COVID-19 News: UK Led Study Finds That The SARS-CoV-2 Protein ORF3c Is A Mitochondrial Modulator Of Innate Immunity
Nikhil Prasad Fact checked by:Thailand Medical News Team Oct 01, 2023 2 years, 7 months, 4 weeks, 1 day, 1 hour, 23 minutes ago
COVID-19 News: The COVID-19 pandemic, caused by the novel coronavirus SARS-CoV-2, has had far-reaching consequences on global health and the economy. Understanding the intricate interactions between the virus and the host's immune system is crucial for devising effective therapeutic strategies. This
COVID-19 News report covers a new study conducted by researchers from the University of Cambridge, University of Manchester, University Medical Center Göttingen, University of Liverpool, University of Bristol, the Broad Institute of MIT and Harvard, and MIT's Computer Science and Artificial Intelligence Laboratory, that sheds light on a previously overlooked accessory protein encoded by SARS-CoV-2, known as ORF3c, and its role as a mitochondrial modulator of innate immunity.
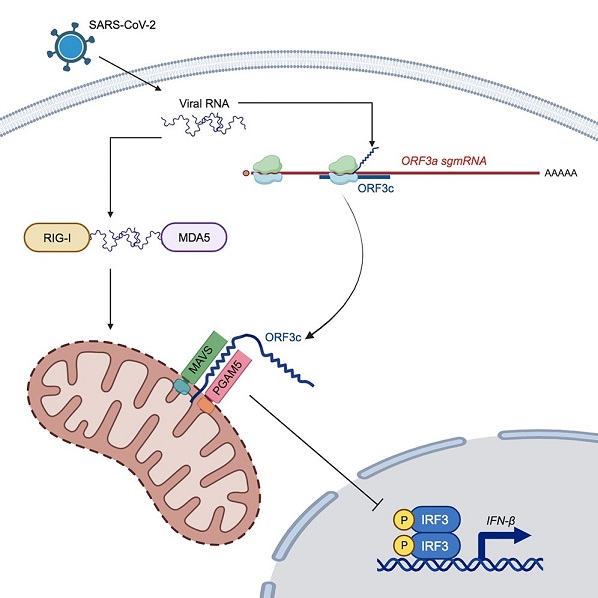 Graphical Abstract
The SARS-CoV-2 Genome and Accessory Proteins
Graphical Abstract
The SARS-CoV-2 Genome and Accessory Proteins
SARS-CoV-2 belongs to the Sarbecovirus subgenus within the Coronaviridae family, characterized by large, positive-sense, single-stranded RNA genomes with approximately 30 kilobases in size. These genomes contain several protein-coding open reading frames (ORFs), including ORF1a, ORF1b, and a set of nested subgenomic mRNAs (sgmRNAs) that encode structural and accessory proteins.
While structural proteins are essential for viral replication, accessory proteins, despite being dispensable in cell culture, play crucial roles in evading host immune responses.
The Challenge of Identifying Novel ORFs
Identifying the complete repertoire of viral proteins encoded by SARS-CoV-2 has been a complex task due to the rapid emergence of the virus and the reliance on comparative genomics. These challenges stem from the genetic diversity within the Coronaviridae family and the lack of experimental validation for many predicted ORFs. As a result, the establishment of a definitive SARS-CoV-2 viral proteome has been hindered, leaving numerous unexplored proteins with potential roles in viral replication and host interaction.
Discovery of ORF3c: A Hidden Accessory Protein
In 2020, utilizing comparative genomic approaches and analyzing the coding capacity of SARS-CoV-2 using available sarbecovirus genomes, researchers identified a previously undetected ORF, ORF3c, overlapping with ORF3a. Notably, this ORF3c was also present in the 2002–2003 SARS-CoV-1 outbreak. Despite its evolutionary conservation, ORF3c remained largely unexplored and absent from prior studies. The study by these researchers brought ORF3c into the spotlight, shedding light on its potential significance in viral replication, immune evasion, or transmission.
The Immune Response and Type I Interferon (IFN)
Understanding the immune response
is essential in the context of SARS-CoV-2 infection. The type I interferon (IFN) response, a critical component of innate immunity, plays a pivotal role in limiting viral infections. Host cells produce type I IFNs in response to activation of pattern recognition receptors (PRRs), which recognize pathogen-associated molecular patterns (PAMPs). Activation of PRRs leads to the transcription of type I IFNs and other antiviral genes, initiating a host defense mechanism against the virus.
Key Cytoplasmic PRRs: RIG-I and MDA5
Two central cytoplasmic PRRs responsible for detecting viral RNA and initiating the antiviral response are retinoic acid-inducible gene (RIG-I) and melanoma differentiation-associated protein 5 (MDA5). RIG-I detects short double-stranded RNA (dsRNA) and 5'-ppp/pp-RNA, while MDA5 senses high molecular weight dsRNA and mRNA lacking 2'-O-methylation at the 5' cap. These PRRs engage with mitochondrial antiviral-signaling protein (MAVS) to trigger downstream signaling events.
The Role of MAVS in Antiviral Response
Upon activation by RIG-I or MDA5, MAVS forms polymers at the mitochondrial outer membrane, activating transcription factors such as IFN regulatory factor 3 (IRF3) and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB). These activated transcription factors translocate into the nucleus and stimulate the transcription of type I IFNs and other pro-inflammatory factors, setting off the antiviral response.
SARS-CoV-2 ORF3c: A Mitochondrial Modulator of Innate Immunity
The researchers in this study conducted a comprehensive analysis of ORF3c's function and localization within host cells. They discovered that ORF3c localizes to the mitochondrial outer membrane (MOM), a crucial platform for innate immune signaling. Importantly, ORF3c was found to subvert the host's antiviral response by inhibiting the production of interferon-beta (IFN-β).
ORF3c's Interactions and Mechanisms
ORF3c's immune-evasive functions were found to involve interactions with two critical proteins, MAVS and PGAM5. ORF3c induces cleavage of MAVS by caspase-3, leading to a reduction in IFN-β production. However, it's noteworthy that ORF3c does not impact NF-κB-driven transcripts, suggesting specificity in its inhibitory effects on the IFN-β pathway.
Comparative Analysis of SARS-CoV-2 Accessory Proteins
The study also highlighted the distinct mechanisms employed by various SARS-CoV-2 accessory proteins to inhibit host immune responses. While some proteins directly reduce IFN-β levels or affect transcription factors, ORF3c stands out as the only conserved sarbecoviral protein that directly binds to MAVS within the MOM.
Comparative Analysis of ORF3c with Other SARS-CoV-2 Proteins
Comparative analysis of ORF3c with other SARS-CoV-2 proteins revealed its unique role as a mitochondrial modulator of innate immunity. Unlike other viral proteins that affect IFN-β signaling, ORF3c operates directly within the mitochondria and interacts with MAVS and PGAM5 to suppress the antiviral response.
Conclusion
The discovery of ORF3c as a critical modulator of innate immunity sheds light on a previously unrecognized aspect of SARS-CoV-2 infection. This research not only expands our understanding of the virus-host interactions but also provides potential targets for therapeutic interventions. ORF3c's ability to specifically inhibit the IFN-β pathway via interactions with MAVS and PGAM5 represents a unique mechanism among SARS-CoV-2 accessory proteins. Further investigations into ORF3c and its implications for viral pathogenesis may pave the way for novel strategies to combat COVID-19 and future coronaviral infections.
The study findings were published in the peer reviewed journal: iScience.
https://www.sciencedirect.com/science/article/pii/S2589004223021570
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
