COVID-19 Scams: White House Controlled FDA Grants EUA Status To Regeneron’s Drug That Supposedly Costs About US$12,800 Per Treatment!
Source: COVID-19 Scams Nov 22, 2020 5 years, 6 months, 1 week, 15 hours, 54 minutes ago
COVID-19 Scams: More emerging news coming out from the land of COVID-19 Scams-America. After the hydrochloroquinone, convalescent plasma and remdesivir sagas, we have the Eli lily monoclonal drug and now the Regeneron’s antibody cocktail drug being granted emergency authorization or EUA status to treat COVID-19. It was already expected news as many were sure that Trump has to oblige his ‘arrangements’ with certain clandestine businessmen headed by billionaire Tom Cahill. We are also expected to see Moderna and Pzifers vaccine being granted approvals in coming weeks.
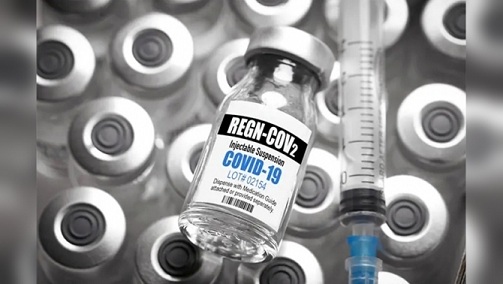
The U.S. Food and Drug Administration has granted emergency authorization for the experimental antibody treatment given to President Trump shortly after he was diagnosed with COVID-19. (No one is really sure whether Trump really had COVID-19 or was it another one of his scams to try to win the elections which he failed.)
REGEN-COV2 or Casirivimab/imdevimab is an artificial "antibody cocktail" designed to produce resistance to the SARS-CoV 2 coronavirus responsible for the COVID-19 pandemic. It consists of a mixture of two monoclonal antibodies, casirivimab (REGN10933) and imdevimab (REGN10987).
Regeneron's novel ‘treatment’ have shown promise in early studies at keeping the infection in check, reducing medical visits in patients who get the drug early in the course of their disease. A similar treatment, made by Eli Lilly, was given emergency approval earlier this month.
The problem is that it is very difficult to assess if such drugs really work or have any efficacy as they are being proposed to be given to individuals who contract SARS-CoV-2 in the early stages. No one can really predict 100 percent even with existing biomarkers that an individual is going to be headed for disease severity and that the drug protocol saved them! It’s a new ruse being deployed by greedy and unethical pharmaceutical and biotech companies and researchers who have been bought.
The emergency authorization for Regeneron’s drug is limited in scope: It is for people 12 and over who have tested positive for the coronavirus and who are at high risk for developing severe Covid-19. So called evidence so far suggests that antibody treatments work best early in the course of the disease, before the virus has gained a foothold in the body.
Like Eli Lilly’s treatment, Regeneron’s is not authorized for use in people who are hospitalized or who need oxygen.
Importantly, the approval raises immediate questions about who will get access to the treatments as an average of more than 165,000 individuals are diagnosed each day with COVID-19 in the United States and hospitals are running out of beds in some regions of the country. Regeneron has said it will have enough of the drug for only about 80,000 people by the end of November, enough for 200,000 patients by the first week of January, and 300,000 by the end of January. After that, the company said it will be able to ramp up production thanks to a partnership with the Swiss
manufacturer Roche.
It was initially speculated that the drug would cost about US$80,000 to US$120,000 per treatment (Monoclonal protocols are known to be extremely expensive.) Then it was speculated that Regeneron would drop the pricings to even as low as US$1,000 per dose but according to inside sources, the drugs are expected to costs US12,800 per treatment.
Interestingly Regeneron has received more than US$500 million from the Trump administration (tax payers monies and also thanks indirectly to Dr Anthony Fauci who also had his hands in the remdesivir saga) to develop and manufacture the treatments, a complex and time-consuming process.
It was said the American government will provide the first 300,000 doses free of charge, patients however may be charged for having the treatment administered; it must be infused in a clinic or hospital.
