COVID-19 New: Scientist Hypothesize that Furin Plays A Key Role In The Pathogenesis Of COVID-19 Associated Neurological Disorders
Nikhil Prasad Fact checked by:Thailand Medical News Team Nov 03, 2023 2 years, 6 months, 1 week, 4 days, 11 hours, 56 minutes ago
COVID-19 News: The COVID-19 pandemic, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has had a devastating impact on global health, resulting in millions of deaths and long-term health issues. Initially considered a respiratory illness, COVID-19 has shown its ability to infiltrate the central nervous system, leading to various neurological complications. These complications include cognitive impairments, cerebrovascular diseases, demyelinating pathologies, encephalopathy, and even brain atrophy. It is vital to understand the mechanisms behind SARS-CoV-2's invasion of the central nervous system and its impact on neurological health.
A novel hypothesis covered in this
COVID-19 News report by researchers from Azerbaijan Medical University-Azerbaijan, Bogomolets National Medical University, Kyiv-Ukraine and Georgetown University Medical Center-USA,
suggests that furin protease-dependent cleavage of the SARS-CoV-2 spike protein plays a significant role in COVID-19-associated neurological disorders. This article explores this hypothesis, shedding light on the potential mechanisms by which SARS-CoV-2 may exacerbate neurological damage, leading to cognitive impairments and neurodegenerative diseases.
The Impact of COVID-19 on Neurological Health
The neurological consequences of COVID-19 have become increasingly apparent. Reports indicate that over 35% of COVID-19 cases are associated with problems affecting the central nervous system. These issues range from mild symptoms such as confusion, anosmia (loss of smell), and anxiety to severe and long-term complications, including cognitive impairments, stroke, and encephalopathy.
Additionally, studies have documented hippocampal shrinkage, brain size reduction, and neurodegeneration following SARS-CoV-2 infection. These observations emphasize the need to understand how the virus affects the central nervous system.
The Role of Renin Angiotensin System in Neurological Health
The renin-angiotensin system (RAS) is a crucial regulator of blood pressure and fluid balance, with its components found in various tissues, including the brain. SARS-CoV-2 enters host cells by binding to the angiotensin-converting enzyme 2 (ACE2), an essential component of the RAS. ACE2 typically converts angiotensin II (Ang II) into angiotensin 1-7 (Ang 1-7), thereby degrading Ang II. Ang II is a vasoconstrictor known to play a role in the development of numerous diseases, including neurological disorders.
The binding of the SARS-CoV-2 spike protein to ACE2 results in ACE2 downregulation. This leads to reduced peptidase activity, inhibiting the conversion of Ang II to Ang 1-7. Consequently, Ang II levels increase, activating the Ang II/Ang II receptor type 1 (AT1R) pathway. Elevated Ang II levels contribute to the production of reactive oxygen species and inflammatory mediators in the brain, which can lead to neuroinflammation, oxidative stress, and reduced cerebral blood flow. Additionally, ACE2 downregulation in the hypothalamus has been associated with stress and anxiety conditions.
Protective effects against neurodegenerative diseases are offered by alternative axes of the RAS, including ACE2/Ang 1
-7/Mas receptor and ACE2/Ang 1-7/AT2 receptor pathways. These pathways reduce oxidative stress, inflammation, and promote vasodilation, antithrombotic effects, and neuroprotection. Medications that inhibit the RAS have shown promise in reducing the risk of developing Alzheimer's disease and other neurological disorders.
The Spike Protein's Impact on ACE2
SARS-CoV-2 relies on the interaction between its spike protein and ACE2 to enter host cells. This interaction leads to ACE2 downregulation, increasing Ang II levels. Animal studies have demonstrated the impact of the spike protein on ACE2 expression and subsequent lung injury. Additionally, the spike protein has been found to induce clathrin-dependent endocytosis and degradation of ACE2.
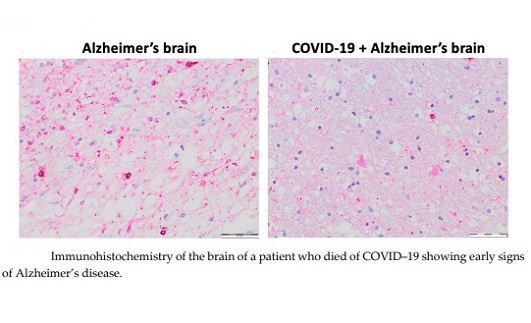
Furthermore, there is evidence that SARS-CoV-2 may affect ACE2 mRNA expression, further reducing ACE2 levels. Immunohistochemical evaluations of COVID-19 patients have shown reduced ACE2 protein expression, even in patients with Alzheimer's disease.
The Hypothesis: Furin Protease Amplification
The central hypothesis presented suggests that furin protease, which cleaves the SARS-CoV-2 spike protein, can lead to an amplification of the spike protein's effects on ACE2 downregulation and Ang II production, ultimately contributing to neurological disorders seen in COVID-19 patients.
Furin-dependent cleavage of the spike protein results in the release of free S1 subunit proteins. This amplification process increases the number of spike protein molecules capable of binding to ACE2, leading to a potentially significant reduction in ACE2 expression and an elevation of Ang II levels. This mechanism may intensify the pathogenesis of COVID-19 and its associated neurological complications.
Evaluation of the Hypothesis
This hypothesis builds upon the viral protein fragment theory, which posits that fragments of the spike protein can target host cells independently of the intact virus. The spike protein consists of two subunits, S1 and S2. The S1 subunit contains the receptor-binding domain (RBD) responsible for binding to ACE2, making it an essential element in host cell entry.
Furin protease plays a vital role in cleaving the spike protein into S1 and S2 subunits. These cleaved fragments can be released into the bloodstream. Studies have detected SARS-CoV-2 S1 protein in the plasma of COVID-19 patients and even in recipients of mRNA COVID-19 vaccines. Notably, this circulating S1 protein can readily cross the blood-brain barrier, making it a potential culprit for neurological complications.
Furin protease cleaves proteins at specific amino acid sequences, releasing fragments that can target ACE2 receptors. Since one viral particle may contain numerous spike protein molecules, the proposed amplification process could lead to the downregulation of ACE2 in a manner 50-100 times more efficient than intact viral particles.
Consequences of the Hypothesis
The loss of ACE2, whose primary function is to degrade Ang II, results in increased Ang II levels. Numerous studies have shown elevated Ang II levels in the plasma of COVID-19 patients, and the virus's spike protein is likely responsible for downregulating ACE2 expression.
This hypothesis carries significant implications for COVID-19 patients and potentially for those receiving vaccines. If proven correct, furin inhibitors may be a viable avenue for reducing COVID-19-related pathologies, including neurological disorders.
Conclusion
The hypothesis presented in this article suggests that furin protease amplification of the SARS-CoV-2 spike protein's effects on ACE2 downregulation may contribute to the neurological complications seen in COVID-19 patients. The release of free S1 subunit proteins could result in significant reductions in ACE2 expression and an increase in Ang II levels, exacerbating the disease's pathogenesis.
Understanding these mechanisms is crucial for developing effective treatment strategies and preventing the adverse neurological effects of COVID-19. Further research and clinical studies are necessary to validate this hypothesis and explore potential therapeutic interventions using furin inhibitors.
The hypothesis was published on a preprint server and is currently being peer reviewed.
https://www.preprints.org/manuscript/202310.2100/v1
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
