GREAT NEWS! Most Post COVID Individuals Will Develop Lung Issues Similar To Idiopathic Pulmonary Fibrosis (IPF) With Potential Serious Outcomes!
Source: Medical News - Post COVID Idiopathic Pulmonary Fibrosis Jul 21, 2022 3 years, 9 months, 2 weeks, 22 hours, 56 minutes ago
A new study by researchers from University of California-San Diego has found that the lung issues that many Post COVID individuals develop are similar to Idiopathic Pulmonary Fibrosis (IPF) with potential serious and fatal outcomes in months or years that follow!
Utilizing an AI-guided approach, the study team analyzed more than 1000 human lung transcriptomic datasets associated with various lung conditions using two viral pandemic signatures (ViP and sViP) and one covid lung-derived signature.
Upon identifying similarities between COVID-19 and idiopathic pulmonary fibrosis (IPF), the study team subsequently dissected the basis for such similarity from molecular, cytopathic, and immunologic perspectives using a panel of IPF-specific gene signatures, alongside signatures of alveolar type II (AT2) cytopathies and of prognostic monocyte-driven processes that are known drivers of IPF.
Subsequently, transcriptome-derived findings were used to construct protein-protein interaction (PPI) network to identify the major triggers of AT2 dysfunction. Key findings were validated in hamster and human adult lung organoid (ALO) pre-clinical models of COVID-19 using immunohistochemistry and qPCR.
The study findings showed that Post COVID-19 lung issues resembles IPF at a fundamental level; it recapitulates the gene expression patterns (ViP and IPF signatures), cytokine storm (IL15-centric), and the AT2 cytopathic changes, e.g., injury, DNA damage, arrest in a transient, damage-induced progenitor state, and senescence-associated secretory phenotype (SASP).
These immunocytopathic features were induced in pre-clinical COVID models (ALO and hamster) and reversed with effective anti-CoV-2 therapeutics in hamsters. PPI-network analyses pinpointed ER stress as one of the shared early triggers of both diseases, and IHC studies validated the same in the lungs of deceased subjects with COVID-19 and SARS-CoV-2-challenged hamster lungs. Lungs from tg-mice, in which ER stress is induced specifically in the AT2 cells, faithfully recapitulate the host immune response and alveolar cytopathic changes that are induced by the SARS-CoV-2 virus.
Similar to IPF, Post COVID-19 lung issues may be driven by injury-induced ER stress that culminates into progenitor state arrest and SASP in AT2 cells. The ViP signatures in monocytes may be key determinants of prognosis. The insights, signatures, disease models identified here are likely to spur the development of therapies for patients with IPF and other fibrotic interstitial lung diseases.
The study findings were published in the peer reviewed journal: eBio Medicine.
https://www.thelancet.com/journals/ebiom/article/PIIS2352-3964(22)00366-8/fulltext
Although most individuals recover from COVID-19 within a week or two, up to one-third of survivors experience persistent or new symptoms weeks and months after initial infection that is now typically recognized as Long COVID or PASC (Post-Acute Sequelae of SARS CoV-2 infection)
Interesting, a sizeable number of "long COVID" patients exhibit interstitial lung disease (ILD), a group of chronic pulmonary disorders characterized by inflammation and scarring (fibrosis) that make it hard for the lungs to get enough oxygen. Little is currently known about ILD, from diagnosis to prognosis to management. I
n its most severe form, the disease is fatal without a lung transplant.
https://www.sciencedirect.com/science/article/pii/S2213260020302228
https://www.atsjournals.org/doi/10.1513/AnnalsATS.202008-1002OC
https://pubs.rsna.org/doi/10.1148/radiol.2021204482
https://www.atsjournals.org/doi/10.1513/AnnalsATS.202102-223ED
https://www.sciencedirect.com/science/article/pii/S2214250920303498
https://onlinelibrary.wiley.com/doi/full/10.1002/jum.15563
https://www.sciencedirect.com/science/article/pii/S2213260020302253
The study findings from the University of California-San Diego study is the first to provide the first insights into the fundamental cellular pathologies that drive ILD.
Co-senior study author Dr Pradipta Ghosh, MD, a Professor in the departments of Medicine and Cellular and Molecular Medicine at University of California-San Diego School of Medicine told Thailand
Medical News, "Utilizing an artificial intelligence (AI) approach,
we found that lung fibrosis caused by COVID-19 resembles idiopathic pulmonary fibrosis (IPF), the most common and the deadliest form of ILD."
