COVID-19 News: Scientists Unveil Molecular Mechanisms Underlying SARS-CoV-2 Hepatotropism And Liver Damage
Nikhil Prasad Fact checked by:Thailand Medical News Team Feb 06, 2024 2 years, 3 months, 3 weeks, 2 days, 20 hours, 52 minutes ago
COVID-19 19 News: The global impact of the COVID-19 pandemic has prompted researchers worldwide to delve deeper into the intricate ways in which the SARS-CoV-2 virus interacts with various organs. While its primary target is the respiratory system, emerging evidence suggests extrapulmonary involvement, notably in the liver.Researchers at Universidad de Buenos Aires (UBA) and Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) in Argentina have unveiled significant findings shedding light on the molecular mechanisms underlying SARS-CoV-2 hepatotropism and its consequential impact on liver health.
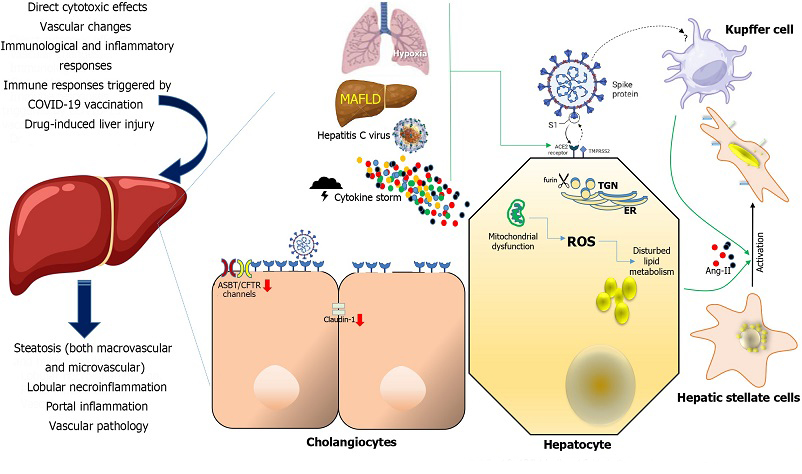 Mechanisms of severe acute respiratory syndrome coronavirus-2 disease-induced liver injury and their consequences at organ level (left). Severe acute respiratory syndrome coronavirus-2 cellular targets involved in liver damage (center and right). Various factors have been postulated to contribute to liver injury in the context of coronavirus disease 2019 (COVID-19), including direct cytotoxic effects, vascular changes, immunological and inflammatory responses associated with COVID-19, immune responses triggered by COVID-19 vaccination, and drug-induced liver injury. In the context of liver injury associated with COVID-19, the histological patterns encompass features such as steatosis (both macrovascular and microvascular), lobular necroinflammation, portal inflammation, and vascular pathology. At the cellular level, hypoxia, metabolic dysfunction-associated fatty liver disease, and concomitant hepatitis C virus infection, and the cytokine storm may upregulate the Angiotensin-converting enzyme-2 (ACE2), transmembrane serine protease 2 and furin expression in hepatocytes. Mitochondrial dysfunction has been affected directly by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection of hepatocytes which in turn may be connected to pre-existing inflammation and the adverse impacts of excessive and dysfunctional adipose tissue. In cholangiocytes, SARS-CoV-2 Leads to a decrease in the mRNA expression of Claudin-1 and downregulates the expression of hepatobiliary transporters, such as ASBT and the chloride channel CFTR. The ACE-2 expression in Kupffer cells is still controversial. Hepatic stellate cells appear do not express ACE2 in any activation state. Their activation is a pivotal event in the progression of chronic liver disease, as these cells serve as the primary source of fibrosis, and it is induced by proinflammatory and profibrotic signals, including angiotensin II, which is generated by the catalytic action of ACE as part of the profibrotic branch of the renin-angiotensin system. Liver and Kupffer cell are created with BioRender.com. ROS: Reactive oxygen species; ACE2: Angiotensin-converting enzyme-2; TMPRSS2: Transmembrane serine protease 2; MAFLD: metabolic dysfunction-associated fatty liver disease; ER: Endoplasmic reticulum; TGN: Trans-Golgi network.
