Japanese Scientists Discover That Ubiquilin2 Drives Dangerous Alpha-Synuclein Clumping in Parkinson’s Disease
Nikhil Prasad Fact checked by:Thailand Medical News Team Nov 29, 2025 6 months, 2 days, 9 hours, 23 minutes ago
Medical News: Ubiquilin2 Identified as a Key Driver of Harmful Protein Build Up in Parkinson’s Disease
A new discovery from scientists at Juntendo University Faculty of Medicine in Tokyo sheds light on how a little-known protein called ubiquilin2 may be helping Parkinson’s disease develop and worsen. Their work finally uncovers how this protein promotes the clumping of another protein known as alpha-synuclein, whose buildup in the brain forms the toxic Lewy bodies that damage nerve cells. This
Medical News report highlights how the discovery of this new mechanism could lead to future treatments.
 Researchers uncover how ubiquilin2 triggers alpha-synuclein clumping linked to Parkinson’s disease
Why Alpha-Synuclein Clumps Are So Dangerous
Researchers uncover how ubiquilin2 triggers alpha-synuclein clumping linked to Parkinson’s disease
Why Alpha-Synuclein Clumps Are So Dangerous
Parkinson’s disease develops when alpha-synuclein changes shape, sticks together and forms long fibers inside brain cells. These fibers gather into Lewy bodies, which slowly destroy the dopamine producing neurons responsible for movement control. Until now, scientists were unsure what triggered the early stages of this clumping inside living brain tissue.
How Ubiquilin2 Helps Alpha-Synuclein Form Toxic Aggregates
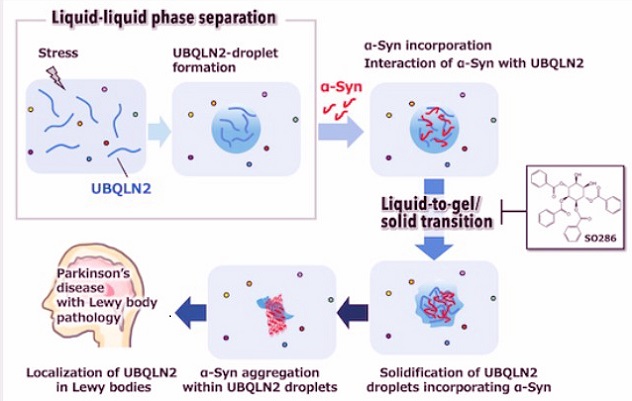
The Juntendo University team led by Professor Masaya Imoto, Professor Nobutaka Hattori, Dr Tomoki Takei and Dr Yukiko Sasazawa discovered that ubiquilin2 forms tiny liquid droplets inside cells through a natural process called liquid-liquid phase separation. These droplets act like microscopic reaction chambers. The researchers found that alpha-synuclein is pulled inside these droplets, where it binds strongly to a particular region of ubiquilin2 known as the STI1 2 domain. Once inside, alpha-synuclein is pushed toward forming fibrils far faster than it normally would. The team also examined brain tissue from patients with sporadic Parkinson’s disease and confirmed that ubiquilin2 was present directly inside the Lewy bodies in the substantia nigra.
Key Experiments Reveal the Hidden Process
Using fluorescent labeling and advanced microscopy, the scientists watched alpha-synuclein enter ubiquilin2 droplets and slowly convert into solid aggregates. Cell studies using the SH-SY5Y human cell line revealed the same pattern. Importantly, only ubiquilin2, not related proteins in the same family, was able to push alpha-synuclein through this dangerous transition. This suggests that its shifting from liquid droplets into gel like or solid like structures creates the perfect environment for alpha-synuclein to stiffen into fibers.
A Potential Treatment Candidate Emerges
The study also identified a promising small molecule called SO286 (1,2,3,6-tetra-O-benzoyl-muco-inositol). Researchers discovered that SO286 attaches to the same STI1 region of ubiquilin2 that alpha-synuclein uses. When bound, SO286 prevents ubiquilin2 from forming its droplets and blocks its interaction with alpha-synuclein. As a result, the harmful clumping process is dramatically reduced both in vitro and in c
ells. Early evidence even shows that SO286 can suppress the aggregation of tau, another damaging protein involved in Alzheimer’s disease.
What This Means for Future Parkinson’s Treatments
These findings suggest that stopping ubiquilin2 from forming droplets or blocking its interaction with alpha-synuclein could slow the development of Parkinson’s disease. The work from the Juntendo University researchers provides a strong new direction for drug development by showing that small molecules targeting this process could prevent Lewy body formation. While more testing is needed in patient derived cells and animal models, this discovery finally explains a missing step in how toxic protein buildup begins and continues in the Parkinson’s brain. It also shows how related neurodegenerative conditions may share similar mechanisms.
The study findings were published in the peer reviewed journal: The EMBO Journal.
https://www.embopress.org/doi/full/10.1038/s44318-025-00591-1
For the latest on neurodegenerative diseases, keep on logging to Thailand
Medical News.
Read Also:
https://www.thailandmedical.news/articles/alzheimer,-dementia-
