COVID-19 News: University of Texas Study Finds That Pannexin-1 Channels, ATP And Purinergic Receptors Play Key Roles In SARS-CoV-2 Pathogenesis!
Nikhil Prasad Fact checked by:Thailand Medical News Team Oct 03, 2023 2 years, 7 months, 2 weeks, 4 days, 14 hours, 36 minutes ago
COVID-19 News: Infectious diseases caused by pathogens like viruses have always posed a significant threat to human health. Two notable examples of such pathogens are the human immunodeficiency virus-1 (HIV) and the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). Both of these viruses utilize host proteins to infiltrate, replicate, and incite inflammation within the host's body. Emerging research from the Department of Neurobiology at The University of Texas Medical Branch (UTMB), USA, has unearthed a crucial link between Pannexin-1 channels, extracellular adenosine triphosphate (ATP), and purinergic receptors in the pathogenesis of these infectious diseases. This
COVID-19 News report delves into the intricate web of interactions within this axis and explores the potential therapeutic applications that could mitigate the devastating consequences of these viral threats.
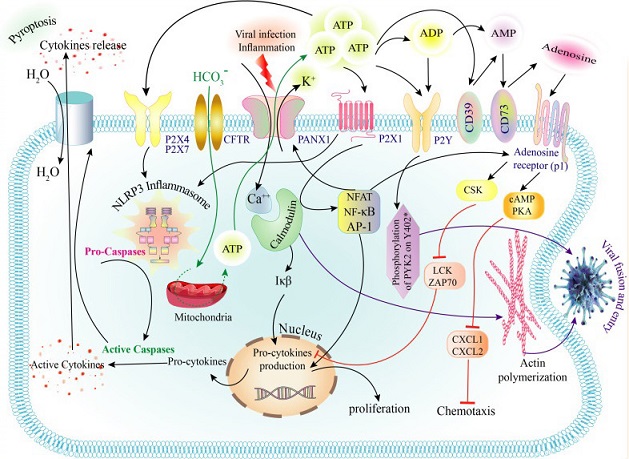 Purinergic receptors play a crucial role in the development of viral infections by regulating various cellular processes. When a viral infection occurs, it triggers inflammation and activates Panx1, leading to the release of ATP and K+ ions and the influx of Ca++. Additionally, CFTR allows bicarbonate to enter the cell, ultimately increasing ATP production in mitochondria. This surplus ATP is released from the cell through the Panx1 channel. The released ATP and ADP molecules bind to P2Y and P2X receptors, while AMP, generated by CD39, can be converted to adenosine via CD73.Intracellular Ca++ binds to calmodulin, which activates IkB, leading to the production of precursor molecules for inflammatory cytokines. Ca++-calmodulin also triggers the activation of transcription factors such as NFAT, NF-κB, and AP-1. These transcription factors further stimulate the adenosine receptor (AR) upon adenosine binding. AR activation suppresses inflammatory cytokines and chemotaxis by initiating CSK, CAMP, and PKA activation and inhibiting LCK, ZAP70, CXCL1, and CXCL2. Binding of ATP to P2X4 and P2X7 receptors activates the NLRP3 inflammasome, which in turn converts pro-caspases into active caspases. These active caspases convert precursor cytokines into their active forms. Subsequently, cytokines are released from the cell through gasdermin-mediated pyroptosis. The P2Y receptor is activated through PYK2 phosphorylation and the Ca++-calmodulin complex formed during actin polymerization, facilitating virus entry.
Purinergic Signaling: A Novel Frontier
Purinergic receptors play a crucial role in the development of viral infections by regulating various cellular processes. When a viral infection occurs, it triggers inflammation and activates Panx1, leading to the release of ATP and K+ ions and the influx of Ca++. Additionally, CFTR allows bicarbonate to enter the cell, ultimately increasing ATP production in mitochondria. This surplus ATP is released from the cell through the Panx1 channel. The released ATP and ADP molecules bind to P2Y and P2X receptors, while AMP, generated by CD39, can be converted to adenosine via CD73.Intracellular Ca++ binds to calmodulin, which activates IkB, leading to the production of precursor molecules for inflammatory cytokines. Ca++-calmodulin also triggers the activation of transcription factors such as NFAT, NF-κB, and AP-1. These transcription factors further stimulate the adenosine receptor (AR) upon adenosine binding. AR activation suppresses inflammatory cytokines and chemotaxis by initiating CSK, CAMP, and PKA activation and inhibiting LCK, ZAP70, CXCL1, and CXCL2. Binding of ATP to P2X4 and P2X7 receptors activates the NLRP3 inflammasome, which in turn converts pro-caspases into active caspases. These active caspases convert precursor cytokines into their active forms. Subsequently, cytokines are released from the cell through gasdermin-mediated pyroptosis. The P2Y receptor is activated through PYK2 phosphorylation and the Ca++-calmodulin complex formed during actin polymerization, facilitating virus entry.
