Japanese Researchers Discover That Exocyst Complex Component 2 (EXOC2) Is An Important Host Factor In SARS-CoV-2 Infections
Source: SARS-CoV-2 Research - EXOC2 Oct 31, 2022 3 years, 6 months, 2 weeks, 2 days, 7 hours, 5 minutes ago
SARS-CoV-2 Research: Researchers from Kyoto University - Japan and AMED-CREST, Japan Agency for Medical Research and Development (AMED)- Japan have in a news study found that Exocyst Complex Component 2 (EXOC2) is an important host factor in SARS-CoV-2 infections.
Exocyst complex component 2 is a protein that in humans is encoded by the EXOC2 gene. The protein encoded by this gene is a component of the exocyst complex, a multiple protein complex essential for targeting exocytic vesicles to specific docking sites on the plasma membrane. Though best characterized in yeast, the component proteins and the functions of the exocyst complex have been demonstrated to be highly conserved in higher eukaryotes. At least eight components of the exocyst complex, including this protein, are found to interact with the actin cytoskeletal remodeling and vesicle transport machinery. This interaction has been shown to mediate filopodia formation in fibroblasts.
The SARS-CoV-2 virus has caused an epidemic and has spread rapidly all over the world. Because the analysis of host factors other than receptors and proteases has not been sufficiently performed, the study team attempted to identify and characterize host factors essential for SARS-CoV-2 infection using iPS cells and airway organoids (AO).
Based on previous CRISPR screening and RNA-seq data, the study team found that exocyst complex component 2 (EXOC2) is one important host factor for SARS-CoV-2 infection.
Importantly, it was found that the intracellular SARS-CoV-2 nucleocapsid (N) expression level was decreased to 3.7 % and the virus copy number in cell culture medium was decreased to 1.6 % by EXOC2 knockdown. Consistently, immunostaining results showed that N protein-positive cells were significantly decreased by EXOC2 knockdown.
The
SARS-CoV-2 Research team also found that EXOC2 knockdown downregulates SARS-CoV-2 infection by regulating IFNW1 expression. In conclusion, controlling the EXOC2 expression level may prevent SARS-CoV-2 infection and deserves further study
Currently, most COVID-19 therapeutics target the SARS-CoV-2 proteins. Because the viral proteins acquire genetic sequence mutations, the emergence of new variants has a risk to make the drugs ineffective.
The detailed analyses of host factors essential for SARS-CoV-2 infection in previous studies have focused on receptors and proteases, such as ACE2 and TMPRSS2, while other host factors have not been investigated sufficiently.
Lead researcher, Dr Kazuo Takayama Center for iPS Cell Research and Application, Kyoto University and his colleagues first predicted that EXOC2 is particularly important for SARS-CoV-2 infection based on previously published data from genetic analyses (CRISPR screening and RNA-seq analysis).
The study team confirmed that suppression of EXOC2 expression by the CRISPR interference system reduced the efficiency of SARS-CoV-2 infection, using ACE2-expressing iPS cells and airway organoids.
The study team also found that the decrease in SARS-CoV-2 infection efficiency by suppression of EXOC2 expression was mediated by increased expression of IFNW1.
The study findings indicate that EXOC2 is an e
ssential host factor for SARS-CoV-2 infection and that regulation of EXOC2 and IFNW1 expression may lead to develop new treatments for COVID-19.
The study findings were published in the peer reviewed journal: iScience.
https://www.sciencedirect.com/science/article/pii/S2589004222016996
The study findings demonstrated that EXOC2 is necessary for SARS-CoV-2 infection. CRISPRi-based and siRNA-based EXOC2 knockdown decreased the infection efficiency of SARS-CoV-2 in human iPS cells and AO, respectively. EXOC2 knockdown also increased the expression level of IFNW1, which has antiviral effects against SARS-CoV-2 and is one of the reasons for the antiviral effect mediated by EXOC2 knockdown.
The study team also found that the F-actin inhibitor Cytochalasin D significantly decreased the SARS-CoV-2 infection efficiency.
The study findings highlight that EXOC2 is a potential target for clinical therapeutics; especially, pharmacological EXOC2 inhibitors may prevent or reduce the impact of SARS-CoV-2 infection.
EXOC2/Sec5 is one of the subunits of the exocyst complex. It is also known as an effector for small GTPases in the Ras subfamily, RalA and RalB, which are involved in multiple cellular events, such as cell polarization, migration, apoptosis, cytokinesis, autophagy, host defense, tumorigenesis, and metastasis.
It is still not clear however as to whether the exocyst complex or other exocyst subunits are necessary for cellular homeostasis. Because cell viability was slightly reduced by EXOC2 knockdown, it is necessary to carefully consider toxicity of this knockdown when conducting drug discovery focusing on EXOC2.
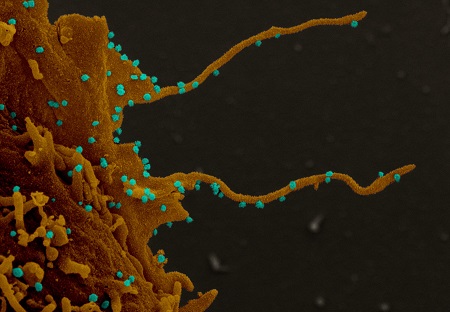
TNTs or Tunneling nanotubes, which are different from other known protrusions (filopodia, microvilli, and dendritic spines), are mainly composed of F-actin and regulated by EXOC2/Sec5.
https://pubmed.ncbi.nlm.nih.gov/19935652/
Tunneling nanotubes (TNTs) have been found particularly in immune cells and neurons cell lines and were proven to associate with many pathogens, especially viruses, such as HIV, herpesviruses, and influenza A.
Interestingly, viruses can hijack TNTs to facilitate viral entry, virus trafficking, cell-to-cell spread, and their protection from immune surveillance.
The signaling pathways that control TNT formation however seem context-dependence, and several EXOC2-independent pathways, such as the Rab35-ACAP2-ARF6-EHD1 cascade in neuron cells 37 and the MAPK pathway in squamous cancer cells 38, have been reported to control TNT formation. Hence, the potency of drugs targeting TNTs should be investigated in other somatic cells.
For the latest
SARS-CoV-2 Research, keep on logging to Thailand Medical News.
Read Also:
https://www.thailandmedical.news/news/breaking-covid-19-research-scientist-discover-unusual-way-sars-cov-2-infects-other-cells-once-inside-human-host-without-need-of-receptors