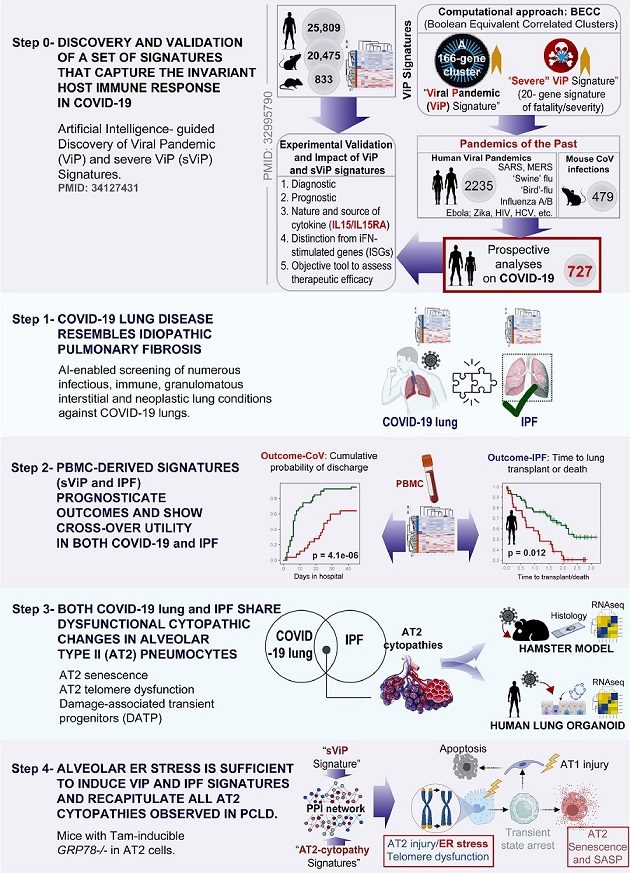 Study design: Artificial Intelligence-guided navigation of COVID-19 lung disease. (From top to bottom) Step 0: Over 45,000 human, mouse, and rat gene expression databases were mined using machine learning tools called Boolean Equivalent Correlated Clusters (BECC133) to identify invariant host response to viral pandemics (ViP). In the absence of a sufficiently large number of COVID-19 datasets at the onset of the COVID-19 pandemic, these ViP signatures were trained on only two datasets from the pandemics of the past (Influenza and avian flu; GSE47963, n = 438; GSE113211, n = 118) and used without further training to prospectively analyze the samples from the current pandemic (i.e., COVID-19; n = 727 samples from diverse datasets). A subset of 20-genes classified disease severity called severe-ViP (sViP) signature. The ViP signatures appeared to capture the ‘invariant’ host response, i.e., the shared fundamental nature of the host immune response induced by all viral pandemics, including COVID-19. Step1: The set of ViP/sViP signatures and a CoV-lung specific13 gene signature was analyzed on diverse transcriptomic datasets representing a plethora of lung diseases; these efforts identified COVID-19 lung disease to be the closest to Idiopathic pulmonary fibrosis (IPF); both conditions induced a common array of gene signatures. Step 2: Clinically useful whole-blood and PBMC-derived prognostic signatures previously validated in IPF27 showed crossover efficacy in COVID-19, and vice versa. Step 3: Gene signatures of alveolar type II (AT2) cytopathic changes that are known to fuel IPF were analyzed in COVID-19 lung, and predicted shared features were validated in human and hamster lungs and lung-organoid derived models. Step 4: Protein- protein interaction (PPI) network built using sViP and AT2 cytopathy-related signatures was analyzed to pinpoint ER stress as a major shared feature in COVID-19 lung disease and IPF, which was subsequently validated in human and hamster lungs.