Purinergic Signaling: A Novel Frontier
Purinergic signaling is a form of extracellular signaling mediated by purine nucleotides and nucleosides such as adenosine and ATP. It involves the activation of purinergic receptors in the cell and/or in nearby cells, thereby regulating cellular functions. The purinergic signalling complex of a cell is sometimes referred to as the “purinome”.
Purine nucleosides are not just essential for cellular metabolism; they also play a pivotal role in extracellular signaling. The role of purinergic signaling has been extensively studied in various acute and chronic diseases, as well as in injury resolution. However, its significance in emerging diseases has only recently come under the spotlight.
Extracellular A
TP, often referred to as a "danger signal," is a potent inflammatory molecule, even at low concentrations. Conversely, adenosine, one of its downstream metabolites, exhibits anti-inflammatory properties. In healthy conditions, the immune system's response to ATP is tightly regulated and transient. This regulation is achieved through the rapid degradation of purine nucleotides by ectonucleotidases like CD39 and ectonucleotidase triphosphate diphosphohydrolase (ENTPD-1). These enzymes catalyze the conversion of ATP and adenosine diphosphate (ADP) into adenosine monophosphate (AMP). Subsequently, CD73, or ecto-5'-nucleotidase (NT5E), further converts AMP into adenosine. This intricate process serves to prevent the overactivation of the purinergic immune system. Consequently, extracellular ATP has an exceedingly short half-life and is rapidly catabolized within minutes.
ATP in Immune Activation
ATP is a crucial player in immune activation. It enhances T cell activation and antigen presentation, initiating an immune response and preventing T cell anergy. In fact, ATP has been shown to increase dendritic cell maturation markers and promote antigen presentation and T cell activation.
Furthermore, ATP and purinergic activation enhance the immune response of various immune cells, including B cells, monocytes/macrophages, eosinophils, and dendritic cells (DCs). Additionally, ATP plays a key role in cellular migration, as demonstrated in dendritic cells. It can either enhance or inhibit T lymphocyte responses, depending on its concentration. High ATP concentrations compromise proper immune T lymphocyte responses by preventing the migration of these cells. Conversely, low ATP concentrations stimulate the proliferation and chemotaxis of regulatory T cells (Tregs).
Intriguingly, low extracellular ATP levels enhance IL-2 secretion and promote lymphocyte migration and adhesion to endothelium, facilitating their transmigration into different tissues. Conversely, high ATP levels on activated CD4+ T lymphocytes inhibit lymphocyte activation and migration. This inhibition can impair surveillance, antigen presentation, and even T cell survival and proliferation.
Pathological Conditions: A Surge in ATP
Under pathological conditions, such as infection, compromised or dying cells release a significant amount of ATP into the extracellular space. This surge in ATP levels disrupts homeostasis, leading to the overactivation of purinergic receptors and associated signaling pathways. This overactivation has been documented in several chronic conditions, including gout, allergen-driven lung inflammation, chronic obstructive pulmonary disease (COPD), diabetes, inflammatory bowel disease, asthma, and rheumatoid arthritis. Furthermore, severe diseases such as cancer, multiple sclerosis, leukemia, and graft-versus-host disease have also been linked to perturbations in the purinergic system, which may contribute to chronic inflammatory states.
The Role of ATP in Macrophages and Microglia
Macrophages, including tumor-associated macrophages, can exhibit two distinct phenotypes: M1 (classically activated macrophages) and M2 (alternatively activated macrophages). M1 macrophages possess proinflammatory properties and anti-tumor activity, whereas M2 macrophages secrete the anti-inflammatory cytokine IL-10, promoting cancer cell survival, proliferation, plasticity, and invasiveness.
