Murine Study Reveals Yet Another Way That SARS-CoV-2 Causes Heart Damage - Targeting Cardiac Pericytes, Causing Heart's Microvasculature Damage!
COVID-19-News - SARS-CoV-2 - Cardiac Pericytes - Heart Microvasculature Damage Jun 08, 2023 2 years, 11 months, 3 weeks, 2 days, 16 hours, 23 minutes ago
How SARS-CoV-2's Spike Protein Damages The Heart
COVID-19 News: Since the onset of the COVID-19 pandemic, the world has grappled with the devastating effects of the disease caused by the SARS-CoV-2 virus. Among the many complications associated with COVID-19, cardiovascular issues have emerged as a significant concern, contributing to a high number of fatalities. Past
COVID-19 News reports have already showed that the SARS-CoV-2 virus can affect the human heart through multiple ways.
https://www.thailandmedical.news/news/russian-scientists-warn-exposure-to-sars-cov-2-will-result-in-various-cardiac-dysfunctions-even-in-healthy-individuals-who-only-had-mild-symptoms
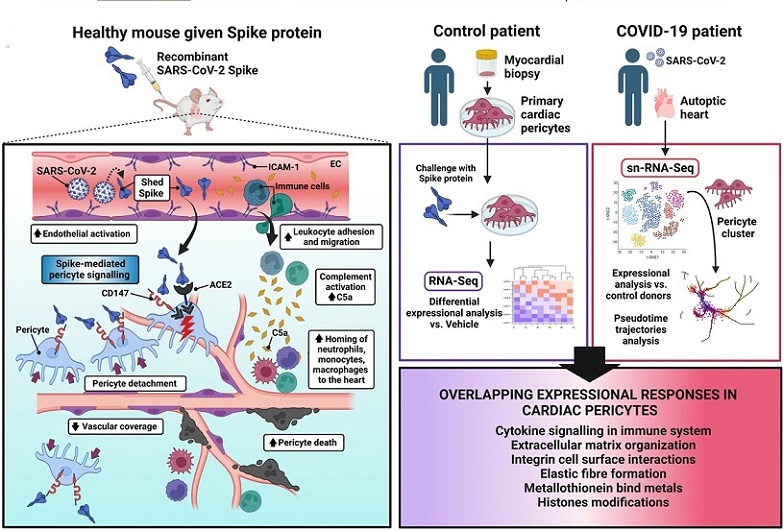 Schematic summarizing major findings and candidate mechanisms underpinning the S-protein damaging action. Left panel: Novel evidence provided that S-protein alone can damage the heart microvasculature of otherwise healthy mice. On one side, the S-protein acts as a ligand activating intracellular pericyte signaling, which results in pericyte detachment, death, and decreased vascular coverage, thus disrupting the coronary microcirculation. On the other, the S-protein triggers endothelial activation (ICAM-1+ endothelial cells), resulting in increased homing of leukocytes to the heart and accumulation of activated complement protein C5a. Right panel: A comparison between the expressional changes induced by the S-protein in primary human cardiac pericytes in vitro and single-nuclei (sn)-RNA-Sequencing pseudo-time trajectories analysis in pericytes extracted from the heart of deceased COVID-19 patients revealed overlapping expressional responses as indicated. These findings suggest that at least some of the in vivo effects of SARS-CoV-2 on human cardiac pericytes may be due to the modulation of inflammatory and epigenetic pathways triggered by the S-protein interaction with its cell surface receptors
Schematic summarizing major findings and candidate mechanisms underpinning the S-protein damaging action. Left panel: Novel evidence provided that S-protein alone can damage the heart microvasculature of otherwise healthy mice. On one side, the S-protein acts as a ligand activating intracellular pericyte signaling, which results in pericyte detachment, death, and decreased vascular coverage, thus disrupting the coronary microcirculation. On the other, the S-protein triggers endothelial activation (ICAM-1+ endothelial cells), resulting in increased homing of leukocytes to the heart and accumulation of activated complement protein C5a. Right panel: A comparison between the expressional changes induced by the S-protein in primary human cardiac pericytes in vitro and single-nuclei (sn)-RNA-Sequencing pseudo-time trajectories analysis in pericytes extracted from the heart of deceased COVID-19 patients revealed overlapping expressional responses as indicated. These findings suggest that at least some of the in vivo effects of SARS-CoV-2 on human cardiac pericytes may be due to the modulation of inflammatory and epigenetic pathways triggered by the S-protein interaction with its cell surface receptors
Now, a groundbreaking study conducted by researchers from the University of Bristol and Imperial College London has uncovered a new aspect of the virus's impact on the heart, specifically targeting cardiac pericytes, cells vital for maintaining vascular integrity. These findings have far-reaching implications for understanding the disease and developing potential treatments.
