Source: Thailand Medical News Dec 23, 2019 6 years, 4 months, 3 weeks, 3 days, 2 hours, 12 minutes ago
Researchers from University Of California, Los Angeles have now reported to
Thailand Medical News that they have devised a
diagnostic blood test that can quickly and sensitively
diagnoses Lyme disease at its early stages.
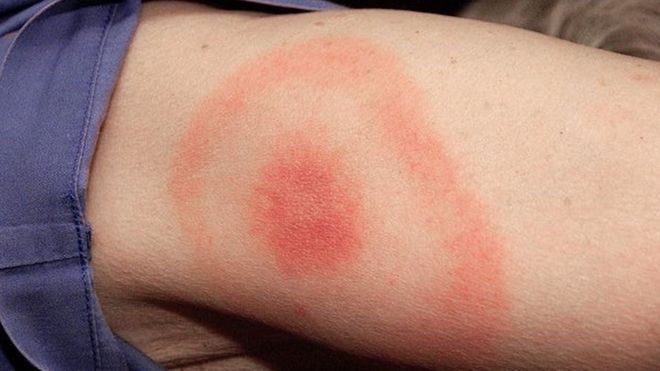
More 300,000 cases of
Lyme disease, which is caused by the tick-borne bacteria Borrelia burgdorferi, are diagnosed in the U.S. each year, according to the U.S. Centers for Disease Control and Prevention and globally the number of people contracting Lyme disease each year runs into millions.
Early symptoms of
Lyme disease include the characteristic skin rash, along with fever, headache, chills and muscle aches. If not treated promptly with antibiotics, more severe symptoms, such as facial palsy, nerve pain, heart palpitations and arthritis, can occur.
The problem is that about 10–20% of infected people do not develop the rash, and existing
diagnostic blood tests are slow, costly or insensitive at early stages, when treatment is most effective. Dr Aydogan Ozcan and colleagues from UCLA wanted to develop a fast, easy-to-use and inexpensive blood test to
diagnose Lyme disease soon after infection.
The medical researchers built a handheld, paper-based device to detect antibodies against the
B. burgdorferi bacteria in serum samples. The device included a sensing membrane that contained several spots covering seven bacterial antigens and a synthetic peptide. Antibodies from serum samples that attached to the spots were detected with a solution that changed color, depending on the amount of antibody captured.
The medical researchers took pictures of the color changes on a smart phone, then analyzed all of the spots with a neural network they developed that could determine whether the sample was positive or negative for
Lyme disease. When tested on 50 blood samples from people with or without early-stage Lyme disease, the assay had a specificity of 96.3% and a sensitivity of 85.7%. In addition to being much more sensitive than existing tests, the assay requires 15 minutes to complete and costs only 42 cents per test.
The team is planning further studies in order to pass necessary regulatory requirements to get the new
diagnostic platform commercially available in the market for clinics, hospitals and healthcare professional’s usage.
Reference : Joung et al. (2019) Point-of-Care Serodiagnostic Test for Early-Stage Lyme Disease Using a Multiplexed Paper-Based Immunoassay and Machine Learning. ACS Nano. DOI: https://doi.org/10.1021/acsnano.9b08151