Studies have shown that M1 macrophages have lower expression of ectonucleotidases and a slower ATP hydrolysis rate compared to M2 macrophages. Consequently, millimolar concentrations of ATP can promote the production of proinflammatory cytokines such as IL-1β, IL-6, IL-18, CCL2, and TNF-α in macrophages/microglia via a P2X7-dependent mechanism.
Low ATP levels, along with other nucleotides, activate G-protein coupled P2Y receptors to mediate the chemotaxis of myeloid cells into damaged tissues. This activation contributes to the differentiation and activation of monocytes/macrophages/microglia.
Panx-1 Channels: The Gateway for ATP
Pannexin-1 (Panx-1) is a protein known for its role in forming hemichannels that facilitate apoptotic signaling, release of inflammatory molecules, and solute trafficking across the plasma membrane. These channels are typically constitutively closed but can open in response to specific stimuli, including hypoxia, coagulation, blood pressure alterations, endothelial permeability changes, and apoptosis.
The Contribution of Panx-1 Channels to ATP Signaling
Panx-1 channels have emerged as key players in the release of ATP during various inflammatory conditions. When Panx-1 channels open, they enable the release of ATP into the extracellular space, where it can act as a signaling molecule. This phenomenon occurs in response to a wide range of triggers, including hypoxia, coagulation, and even changes in blood pressure and endothelial permeability.
Mechanisms of PANX1 Channel/Purinergic Receptors Involvement in COVID-19 Pathogenesis
The PANX1/purinergic receptors signaling system plays a role in modulating the immune response, acting as both proviral and antiviral factors.
SARS-CoV-2 primarily infects cells by binding to ACE2 receptors, leading to the dysregulation of the renin-angiotensin signaling pathway. Angiotensin II, which increases due to viral attachment, can activate PANX1 currents and sustain PANX1 channel activity, releasing ATP into the extracellular space. This persistent release of ATP can activate signaling pathways that promote oxidative stress and tissue injury, contributing to COVID-19 complications.
Oxidative stress, resulting from an imbalance between ROS production and clearance, plays a role in viral infections, leading to cell damage and immune-inflammatory injury. PANX1-mediated ATP release can activate P2 receptors, promoting ROS production and oxidative stress. Excessive ATP release through PANX1 channels can induce proapoptotic mechanisms and DNA damage, further exacerbating oxidative stress and apoptosis.
Endothelial dysfunction in COVID-19 can lead to organ ischemia, increased oxidative stress, inflammation, and pro-coagulant states. PANX1 channels within endothelial cells can regulate ATP release, influencing vasodilation and inflammatory cell adhesion. Dysfunction of endothelial cells may be mediated by PANX1 activation through TNF signaling, which results in Ca2+ increase and inflammatory cell recruitment.
PANX1 is involved in multiple physiological functions, making it a potential contributor to COVID-19 clinical manifestations. These functions include inflammation, regulation of blood flow, tissue development, and more.
Targeting PANX1 and Purinergic Signaling in COVID-19 Therapy
Targeting PANX1 and purinergic receptors may offer novel therapeutic options for COVID-19 prevention and treatment. Potential approaches include:
-Repurposed FDA-Approved Drugs:
Drugs such as PBN, Spironolactone, and Tenofovir have shown potential in inhibiting PANX1 activity or blocking purinergic signaling pathways.
These drugs may help reduce inflammation and viral infection associated with COVID-19.
-Directly Targeting PANX1 Channels:
PANX1-specific inhibitors like PxIL2P can regulate intracellular calcium and reduce inflammatory responses.
Developing new drugs that directly target PANX1 channels may be beneficial for COVID-19 treatment if further research confirms their involvement.
-Purinergic Receptor Antagonists:
Purinergic receptor blockers such as Suramin, PPADS, and BBG have been used to treat HBV and other viral infections and may be considered for COVID-19 therapy.
Conclusion
In conclusion, the intricate interplay between Pannexin-1 channels, extracellular ATP, and purinergic receptors holds immense promise in the realm of infectious disease research. Understanding these molecular mechanisms not only sheds light on the pathogenesis of diseases like HIV and SARS-CoV-2 but also opens doors to novel therapeutic interventions. As we continue to unravel the mysteries of purinergic signaling, we may uncover new strategies to combat infectious diseases and safeguard human health.
The study findings were published in the peer reviewed journal: Current Opinion in Pharmacology.
https://www.sciencedirect.com/science/article/abs/pii/S1471489223000590
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
