BREAKING! Saudi Arabia Study Uncovers That Mutations In Nucleocapsid Protein Of SARS-CoV-2 Affects Viral Load, Host Response And Disease Severity
Source: Medical News - Mutations In Nucleocapsid Proteins Mar 28, 2022 4 years, 1 month, 5 days, 7 hours, 21 minutes ago
A new study led by researchers from King Abdullah University of Science and Technology (KAUST)-Saudia Arabia has found that mutations in nucleocapsid protein of SARS-CoV-2 affects viral load, host response and disease severity.
The study team sequenced 892 SARS-CoV-2 genomes collected from patients in Saudi Arabia from March to August 2020.
The team found that two consecutive mutations (R203K/G204R) in the nucleocapsid (N) protein were associated with higher viral loads in COVID-19 patients.
Further detailed comparative biochemical analysis revealed that the mutant N protein displays enhanced viral RNA binding and differential interaction with key host proteins.
The study team found increased interaction of GSK3A kinase simultaneously with hyper-phosphorylation of the adjacent serine site (S206) in the mutant N protein.
Also, the host cell transcriptome analysis suggests that the mutant N protein produces dysregulated interferon response genes.
The study findings provide crucial information in linking the R203K/G204R mutations in the N protein to modulations of host-virus interactions and underline the potential of the nucleocapsid protein as a drug target during infection.
The study findings were published in the peer reviewed journal: Nature Communications.
https://www.nature.com/articles/s41467-022-28287-8
The multicenter collaboration that also involved other researchers from King Faisal Specialist Hospital and Research Center, Al Noor Specialist Hospital Makkah, Dr Suliman Al-Habib Medical Group, King Abdulaziz University, General Directorate of Health Affairs Makkah Region, Taibah University, Ministry of National Guard - Health Affairs along with support by scientists from Imperial College-UK, tracked the spread and evolution of the SARS-CoV-2 virus in Saudi Arabia and also identified mutations in the virus's N protein associated with increased viral loads in COVID-19 patients.
The study findings provide insight into the function of this nucleocapsid protein, which could help develop drugs that reduce the impact of coronavirus infection.
KAUST research scientist Muhammad Shuaib told Thailand
Medical News , "The nucleocapsid (N) protein is the most abundant protein in all coronaviruses, including SARS-CoV-2."
He added, “This protein binds to various parts of the viral RNA, affecting how it is packaged within the virus. It also plays roles inside host cells related to viral replication and host immune responses.”
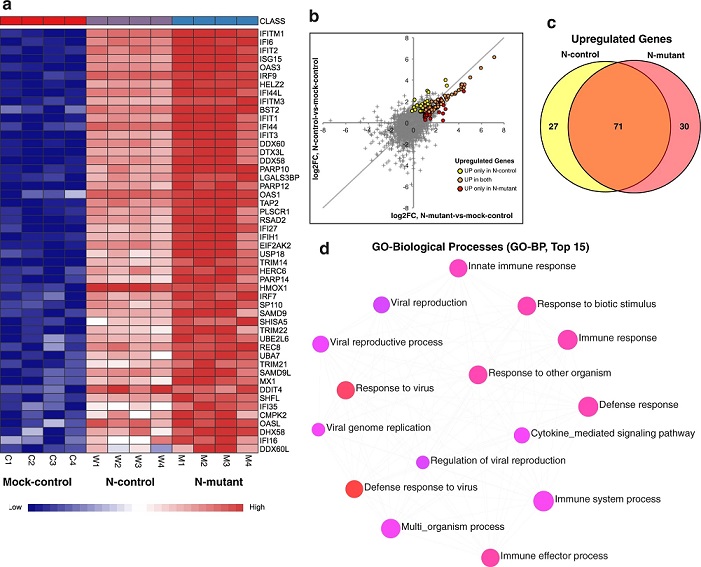 Calu-3 cells were transfected with plasmids expressing the full-length N-control and N-mutant protein along with mock control (4 biological replicates per condition). 48-h post-transfection total RNA was isolated and subjected to RNA-sequencing using illumina NovaSeq 6000 platf
orm. a Heatmap shows normalized expression of top significantly differentially expressed genes in N-mutant and N-control conditions (adj p-value <0.05 and log2 fold-change cutoff ≥1). Genes enriched in interferon and immune-related processes are overexpressed in the N-mutant transfected cells. The heatmap was generated by the visualization module in the NetworkAnalyst. b Plot showing comparison of fold-changes for all DE genes in N-mutant and N-control conditions. Differentially expressed genes display higher up-regulation in the N-mutant condition (as orange dots that represent common up-regulated genes are skewed towards the lower half of the diagonal). c Venn diagram shows the common and unique up-regulated genes in both conditions. d GO-enrichment analysis (top 15 pathways based on p-value and FDR are shown) of up-regulated genes. The enriched GO BP (Biological Processes) term is related to defense and interferon response. The enriched terms display an interconnected network with overlapping gene sets. Each node represents an enriched term and colored by its p-value from red to blue in ascending order (red shows the smallest p-value). The size of each node corresponds to number of linked genes from the list.