Cardiac pericytes, known for their role in preserving the health of the microvasculature, have been identified as a primary target of SARS-CoV-2 infection. These perivascular cells engage in vital communication with capillary endothelial cells, ensuring the integrity of the blood vessels. However, when pericytes become dysfunctional and detach, the virus gains a foothold, spreading from the bloodstream and inflicting damage on the heart muscle.
S-Protein's Dark Side
The SARS-CoV-2 virus gains entry into cell
s through its Spike (S) protein, which binds to ACE2 and CD147 receptors. Intriguingly, this study reveals that the S-protein not only facilitates viral entry but also directly activates the ERK1/2 MAPK signaling pathway in cardiac pericytes. This finding indicates that the S-protein acts as a double-edged sword, both enabling viral infection and wreaking havoc on the heart cells. Moreover, this study provides compelling evidence linking the S-protein to the initiation of the cytokine storm, a dangerous inflammatory response implicated in severe COVID-19 cases.
To unravel the consequences of S-protein exposure, the study team conducted a series of experiments in healthy mice. They administered the S-protein intravenously and observed its acute effects on the heart's microvasculature. The results were striking. While the S-protein did not alter capillary density, it led to increased expression of ICAM-1, a molecule involved in immune cell-endothelial interactions. Furthermore, the density, coverage, and viability of cardiac pericytes were significantly reduced, exposing the vulnerability of the heart's defense system.
To gain further insights into the impact of the S-protein, the researchers turned to human cardiac pericytes in vitro. By subjecting these cells to the S-protein, they conducted RNA sequencing analysis to uncover changes in gene expression. This analysis revealed the differential modulation of hundreds of RNA transcripts, with inflammatory pathways and infectious diseases prominently featured.
Importantly, the study also showed the downregulation of processes related to healing and angiogenesis, shedding light on the mechanisms underlying the heart's struggle to recover from SARS-CoV-2 damage.
Connecting the Dots
Taking their investigation a step further, the study team compared their findings with single-nuclei RNA sequencing datasets obtained from the hearts of COVID-19 patients. This cross-referencing approach revealed a remarkable overlap in expressional changes induced by the S-protein and those observed in infected human hearts. Specific gene clusters associated with cytokine signaling, extracellular matrix organization, and integrin cell surface interactions were identified, further substantiating the link between the S-protein and the transcriptional changes observed in COVID-19 patients!
This groundbreaking study provides compelling evidence that the SARS-CoV-2 S-protein directly inflicts damage on cardiac pericytes, causing microvascular harm in the heart. These findings suggest that the detrimental effects observed in healthy mice just three days after exposure to the S-protein could be even more pronounced in individuals with cardiovascular risk factors or prolonged exposure to the virus. This discovery highlights the urgent need for further investigation into the implications for COVID-19 patients and the development of targeted therapies.
Potential Treatment Approaches
The study's findings not only deepen our understanding of the virus's mechanisms but also open up exciting avenues for potential treatments. Through drug target enrichment analysis, the study team identified compounds that could reverse the expressional changes induced by the S-protein in pericytes. Anti-tumoral, pro-apoptotic, anti-viral, anti-inflammatory, and anti-thrombotic drugs emerged as promising candidates, some of which have already been tested in COVID-19 patients. While further research is needed to determine the efficacy of these interventions, this discovery offers hope in the fight against COVID-19.
Also, based on these findings, anti-S-protein engineering approaches could hold promise in protecting vascular cells from the detrimental effects of SARS-CoV-2. By blocking the interaction between the S-protein and host receptors, it may be possible to prevent viral entry and subsequent damage to the heart and other organs. This strategy represents an exciting avenue for future research and therapeutic development.
Conclusion
This new study uncovers the dark side of the SARS-CoV-2 S-protein, revealing its direct pathogenic action on cardiac pericytes and the heart's microvasculature. The study provides crucial insights into the mechanisms underlying COVID-19-related cardiovascular complications and offers potential avenues for targeted therapies. By deepening our understanding of the virus's impact on the heart, researchers are paving the way for innovative treatments that may alleviate the devastating effects of COVID-19 and save lives.
The study findings were published in the peer reviewed journal: Signal Transduction And Targeted Therapy.
https://www.nature.com/articles/s41392-023-01489-2
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
