For the latest on Thailand Medical Industry, Thailand Doctors, Thailand Medical Research, Thailand Hospitals, Thailand Wellness Initiatives and the latest Medical News
Sepsis, a syndrome in which the body’s immune system overreacts to an infection, can have several life-threatening complications.
One of those is acute respiratory distress syndrome (ARDS), a lung complication involving inflammation and dysfunction of the endothelial layer. Sepsis induced ARDS is deadlier than other forms of ARDS, making it a top priority for hospitals and researchers alike.
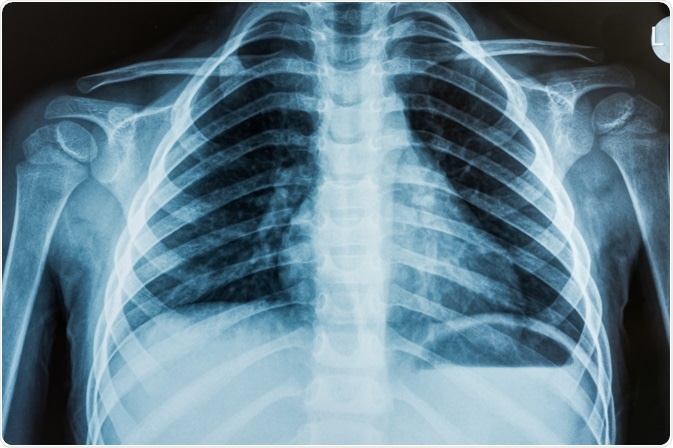
ARDS was initially called adult respiratory syndrome when it was first characterized in 1967. The first criteria to define the disease came in 1988 with the use of a four-point lung-scoring system.
This scoring system looks at measures of pressure in the lung, specifically positive end-expiratory pressure and ratio of partial pressure of arterial oxygen to the fraction of inspired oxygen, the compliance of the lung and the degree of infiltration as seen by radiographs of the chest.
While it is widely used in research, this criterion does not indicate survivability or severity when used in the first three days after onset and is therefore often not clinically useful.
In relation to sepsis, ARDS has a more rapid onset and a higher chance of resulting in mortality. In patients with severe sepsis, initial serum lactate levels and a microbiologically proven infection were both associated with increased occurrence of ARDS.
ARDS is a heterogeneous syndrome, meaning it can be caused by several different pathways. The general pathology involves increased permeability of the endothelial cells of the pulmonary capillary and of the epithelial cells of the alveoli.
Sepsis induced ARDS is often caused by indirect injury to the lung. However, pneumonia can directly injure the lung leading to ARDS and can also cause sepsis.
In preclinical model studies, the different mechanisms of direct and indirect injury pathways are being explored.
The pathway sepsis is more likely to work indirectly, as this pathway of injury has been shown to be due to inflammatory mediators. These mediators cause systemic endothelial damage, thereby causing the lung damage which precedes ARDS.
One example of this is defective signaling between inflammatory and hemostatic effector cells and the formation of neutrophil extracellular traps (NETs), which can cause injury to endothelial cells.
ARDS, once initiated, passes through several phases. First, the acute phase sees rapid respiratory failure in patients with risk factors for the disease.
Here, increased permeability of the alveolar-capillary barrier leads to protein-rich edema fluid moving into the air spaces. It is also linked to leukocytes and red blood cells translocating into the alveolar space.
In some cases, ARDS does not progress any further and comes to an end. In other cases, it progresses to fibrosing alveolitis accompanied by hypoxemia (low blood oxygen levels).
During recovery from this, the hypoxemia gradually dissolves and the lung compliance is improved. In most patients, lung function returns to normal levels.
As of now, there is no single treatment for ARDS. The treatment of sepsis-related ARDS does not differ from standard ARDS, where oxygenating tissue is the main goal.
The decreased lung function of ARDS means many patients are fitted with mechanical ventilators to mechanically push air into the lungs. The type of ventilation can differ and have significant effects on outcome.
Noninvasive ventilation has indicated to be useful for some patients, but not for those with hypoxemic respiratory failure. Invasive mechanical ventilation is generally more strongly advised.
This is occasionally used in addition to permissive hypercapnia (elevated carbon dioxide levels in the blood), especially in sepsis-induced ARDS.
Biological agents can be injected to reduce ARDS symptoms. One trial showed that injection with a neuromuscular blocking agent improved the outcome of sepsis-induced ARDS, while not affecting muscle weakness.
When neuromuscular blocking agents are administered early, the hospital mortality rate of patients with severe sepsis and ARDS was reduced. It therefore seems that these blocking agents can be used in patients afflicted with both conditions on a short-term basis when administered early on in disease onset.
Platelets are central to early phases of ARDS induced by sepsis. A common over the counter drug, aspirin, acts as a platelet inhibitor.
Preclinical models have shown that aspirin can be used to both prevent and treat sepsis-induced ARDS by hindering recruitment of neutrophils, neutralizing the inflammatory cascade, and reducing sequestration of platelets in the lungs.
This was backed up by clinical studies, where administration of aspirin reduced the development and mortality of severe sepsis and ARDS.