For the latest on Thailand Medical Industry, Thailand Doctors, Thailand Medical Research, Thailand Hospitals, Thailand Wellness Initiatives and the latest Medical News
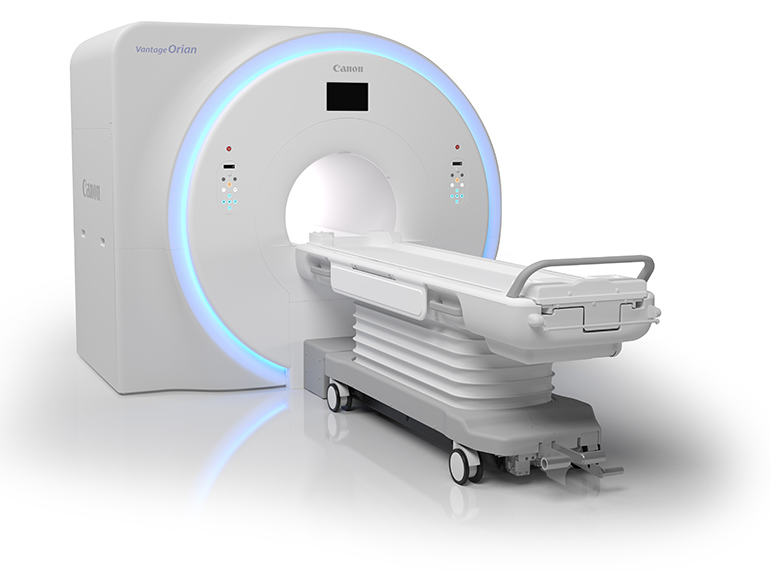
Won FDA clearance to bring to the U.S. market its brand new Vantage Orian 1.5 Tesla MRI scanner. It features a wide 71 centimeter bore and quiet operation thanks to its Pianissimo and Pianissimo Zen technologies. Pianissimo makes all scanning sequences quieter, while Pianissimo Zen allows certain scans to be performed at near ambient noise levels. To help young patients and those uncomfortable inside a scanner, Canon offers an immersive virtual experience called MR theater that combines audio and video to keep the person’s attention on something other than the scan.
The scanner was developed to maximize productivity and lower operating costs. Rapid Scan, an offering built into the scanner, allows many scans to be performed faster than before. The ForeSee View planning tool helps to prepare for scans to make sure rescans are not necessary. Additional technologies allow for cardiac and perfusion scans without the patient having to hold the breath.

“We are committed to offering our customers the premium diagnostic imaging tools they need to deliver accurate, confident and effective patient care,” said Dominic Smith, senior director, CT, PET/CT and MR Business Units, Canon Medical Systems USA, Inc. “The Vantage Orian was designed to increase productivity while ensuring patient comfort and delivering uncompromised clinical confidence.”