Calu-3 cells were transfected with plasmids expressing the full-length N-control and N-mutant protein along with mock control (4 biological replicates per condition). 48-h post-transfection total RNA was isolated and subjected to RNA-sequencing using illumina NovaSeq 6000 platf
orm. a Heatmap shows normalized expression of top significantly differentially expressed genes in N-mutant and N-control conditions (adj p-value <0.05 and log2 fold-change cutoff ≥1). Genes enriched in interferon and immune-related processes are overexpressed in the N-mutant transfected cells. The heatmap was generated by the visualization module in the NetworkAnalyst. b Plot showing comparison of fold-changes for all DE genes in N-mutant and N-control conditions. Differentially expressed genes display higher up-regulation in the N-mutant condition (as orange dots that represent common up-regulated genes are skewed towards the lower half of the diagonal). c Venn diagram shows the common and unique up-regulated genes in both conditions. d GO-enrichment analysis (top 15 pathways based on p-value and FDR are shown) of up-regulated genes. The enriched GO BP (Biological Processes) term is related to defense and interferon response. The enriched terms display an interconnected network with overlapping gene sets. Each node represents an enriched term and colored by its p-value from red to blue in ascending order (red shows the smallest p-value). The size of each node corresponds to number of linked genes from the list.
The study team found that two consecutive mutations in the N protein, called R203K and G204R, were associated with increased severity of COVID-19 in patients. Analyses showed that the changes to the protein made it bind more strongly to the viral RNA.
Detailed analysis in laboratory cells suggested that the changes in the N protein allowed the virus to more efficiently hijack host cell translation machinery to facilitate virus replication.
These mutations were also associated with increased expression of genes involved in interferon and chemokine production. This could be behind the life-threatening cytokine storm that happens in some COVID-19 patients, making it very difficult for them to breathe.
The study findings were the result of analyses of viral genome sequences from 892 patient samples taken from various parts of Saudi Arabia between March and August 2020, relatively early in the pandemic. This was followed by comparisons with patient data to understand how mutations affected viral load and virulence.
Dr Tobias Mourier, a consultant research scientist working on the research added, "Compared to the spike protein, the N protein is highly conserved in the different coronaviruses, like SARS and MERS; yet attempts to design vaccines against it have not been successful. Understanding N protein's function could help develop drugs that target it and potentially limit disease severity in COVID-19 and other coronavirus infections."
The study team warned, "Sequencing virus genomes and reporting genomic changes from regions of the world that are severely underrepresented in current databases is essential for tracking and assessing new variants of concern.”
They added, “In conclusion, our results suggest a major influence of the R203K/G204R mutations on the essential properties and phosphorylation status of SARS-CoV-2 N protein that may lead to increased host response and heightened efficacy of viral infection.”
For more about
SARS-CoV-2 Nucleocapsid Proteins, keep on logging to Thailand Medical News.

 Calu-3 cells were transfected with plasmids expressing the full-length N-control and N-mutant protein along with mock control (4 biological replicates per condition). 48-h post-transfection total RNA was isolated and subjected to RNA-sequencing using illumina NovaSeq 6000 platf
orm. a Heatmap shows normalized expression of top significantly differentially expressed genes in N-mutant and N-control conditions (adj p-value <0.05 and log2 fold-change cutoff ≥1). Genes enriched in interferon and immune-related processes are overexpressed in the N-mutant transfected cells. The heatmap was generated by the visualization module in the NetworkAnalyst. b Plot showing comparison of fold-changes for all DE genes in N-mutant and N-control conditions. Differentially expressed genes display higher up-regulation in the N-mutant condition (as orange dots that represent common up-regulated genes are skewed towards the lower half of the diagonal). c Venn diagram shows the common and unique up-regulated genes in both conditions. d GO-enrichment analysis (top 15 pathways based on p-value and FDR are shown) of up-regulated genes. The enriched GO BP (Biological Processes) term is related to defense and interferon response. The enriched terms display an interconnected network with overlapping gene sets. Each node represents an enriched term and colored by its p-value from red to blue in ascending order (red shows the smallest p-value). The size of each node corresponds to number of linked genes from the list.
Calu-3 cells were transfected with plasmids expressing the full-length N-control and N-mutant protein along with mock control (4 biological replicates per condition). 48-h post-transfection total RNA was isolated and subjected to RNA-sequencing using illumina NovaSeq 6000 platf
orm. a Heatmap shows normalized expression of top significantly differentially expressed genes in N-mutant and N-control conditions (adj p-value <0.05 and log2 fold-change cutoff ≥1). Genes enriched in interferon and immune-related processes are overexpressed in the N-mutant transfected cells. The heatmap was generated by the visualization module in the NetworkAnalyst. b Plot showing comparison of fold-changes for all DE genes in N-mutant and N-control conditions. Differentially expressed genes display higher up-regulation in the N-mutant condition (as orange dots that represent common up-regulated genes are skewed towards the lower half of the diagonal). c Venn diagram shows the common and unique up-regulated genes in both conditions. d GO-enrichment analysis (top 15 pathways based on p-value and FDR are shown) of up-regulated genes. The enriched GO BP (Biological Processes) term is related to defense and interferon response. The enriched terms display an interconnected network with overlapping gene sets. Each node represents an enriched term and colored by its p-value from red to blue in ascending order (red shows the smallest p-value). The size of each node corresponds to number of linked genes from the list.