Study design: Artificial Intelligence-guided navigation of COVID-19 lung disease. (From top to bottom) Step 0: Over 45,000 human, mouse, and rat gene expression databases were mined using machine learning tools called Boolean Equivalent Correlated Clusters (BECC133) to identify invariant host response to viral pandemics (ViP). In the absence of a sufficiently large number of COVID-19 datasets at the onset of the COVID-19 pandemic, these ViP signatures were trained on only two datasets from the pandemics of the past (Influenza and avian flu; GSE47963, n = 438; GSE113211, n = 118) and used without further training to prospectively analyze the samples from the current pandemic (i.e., COVID-19; n = 727 samples from diverse datasets). A subset of 20-genes classified disease severity called severe-ViP (sViP) signature. The ViP signatures appeared to capture the ‘invariant’ host response, i.e., the shared fundamental nature of the host immune response induced by all viral pandemics, including COVID-19. Step1: The set of ViP/sViP signatures and a CoV-lung specific13 gene signature was analyzed on diverse transcriptomic datasets representing a plethora of lung diseases; these efforts identified COVID-19 lung disease to be the closest to Idiopathic pulmonary fibrosis (IPF); both conditions induced a common array of gene signatures. Step 2: Clinically useful whole-blood and PBMC-derived prognostic signatures previously validated in IPF27 showed crossover efficacy in COVID-19, and vice versa. Step 3: Gene signatures of alveolar type II (AT2) cytopathic changes that are known to fuel IPF were analyzed in COVID-19 lung, and predicted shared features were validated in human and hamster lungs and lung-organoid derived models. Step 4: Protein- protein interaction (PPI) network built using sViP and AT2 cytopathy-related signatures was analyzed to pinpoint ER stress as a major shared feature in COVID-19 lung disease and IPF, which was subsequently validated in human and hamster lungs.
He added, "At a fundamental level, both conditions display similar gene expression patterns in the lungs and blood, and dysfunctional processes within alveolar type II (AT2) cells."
Alveolar type II (AT2) cells play several critical roles in pulmonary function, including the production of lung surfactant that keeps lung cells from collapsing after exhalation and regeneration of lung cells after injury.
Professor Ghosh added, "The study findings are insightful because AT2 cells are known to contain an elegant quality control network that responds to stress, internal or external. Failure of quality control leads to broader organ dysfunction and, in this case, fibrotic remodeling of the lung."
In order to conduct the study, Professor Ghosh collaborated with co-senior author Debashis Sahoo, Ph.D., an Associate Professor in the departments of Computer Science, Engineering and Pediatrics at UC San Diego to access transdisciplinary approaches, such as AI-assisted ‘big data’ analysis.
Professor Ghosh and Asst Professor Sahoo said the strategy would help them stay unbiased in navigating the unknowns of an emerging, post-pandemic disease.
The combined study team analyzed more than 1,000 human lung transcriptomic datasets associated with various lung conditions, specifically looking for gene expression patterns, inflammation signaling and cellular changes. The disease with the closest match: IPF.
The study team were able to successfully induce these tell-tale elements in human lung organoids, in a hamster model of COVID-19, and could confirm their presence in the lungs of deceased individuals with COVID-19. Key elements were also reversed in the hamsters using anti-SARS-COV-2 therapeutics. A deeper analysis pinpointed endoplasmic reticulum stress as the shared early trigger of both post-COVID lung disease and ILD.
Professor Ghosh added that the use of computational models to identify shared gene expression and cellular processes between COVID-19 and IPF suggests utility of the findings beyond the current pandemic.
He said, "The insights, biomarkers, tools, mechanisms and promising therapeutic avenues identified here are likely to spur the development of therapies for patients with IPF and other fibrotic interstitial lung diseases, all of whom have limited or no treatment options."
Idiopathic Pulmonary Fibrosis current affects approximately 100,000 persons in the United States, with 30,000 to 40,000 new cases annually. The condition has a poor prognosis, with an estimated mean survival of 2 to 5 years from time of diagnosis. In the United Kingdom, about 34,000 people are affected with the condition with about 7,000 new cases per year.
With the ongoing COVID-19 surges and the fact that almost 91.7 million people in the United States and 23.2 million people in the United Kingdom have already been exposed to the SARS-CoV-2 virus according to official figures (In reality the actual numbers can be as high as between 1.5 to 2.2-fold!), we can expect to see a rise in IPF in both these countries and elsewhere globally and more people dying from it and contributing to excess death rates.
This is good news for us at Thailand
Medical News as it adds to the list of conditions that will contribute to our hypothesis that a majority of the individuals who have been exposed to the SARS-CoV-2 virus will have shortened lifespans with only about 5 to 8 years left on an optimistic level due to a variety of health conditions that will develop in them.
https://www.thailandmedical.news/news/breaking-hypothesis-that-majority-exposed-to-sars-cov-2-will-have-shortened-lifespans-validated-by-study-showing-nsp2-impairs-human-4ehp-gigyf2-comple
Fortunately, reinfections with newer variants will help shorten that remaining time frame. With so many and ignorant stupid people especially those who think that they are far more smarter and keep on claiming that the new Omicron variants are mild or that COVID-19 is nothing but like common flu!, it will be nice to see such excess deaths rates occurring in coming years while ridding the world of such stupid garbage!
For more on
Post COVID Idiopathic Pulmonary Fibrosis, keep on logging to Thailand
Medical News.

