple comparison correction both within each treatment group and across the groups for each day. * denotes p ≤ 0.05, ** for p ≤ 0.01, *** for p ≤ 0.001 and **** for p ≤ 0.0001. ns, not significant
Non-neutralizing antibodies can protect against SARS-CoV-2 infection. (A) Humanized ACE2 mice were infected intranasally with SARS-CoV-2 (Wuhan strain). One day after infection, the animals (N=7 per group) were treated intraperitoneally with antibodies. Relative body weights were recorded and tabulated. (B) Body weights relative to each individual mouse over the time course of viral infection and treatment. (C) Quantitative PCR analysis was performed on the BAL fluids obtained from the infected mice at the experiment termination. The cycle threshold (Ct) values were detected and graphed. Error bars represent the SD. Statistical significance was assessed using two-way ANOVA with Dunnett’s multiple comparison correction both within each treatment group and across the groups for each day. * denotes p ≤ 0.05, ** for p ≤ 0.01, *** for p ≤ 0.001 and **** for p ≤ 0.0001. ns, not significant
The study findings are supported by experiments on mice, in which the non-neutralizing antibodies protected the animals from COVID-19 infection.
The study team also discovered that the amount of neutralizing antibodies affects the process of phagocytosis.
The research team identified and isolated immune system cells from the blood of recovered COVID-19 patients.
Utilizing several techniques, they mapped which immune cells interact with the virus's spike protein and identified the antibody binding sites. Then, using flow cytometry, they studied how these antibodies affect phagocytosis.
The study team coated micrometer-sized beads with the spike protein and in doing so observed that the dosage of neutralizing antibodies had an effect on the phagocytosis.
The process of opsonization is the immune system mechanism in which an increase in bound antibodies triggers a stronger phagocyte response.
Interestingly, maximum opsonization is already achieved at relatively low levels of antibodies. Beyond this, the study team observed a reduction in the effect. The antibody-binding itself continued to increase linearly, even when the phagocytosis decreased, demonstrating that it is the spike protein that does something that causes this reduced phagocytosis.
In order to ensure that this was not due to other factors, the study team set up the same experiment, but this time they used only monoclonal antibodies. This meant that just one type of antibody (clone) was used instead of the standard combination of thousands of different antibodies.
First author of the study, Dr Wael Bahnan, researcher in infection medicine at Lund University said, "It showed the same trend. Up to a certain level, there is a linear dosage effect from the antibodies. After that, it dips. Basically, when you give a dose that is too high you don't get the protective effect."
Dr Bahnan further added, "It looks like something is happening to the spike protein that results in reduced interaction with the white blood cells, leading to the immune system no longer binding to and neutralizing the virus through phagocytosis. Similar phenomena have been described as the prozone effect and have been reported in experiments with pneumococcus and malaria. However, it has not been linked to reduced phagocytosis, so it is not certain that the effect relates to what we are observing in our experiments.”
The study team concluded, “Overall, the study findings presented highlight a concentration-dependent modulation of phagocytosis by anti-Spike antibodies. This modulation phenomenon might help explain the unclear clinical benefit seen with monoclonal antibody treatment for COVID19. This modulation is seen in patient material and translates well to animal infection experiments. The biophysical mechanism underlying the antibody-mediated phagocytic modulation is an exciting topic to pursue, as are the bridging immune steps between phagocytosis and animal protection.”
For more about
Non-Neutralizing Antibodies and SARS-CoV-2 Infections, keep on logging to Thailand Medical News.

