Taiwanese Study Shows That SARS-CoV-2 Variants More Importantly Have Unique Differences In Spike Structure Besides Having Different Mutations!
Source: SARS-CoV-2 Variants Sep 19, 2021 4 years, 8 months, 1 week, 3 days, 12 hours, 32 minutes ago
SARS-CoV-2 Variants: A new study by Taiwanese researchers from the Academia Sinica and National Taiwan University along with support by scientists from the Medical University of Lublin-Poland and Johns Hopkins University-USA has found that the different SARS-CoV-2 variants not only have different mutations and deletions on them but most importantly they exhibit different but unique spike structures.

The study team reported 38 cryo-EM structures, corresponding to the spike protein of the Beta (B.1.351), Gamma (P.1), Delta (B.1.617.2) and Kappa (B.1.617.1) variants in different functional states with and without its receptor, ACE2.
Interestingly it was found that mutations on the N-terminal domain not only alter the conformation of the highly antigenic supersite of the Delta variant, but also remodel the glycan shield by deleting or adding N-glycans of the Delta and Gamma variants, respectively. Substantially enhanced ACE2 binding was observed for all variants, whose mutations on the receptor binding domain modulate the electrostatics of the binding interfaces. Despite their abilities to escape host immunity, all variants can be potently neutralized by three unique antibodies.
The study findings were published on a preprint server and are currently being peer reviewed.
https://www.biorxiv.org/content/10.1101/2021.09.12.459978v1
The
SARS-CoV-2 Variants have been an ongoing concern since their first emergence initially with the Alpha variant or B1.1.7 that was discovered in the UK and which quickly became dominant globally. Since then, more variants have been discovered, including Beta (B1.351), Gamma (P.1), and the current biggest threat, the Delta variant (B.1.617.2). The Delta variant emerged in India in early 2021 and now accounts for more than 90% of new cases.
The various emerging variants are recognized by mutations of the highly glycosylated spike protein.
This spike protein is largely responsible for the pathogenicity of the SARS-CoV-2 coronavirus. The receptor-binding domain (RBD) within the N-terminal S1 subunit of the spike protein binds to the angiotensin-converting enzyme 2 (ACE2) receptors presented by human cells, allowing viral entry.
Importantly the alterations caused by the mutations to this protein can reduce the effectiveness of treatments such as monoclonal antibodies and lower the effectiveness of vaccines.
It has been found for instance that both the Pfizer and Moderna vaccines have shown up to a 12-fold drop in vaccine effectiveness against the Beta variant.
In order to investigate the impact of these mutations, the study team performed cryo-electron microscopy analysis on the variants spike proteins to determine any structural changes.
The study team examined the structural effect of the mutations in the Beta, Gamma, Delta, and Kappa variants and determined the structures of the 25 spike protein variants.
Importantly it was found that in the Beta variant, the RBD showed three states that were mostly fully open (all RBDs in the up conformation) and two partial
ly open, with one or two RBDs in the up conformation. The structure was also solved without any furin cleavage site substitutions or stabilizing tandem profile to confirm the unusual triple RBD-up conformation.
However the Gamma spike protein did not show the same fully open state, only the two partially open states (1 RBD-up and 2 RBD-up), but there were two substates for the 1-RBD-up state when the structure was examined three-dimensionally.