Mechanisms of severe acute respiratory syndrome coronavirus-2 disease-induced liver injury and their consequences at organ level (left). Severe acute respiratory syndrome coronavirus-2 cellular targets involved in liver damage (center and right). Various factors have been postulated to contribute to liver injury in the context of coronavirus disease 2019 (COVID-19), including direct cytotoxic effects, vascular changes, immunological and inflammatory responses associated with COVID-19, immune responses triggered by COVID-19 vaccination, and drug-induced liver injury. In the context of liver injury associated with COVID-19, the histological patterns encompass features such as steatosis (both macrovascular and microvascular), lobular necroinflammation, portal inflammation, and vascular pathology. At the cellular level, hypoxia, metabolic dysfunction-associated fatty liver disease, and concomitant hepatitis C virus infection, and the cytokine storm may upregulate the Angiotensin-converting enzyme-2 (ACE2), transmembrane serine protease 2 and furin expression in hepatocytes. Mitochondrial dysfunction has been affected directly by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection of hepatocytes which in turn may be connected to pre-existing inflammation and the adverse impacts of excessive and dysfunctional adipose tissue. In cholangiocytes, SARS-CoV-2 Leads to a decrease in the mRNA expression of Claudin-1 and downregulates the expression of hepatobiliary transporters, such as ASBT and the chloride channel CFTR. The ACE-2 expression in Kupffer cells is still controversial. Hepatic stellate cells appear do not express ACE2 in any activation state. Their activation is a pivotal event in the progression of chronic liver disease, as these cells serve as the primary source of fibrosis, and it is induced by proinflammatory and profibrotic signals, including angiotensin II, which is generated by the catalytic action of ACE as part of the profibrotic branch of the renin-angiotensin system. Liver and Kupffer cell are created with BioRender.com. ROS: Reactive oxygen species; ACE2: Angiotensin-converting enzyme-2; TMPRSS2: Transmembrane serine protease 2; MAFLD: metabolic dysfunction-associated fatty liver disease; ER: Endoplasmic reticulum; TGN: Trans-Golgi network.
Thailand
Medical News had previously covered a number of studies that also warned of the growing threat of SARS-CoV-2 infecting the liver and causing liver damage!
https://www.thailandme
dical.news/news/breaking-news-brazilian-study-shockingly-finds-that-sars-cov-2-directly-infects-liver-cells-or-hepatocytes-to-cause-hyperglycemia
https://www.thailandmedical.news/news/argentinian-researchers-warn-that-sars-cov-2-could-be-a-hepatotropic-virus-that-also-manipulates-glucose-metabolism
https://www.thailandmedical.news/news/new-york-clinicians-sound-the-alarm-covid-19-asymptomatic-individuals-can-suffer-acute-liver-injury
https://www.thailandmedical.news/news/covid-19-research-the-detrimental-effects-of-tnf%CE%B1-on-liver-during-sars-cov-2-infection-and-its-impact-on-patient-survival
https://www.thailandmedical.news/news/breaking-covid-19-news-brazil-study-shows-that-most-long-covid-individuals-continue-to-sustain-damaged-liver-functions-up-to-20-months
https://www.thailandmedical.news/news/covid-19-news-fatty-acids-impact-ace2-receptor-expression-in-metabolic-dysfunction-associated-steatotic-liver-disease
https://www.thailandmedical.news/news/breaking-covid-19-news-scientists-from-university-of-texas-warn-that-sars-cov-2-can-also-cause-seronegative-autoimmune-hepatitis-in-adults
https://www.thailandmedical.news/news/covid-19-news-german-researchers-warn-that-sclerosing-cholangitis-emerging-as-a-consequence-of-severe-covid-19-infection
https://www.thailandmedical.news/news/coronavirus-news-german-study-confirms-that-sars-cov-2-coronavirus-can-invade-and-damage-the-liver
https://www.thailandmedical.news/news/breaking-covid-19-research-studies-warn-of-high-prevalence-of-liver-injury-in-covid-19-patients
https://www.thailandmedical.news/news/covid-19-research-university-of-bristol-researchers-warn-that-liver-complications-are-common-in-covid-19-patients
https://www.thailandmedical.news/news/breaking-more-emerging-chinese-research-studies-shows-that-the-sars-cov-2-coronavirus-also-attacks-the-kidneys,-pancreas-and-liver
https://www.thailandmedical.news/news/breaking-covid-19-news-sars-cov-2-that-causes-liver-impairment-in-many-also-cause-hypoalbuminemia
https://www.thailandmedical.news/news/breaking-sars-cov-2-impairs-lipid-metabolic-and-autophagic-pathways,-causing-damage-to-heart,-liver-and-kidneys-the-phytochemical-trigonelline-helps
https://www.thailandmedical.news/news/researchers-alarmingly-find-that-metabolic-associated-fatty-liver-disease-mafld-may-be-a-prevalent-long-covid-manifestation
https://www.thailandmedical.news/news/breaking-sars-cov-2-infection-induces-increase-of-gp73-that-causes-dysglycaemia-increased-gp73-could-also-imply-future-liver-disease-and-liver-cancer
https://www.thailandmedical.news/news/breaking-study-finds-that-upregulated-interleukin-6-drives-development-of-rare-liver-cancers-covid-19-infections-causes-elevated-il-6-levels-in-many
https://www.thailandmedical.news/news/are-the-new-sars-cov-2-variants-circulating-in-europe-more-neuropathogenic-with-a-modified-pathogenesis-brain-and-liver-issues-on-the-rise
https://www.thailandmedical.news/news/belgium-researchers-warn-that-more-hospitalized-covid-19-patients-are-manifesting-a-rare-chronic-liver-disease-known-as-sclerosing-cholangitis-could-b
Liver damage may be associated with COVID-19 due to direct injury of the viral infection to the liver cells, delayed resolution of inflammation, and viral persistence.