When questioned if Regeneron was giving these 300,000 doses or if the Trump administration was buying these 300,000 doses, both sides refused to answer.
Such antibody treatments have gotten less attention than vaccines, but health officials have long held out hope that they may serve as a possible bridge until a COVID-19 vaccine is more broadly available.
Two COVID-19 vaccines, one made by Pfizer and another by Moderna, recently claimed to be more than 90 percent effective in early analyses. Pfizer, which has completed its trial, submitted an application on Friday for emergency authorization of the vaccine, and Moderna said it also planned to apply soon. Still, it will be weeks before a vaccine is available, and even then, access will be limited to people in high-risk groups.
Regeneron’s president and chief scientific officer, Dr George D. Yancopoulos, said in a statement that he was encouraged by the recent vaccine results, but “there remains a need to treat patients who develop COVID-19, especially as some may not have had access to or were not protected by vaccination.”
The previously unknown and without a track record new biotech startup Regeneron enjoyed a burst of publicity in October, when Trump received an infusion of its cocktail and then enthusiastically promoted the drug as lending him a superpower-like feeling. In a video released on Oct. 7, the soon tie be outgoing American president claimed without evidence that it had cured him, and that he had authorized it ie something he does not have the power to do.
Interestingly it remains impossible to know whether the Regeneron treatment helped Mr. Trump. He was given multiple drugs while at Walter Reed National Military Medical Center, and while many people recover from the SARS-CoV-2 virus on their own.
The president and two of his top advisers Mark Meadows, the White House chief of staff, and Jared Kushner, Trump’s son-in-law have called Dr Stephen M. Hahn, the F.D.A. commissioner and a mere minion, to press for speed in agency reviews.
Dr Leonard S. Schleifer, the billionaire co-founder and chief executive of Regeneron, who has known Trump casually for years, has told associates that Trump calls him to ask about the status of the treatment.
To date, clinical evidence also shows that the Regeneron antibody treatments do not work well once people are sick enough to be hospitalized. Eli Lilly stopped giving its treatment to hospitalized patients in a government-run trial, because it said it did not seem to be helping those patients. And Regeneron paused enrolling the sickest hospitalized patients in one of its trials.
This causes a challenge for distributing the drugs, since they are only authorized for people who are not hospitalized, yet must be infused intravenously in a clinic or hospital.
Regeneron has claimed that the people who benefit the most from the treatment are those who have not yet mounted an antibody response and who also have high viral loads but learning who those people are would require separate tests that are not routinely given to patients who test positive for the virus. Company executives have acknowledged that at first, such tests may not be available, and the emergency authorization notes that people at high risk include those who are over 65, or have underlying conditions such as obesity or diabetes.
Giving the treatment to the right people will require quick turnarounds in testing, as well as coordination among federal, state and hospital officials many of the same challenges that have complicated the U.S. response to the pandemic.
It was reported that the distributor AmerisourceBergen will deliver Regeneron’s treatment on a weekly schedule based on the number of Covid-19 cases in each state. The federal government plans to work with state health officials to determine which hospitals and clinics should receive it.
The EUA or emergency use authorization was previously an obscure corner of regulatory law that mostly escaped broad public attention. But during the pandemic, it has become a centerpiece of the administration’s health policy: Since February, the agency has granted hundreds of emergency authorizations related to Covid-19, many for diagnostic tests and others for personal protective equipment, blood-purification devices, ventilators and therapies.
The EUA for Regeneron may still have the unintended effect of complicating the role of clinical trials in proving the treatment’s effectiveness for different age groups. If the drug becomes available more broadly, fewer people may want to sign up for clinical trials and risk the possibility that they may receive a placebo.
Other countries are also advised not to rush and follow suit in granting approvals for Regenerons or Eli lily drug protocols and even vaccines till after a 6 month period to see if there is truly any efficacy and these things work in America. The American regulatory agencies do not have a credible track record considering the previous sagas and nothing that is approved or claimed by America should be taken seriously considering the COVID-19 scams originating from that country with regards to drugs, therapeutics and even diagnostics.
For more on
COVID-19 Scams, keep on logging to Thailand Medical News.
