Study Reveals Correlation Between Neurological Long-COVID Symptoms And Autoantibodies Targeting Vasoregulatory And Autonomic Nervous Receptors
Nikhil Prasad Fact checked by:Thailand Medical News Team Sep 11, 2023 2 years, 8 months, 3 weeks, 1 day, 21 hours, 13 minutes ago
Long COVID: As the world grappled with the SARS-CoV-2 pandemic for more than two years, the prevalence of severe acute COVID-19 cases gradually decreased. However, a new challenge emerged in the form
of Long-COVID, a syndrome characterized by persisting symptoms that can affect multiple organ systems. Neurological disorders are among the most debilitating aspects of Long-COVID, causing profound impacts on individuals' health and quality of life.
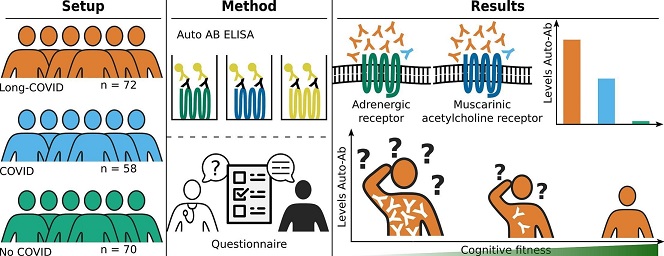 Graphical Abstract
Graphical Abstract
A new study by researchers from Universitätsklinikum der Ruhr-Universität Bochum-Germany, Charité - Universitätsmedizin Berlin- Germany, Universität zu Berlin- Germany and Berlin Institute of Health-Germany sheds light on the potential role of autoantibodies targeting vasoregulatory and autonomic nervous receptors in exacerbating neurological Long-COVID symptoms.
Defining Long-COVID
Long-COVID is a term used to describe a wide range of symptoms that persist beyond the acute phase of SARS-CoV-2 infection. According to the World Health Organization (WHO), Long-COVID is characterized by the continuation or development of new symptoms three months after the initial infection, with these symptoms lasting for at least two months and no other explanation for their presence. These persistent symptoms can affect various organ systems, including the respiratory, neurological, mental health, cardiovascular, and gastrointestinal systems. Common symptoms include fatigue, shortness of breath, myalgia (muscle pain), cognitive dysfunction, and more. Long-COVID can manifest as a single symptom or as a combination of multiple symptoms, often referred to as a syndrome.
One of the most significant and impactful syndromes associated with Long-COVID is chronic fatigue syndrome, also known as myalgic encephalopathy (ME/CFS). This syndrome, which has been recognized for decades, is characterized by extreme fatigue that impairs daily functioning in both personal and professional life. Despite ongoing research, the exact underlying mechanisms of ME/CFS remain incompletely understood. However, one hypothesis suggests that the generation of autoantibodies to specific receptors, such as G-protein coupled receptors (GPCRs), may contribute to the development of this syndrome.
Autoantibodies and G-Protein Coupled Receptors
G-protein coupled receptors (GPCRs) are a diverse and extensive family of receptors that play a crucial role in transmitting extracellular signals into cellular responses. Among their functions, GPCRs are involved in regulating local vasomotor tone by mediating signaling of molecules like catecholam
ines, acetylcholine, endothelin 1, and angiotensin II.
Autoantibodies targeting GPCRs can have both agonistic and antagonistic effects on these receptors, potentially leading to autonomic dysfunction and microcirculatory hypoperfusion. These effects may contribute to the symptoms experienced by individuals with Long-COVID.
Previous research has indicated a correlation between the severity of fatigue and muscle pain in patients with infection-induced ME/CFS and the concentrations of GPCR autoantibodies. With SARS-CoV-2 being a virus known to induce chronic fatigue syndrome, investigating the presence and impact of autoantibodies targeting GPCRs in Long-COVID becomes a critical area of study.