https://pubmed.ncbi.nlm.nih.gov/32489653/
https://pubmed.ncbi.nlm.nih.gov/35105985/
Past studies suggested that angiotensin-converting enzyme 2 (ACE2) expression is enriched in cholangiocytes and that SARS-CoV-2 can bind directly to ACE-positive cholangiocytes, thereby deregulating liver function.
https://pubmed.ncbi.nlm.nih.gov/33332624/
https://pubmed.ncbi.nlm.nih.gov/32170806/
Many studies have reported changes in serum markers in long-term COVID, including inflammatory (C-reactive protein) and liver markers (alanine aminotransferase [ALT] and aspartate aminotransferase [AST]).
https://pubmed.ncbi.nlm.nih.gov/33887749/
https://pubmed.ncbi.nlm.nih.gov/33347790/
https://pubmed.ncbi.nlm.nih.gov/33532785/
https://pubmed.ncbi.nlm.nih.gov/33172844/
Background on SARS-CoV-2
SARS-CoV-2, a member of the Betacoronavirus genus within the Coronaviridae family, is a positive-sense, single-stranded RNA virus with an enveloped structure. Sharing genetic similarities with SARS-CoV-1 and Middle East respiratory syndrome CoV, SARS-CoV-2's genome encodes essential structural proteins like spike (S), envelope (E), membrane (M), and nucleocapsid (N). The spike protein plays a critical role in viral attachment, fusion, entry, and transmission.
Hepatotropism and Liver Damage
While the respiratory system is the primary battleground for SARS-CoV-2, extrapulmonary organ involvement, particularly in the liver, has become a growing concern. Hepatic tissues have been shown to harbor viral RNA, and in situ hybridization revealed the presence of virions in blood vessels and endothelial cells. Electron microscopy further confirmed viral particles within hepatocytes, emphasizing the need for a comprehensive understanding of hepatotropism and direct cytopathic effects contributing to COVID-19-related liver injury.
Factors Contributing to Liver Injury
Several factors contribute to liver injury in the context of COVID-19, including direct cytotoxicity, vascular changes, inflammatory responses, immune reactions triggered by COVID-19 and vaccinations, and drug-induced liver injury. Though a typical hepatitis presentation is not extensively documented, elevated liver biochemical markers are common in hospitalized COVID-19 patients, primarily showing a hepatocellular pattern of elevation. Long-term studies suggest that progressive cholestasis may affect 20% of patients with chronic liver disease post-SARS-CoV-2 infection.
Molecular Mechanisms Underlying SARS-CoV-2 Hepatotropism
Understanding the molecular mechanisms underlying SARS-CoV-2 infection in the liver is a complex endeavor. This
COVID-19 News report, highlights key aspects, including the expression of the Angiotensin-converting enzyme-2 (ACE2) receptor in liver cells, the role of inflammatory responses, the impact of hypoxia, the involvement of the liver's vascular system, the infection of bile duct epithelial cells, the activation of hepatic stellate cells, and the contribution of monocyte-derived macrophages. Pre-existing liver conditions are noted to worsen outcomes in COVID-19 patients.