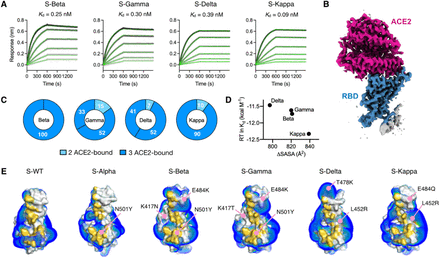 Kinetics and structural basis of receptor ACE2 binding by the SARS-CoV-2 variants. (A) BLI sensorgrams of immobilized ACE2 binding to the S variants. The sensorgrams of the 100, 50, 25, 12.5, 6.25 and 3.125 nM S variants are colored from black to light grey in descending orders superimposed with the fitting results in green. The corresponding Kd values are shown above. (B) Representative cryo-EM map of the RBD S-Delta in complex with ACE2 is colored in blue and magenta for RBD and ACE2, respectively. (C) Distributions of distinct receptor ACE2 binding stoichiometry and quaternary conformations classified according to the number of RBD-bound ACE2. The coloring scheme is shown below. (D) Correlation between the free energy of ACE2 binding and ΔSASA. (E) Surface representations of the RBD of the individual variants with the binding interfaces of the S variants highlighted in gold. Positive isosurfaces of the RBD of the S variants were generated by the APBS module within PyMol. The positions of individual mutations are indicated by pink lines with their changes indicated. The structure of S-WT RBD in complex with ACE2 is taken from the PDB ID 6M0J. The structure of S-Alpha RBD in complex with ACE2 is taken from the PDB ID 7EDJ
Kinetics and structural basis of receptor ACE2 binding by the SARS-CoV-2 variants. (A) BLI sensorgrams of immobilized ACE2 binding to the S variants. The sensorgrams of the 100, 50, 25, 12.5, 6.25 and 3.125 nM S variants are colored from black to light grey in descending orders superimposed with the fitting results in green. The corresponding Kd values are shown above. (B) Representative cryo-EM map of the RBD S-Delta in complex with ACE2 is colored in blue and magenta for RBD and ACE2, respectively. (C) Distributions of distinct receptor ACE2 binding stoichiometry and quaternary conformations classified according to the number of RBD-bound ACE2. The coloring scheme is shown below. (D) Correlation between the free energy of ACE2 binding and ΔSASA. (E) Surface representations of the RBD of the individual variants with the binding interfaces of the S variants highlighted in gold. Positive isosurfaces of the RBD of the S variants were generated by the APBS module within PyMol. The positions of individual mutations are indicated by pink lines with their changes indicated. The structure of S-WT RBD in complex with ACE2 is taken from the PDB ID 6M0J. The structure of S-Alpha RBD in complex with ACE2 is taken from the PDB ID 7EDJ
Significantly however, the Delta variant showed the most structural variation, including an unusually fully closed conformation in which all RBD’s were in the down conformation. There were also five 1-RBD up substates and three 2-RBD up substates.
Also the most unusual or strange results were seen in the Kappa spike protein, with a previously unseen head-to-head dimer, with all three RBD’s in all three spike proteins binding to another RBD from another spike protein. This structure could be resolved into three substates. Two 1 RBD-up substates were seen, and a single 2 RBD-up conformation and the uncommon all RBD down conformation.
According to the study team, it is thought that these altered conformations allow at least the Gamma and Delta variants to escape immune responses from previous strains, as the chromosomal rearrangements alter the glycan shield around the supersite normally targeted by neutralizing antibodies.
It was also found that almost all of these structural variants also showed greatly enhanced ACE2 binding, with the Kappa variant binding the strongest.
Upon analysis using cryo-electron microscopy, the study team found that the Kappa spike protein dimers showed a reduced negative charge, allowing a much stronger electrostatic binding to the negatively charged ACE2.
Several other mutations in alternate variants had the same effect, to a lesser extent.
The study team had also identified three effective monoclonal neutralizing antibodies against wild-type SARS-CoV-2, the Alpha variant, and D614G, a mutation common to both the Delta and Kappa strains. These are RBD-chAb -15, -25 and -45.
However it was found that RBD-chAb-15 is not effective against the Alpha, Beta, or Gamma variants protected by the N501Y mutation. Still, a mix of the other two antibodies can neutralize both the Alpha variant and variants carrying D614G.
The study findings help further the understanding of the COVID-19 pandemic.
The stud team highlights the benefit of knowing the different structural conformations of the variants and the mechanisms by which they escape immunity from common vaccines and monoclonal antibody treatments.
Importantly the ability of all three discovered neutralizing antibodies to combat the widely spread Delta variant is of great importance, as is the versatility of RBD-chAB-45 in recognizing different states of conformation of the RBD, as it binds to the site la/class 2 on the RBD of the spike protein, which is accessible in either up or down conformations.
Important takeaways from this study include the insight into N-terminal mutations in the variants, which in many cases completely avoid the neutralizing ability of N-terminal domain-specific neutralizing antibodies, further highlighting the importance of targeting the RBD. Therefore, nAbs that target the RBD remain a preferred route for success, and several RBD-specific nAbs are currently approved for emergency use to treat COVID-19 patients.
https://pubmed.ncbi.nlm.nih.gov/34416148/
For more on
SARS-CoV-2 Variants, keep on logging to Thailand Medical News.