Autoimmunity and Long-COVID
Long-COVID is believed to result from a combination of virus-specific pathophysiological changes, persistent inflammation, and humoral autoimmunity, all contributing to immune-mediated damage. While autoantibodies have been identified in acute COVID-19 cases, including those targeting platelet receptors involved in blood clotting, the relationship between autoantibodies targeting GPCRs and inflammation in the context of COVID-19 remains unclear.
The Study: Investigating Autoimmunity in Long-COVID
To better understand the role of autoimmunity in Long-COVID, the study team conducted a monocentric cross-sectional study at the University Hospital of Ruhr-University Bochum, Germany. The study recruited patients who had experienced mild to moderate SARS-CoV-2 infections up to 12 months prior to enrollment.
Three groups were established: Long-COVID, individuals with a history of SARS-CoV-2 infection without persisting symptoms (COVID), and a control group with no known history of SARS-CoV-2 infection (No COVID).
In total, 200 individuals participated in the study, with 72 classified as Long-COVID, 58 as COVID, and 70 as No COVID. The median age across all groups was similar (48 years), ensuring age-related factors did not significantly impact the results. Long-COVID patients were administered a questionnaire to self-assess their symptoms, providing valuable insight into their experiences.
Findings and Implications
The study yielded intriguing results that shed light on the potential link between autoimmunity and neurological Long-COVID symptoms. Autoantibodies targeting vasoregulatory and autonomic nervous system receptors were found to be significantly more prevalent and present at higher concentrations in the Long-COVID group compared to the COVID and No COVID groups. This observation suggests a potential association between these autoantibodies and the development of Long-COVID.
Furthermore, the Long-COVID group exhibited a higher number of patients with simultaneous detection of multiple autoantibodies, indicating a complex interplay of autoimmunity in the syndrome. Importantly, despite the presence of these autoantibodies, the overall inflammatory state in all three groups was relatively low, challenging previous assumptions about the role of inflammation in Long-COVID.
Correlations between autoantibody concentrations and specific neurological symptoms were also identified. Autoantibodies targeting Adrenergic Receptor B1 and B2 (ARB1 and ARB2) showed negative correlations with cognitive tasks, such as the CERAD Trail Making A and B tests, suggesting a potential link between these autoantibodies and impaired psychomotor speed and attention. Conversely, autoantibodies targeting muscarinic acetylcholine receptor M3 (CHRM3) correlated positively with the Chadler Fatigue Scale, indicating a connection between CHRM3 autoantibodies and increased fatigue.
These findings provide critical insights into the potential mechanisms underlying neurological Long-COVID symptoms. Autoimmunity, particularly the presence of autoantibodies targeting GPCRs, may play a significant role in the intensity and persistence of cognitive and physical impairment in Long-COVID patients.
The Global Impact of Long-COVID
The WHO's recognition of Long-COVID as a widespread and persistent condition affecting millions of individuals underscores the urgency of addressing this complex syndrome. Long-COVID has far-reaching implications, not only for the affected individuals but also for healthcare systems and economies worldwide.
This study's findings emphasize the need for further research into the causative role of autoantibodies in Long-COVID. While the current study highlights correlations, establishing causation would open the door to potential therapeutic interventions targeting autoimmunity in Long-COVID. This approach aligns with the WHO's call for increased investment in research, recovery, and rehabilitation to address the challenges posed by Long-COVID.
In conclusion, the study conducted by German researchers offers valuable insights into the potential role of autoantibodies targeting vasoregulatory and autonomic nervous system receptors in the severity of neurological Long-COVID symptoms. While the study does not confirm a causative relationship, it opens new avenues for future research and therapeutic interventions, holding promise for the millions of individuals affected by Long-COVID worldwide.
The study findings were published as a journal Pre-proof in the journal: Autoimmunity reviews.
https://www.sciencedirect.com/science/article/abs/pii/S1568997223001799
For the latest on
Long COVID, keep on logging to Thailand Medical News.