ACE2 and Viral Entry Co-Factors Expression in Hepatic Cells
The expression of ACE2, the primary receptor for SARS-CoV-2, in liver cells remains a critical research challenge. While the liver exhibits minimal ACE2 and TMPRSS2 protein expression, studies using single-cell RNA sequencing on healthy human livers revealed detectable ACE2 expression in cholangiocytes, the epithelium lining the bile duct. Notably, ACE2 expression increases in hepatocytes in the presence of liver fibrosis or cirrhosis. Hepatocellular carcinoma-derived cell lines have shown the ability to support the entire viral life cycle, emphasizing the role of ACE2 and TMPRSS2 in liver parenchymal cells.
ACE2-Independent SARS-CoV-2 Hepatotropism
Beyond ACE2, additional receptors, such as the high-density lipoprotein scavenger receptor B type 1 (SR-B1), may play a facilitating role in ACE2-dependent coronavirus attachment. Immunohistochemistry analysis of liver tissue has confirmed sporadic ACE2 expression, while ganglioside (GM1) may influence the interaction between the SARS-CoV-2 spike protein and ACE2. Comprehensive research into the S protein-ACE2 interactome is crucial for understanding the molecular mechanisms involved in SARS-CoV-2 infection.
Experimental Models for Studying SARS-CoV-2 Hepatotropism
Various experimental models, including HuH7 cells and human liver organoids, have been employed to study SARS-CoV-2 hepatotropism. HuH7 cells, derived from hepatocellular carcinoma, have been identified as a permissive model for studying viral interactions. Post-mortem autopsies have provided evidence supporting the direct infection of liver cells by SARS-CoV-2, with electron microscope imaging revealing viral structures within hepatocytes. Organoids generated from human hepatocytes have confirmed SARS-CoV-2 hepatotropism, showcasing substantial viral replication and associated changes in gene expression.
The Relevance of Cholangiocytes as SARS-CoV-2 Cellular Targets in the Liver
Cholangiocytes, or bile duct epithelial cells, play pivotal roles in bile generation, regulation, and immune responses. Single-cell sequencing of long-term liver ductal organoid cultures has revealed the persistence of ACE2 and TMPRSS2 expression in cholangiocytes. In vitro studies have demonstrated SARS-CoV-2 infection in human liver ductal organoids, raising the possibility of viral replication within bile duct epithelium in vivo. Disruptions in tight junctions in cholangiocytes could contribute to liver injury, allowing toxic bile components to leak into adjacent liver parenchyma.
Are Kupffer Cells and Hepatic Stellate Cells Susceptible to SARS-CoV-2 Infection?
Kupffer cells, liver-resident macrophages, do not express ACE2 according to current evidence. However, monocyte-derived macrophages, infiltrating the liver in response to inflammation, may act as carriers of the virus, facilitating infection of ACE2-expressing cells. Hepatic stellate cells, crucial in fibrosis development, do not express ACE2, but the pro-inflammatory environment initiated by COVID-19 may activate these cells, contributing to fibrosis. Pre-existing chronic liver diseases, especially metabolic dysfunction-associated fatty liver disease (MAFLD), may exacerbate the severity of COVID-19 and contribute to long-term effects such as fibrosis.
Conclusion
The evolving understanding of SARS-CoV-2 hepatotropism and its impact on liver health underscores the need for continued research. The intricate interplay between the virus and the liver, involving ACE2, cholangiocytes, Kupffer cells, and hepatic stellate cells, presents a complex landscape that necessitates thorough investigation. Monitoring and managing liver function in COVID-19 patients remains an important feature and individuals exposed to the SARS-CoV-2 virus should go for frequent health screenings.
The study findings were published as an editorial in the peer reviewed journal: World Journal of Hepatology.
https://www.wjgnet.com/1948-5182/full/v16/i1/1.htm
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
Read Also:
https://www.thailandmedical.news/news/breaking-covid-19-news-greek-researchers-warn-that-mild-covid-19-infections-can-cause-hepatitis-b-virus-hbv-reactivation
https://www.thailandmedical.news/news/brazilian-plasma-metabolome-study-finds-massive-alterations-in-COVID-19-lipid-and-amino-acid-metabolism-in-severe-and-fatal-covid-19-cases
https://www.thailandmedical.news/news/breaking-a-must-for-all-doctors-to-read-scientists-warn-that-covid-19-affects-cytochrome-p450-3a4-mediated-drug-metabolism-and-drug-interactions
https://www.thailandmedical.news/news/breaking-news-greek-researchers-warn-that-mild-covid-19-can-reactivate-hepatitis-b-virus-hbv-even-in-absence-of-immunosuppressive-therapy
