Breaking! SARS-CoV-2 Dysregulates One-Carbon Metabolism In Human Host With Implications For Various Long Term Diseases! N-Acetyl Cysteine Helps!
Source: Medical News - NAC Apr 14, 2022 4 years, 1 month, 1 week, 5 days, 21 hours, 28 minutes ago
Yet another worrisome study finding has emerged that adds to the growing list of human host cellular and biological pathways… now numbering in hundreds, that are dysregulated or disrupted by the SARS-CoV-2 coronavirus with implications for long term medical and health issues, some that can end up with fatal outcomes.

Already Thailand
Medical News has issued numerous warnings that majority who have been exposed to the viral proteins of the SARS-CoV-2 coronavirus either by natural infections or via therapeutics, will have reduced life spans with many only having about 5 to 8 years left. Those number of years will be shortened with reinfections and coinfections from the various emerging SARS-CoV-2 variants and subvariants. Many of these expected fatal outcomes include heart failures, strokes and other thrombotic events, CVST, organ failure especially of kidneys, lungs and liver, gastrointestinal issues that is accompanied by sepsis, secondary opportunistic infections, accelerated and aggressive cancers, accelerated and aggressive neurodegenerative issues etc. Most of these subsequent fatal outcomes in otherwise seemingly healthy individuals will be deemed as non-COVID-19 deaths as planned by those controlling the COVID-19 narratives!
Initial conditions upon infection by the SARS-CoV-2 virus or its variants or subvariants are not the only manner to categorize as to whether the variant is mild or more lethal, rather studies should be conducted as to how the human host is affected in also the mid-term and long term especially in terms of the various cellular and biological pathways. With more of the emerging variants displaying the capability to evade both vaccine-induced immunity and natural immunity, the issues of viral persistence is going to be a new worrying norm!
In this new study by researchers from the the University of Life Sciences, Poznań-Poland and Rutgers-New Jersey Medical School, New Jersey-USA, it was found that the SARS-CoV-2 coronavirus is able to dysregulated One-Carbon Metabolism in the human host!
One-carbon (1C) metabolism comprises a series of interlinking metabolic pathways that include the methionine and folate cycles that are central to cellular function, providing 1C units (methyl groups) for the synthesis of DNA, polyamines, amino acids, creatine, and phospholipids. S-adenosylmethionine is a potent aminopropyl and methyl donor within these cycles and serves as the principal substrate for methylation of DNA, associated proteins, and RNA.
Dysregulation of one-carbon metabolism affects a wide range of biological processes and is associated with a number of diseases, including cardiovascular disease, dementia, neural tube defects, and cancer.
To date, accumulating evidence suggests that one-carbon metabolism plays an important role in COVID-19. The symptoms of long COVID-19 are similar to those presented by subjects suffering from vitamin B12 deficiency (pernicious anemia).
The metabolism of a cell infected by the SARS-CoV-2 virus is reshaped to fulfill the need for massive viral RNA synthesis, which requires de novo purine biosynthesis involving folate and one-carbon metabolism. Many aspects of host sulfur amino acid metabolism, particularly glutathione metabolism underlying antioxidant defenses, are also taken over by the SARS-CoV-2 virus.
This review study f
indings summarize recent findings related to one-carbon metabolism and sulfur metabolites in COVID-19 and discuss how they inform strategies to combat the disease.
The review findings were published in the peer reviewed International Journal of Molecular Sciences.
https://www.mdpi.com/1422-0067/23/8/4181
The SARS-CoV-2 coronavirus is the pathogen responsible for the ongoing COVID-19 pandemic. The SARS-CoV-2 infection has a wide range of symptoms, ranging from none to extremely critical symptoms.
To date, the pathophysiology of COVID-19 is still not well understood, even after more than two years; however, many scientists consider that critical and severe COVID-19 is related to the severity of the immune-inflammatory response.
This new study addresses the relationship between SARS-CoV-2 to the metabolism of one-carbon molecules in the host, suggesting new therapeutic pathways.
Already many earlier protein-based research in COVID-19 patients showed that several pathways were dysregulated, some of which include the complement and coagulation cascades, interactions between different cytokines, and cholesterol metabolism. Even as biomarkers were identified that indicated increased severity and mortality from COVID-19, these were found to be largely involved in inflammatory pathways.
Besides acute COVID-19, “long COVID” or PASC (Post-Acute Sequelae of SARS-CoV-2 infection) has become a recognized syndrome, though it is far from being clearly defined.
The many symptoms of long COVID may include neurological sequelae, chest pain, fatigue, dyspnea, respiratory symptoms, joint pain, and insomnia. These symptoms resemble those of pernicious anemia, which is a condition caused by the deficiency of vitamin B12 or cobalamine.
Importantly, the underlying mechanism here is a one-carbon pathway of methylation, as B12 is a cofactor of vitamin B12-dependent methionine (Met) synthase (MS), which is a key enzyme in this pathway. MS is the source of methionine in the body and is required for the production of the universal methyl donor S-adenosylmethionine (SAM).
It is already known that the one-carbon pathway is involved in many biological processes including purine and thymidine synthesis, nucleic acid synthesis, homeostasis of glycine, serine, and methionine, as well as the generation of glutathione through the homocysteine-cysteine pathway, wherein glutathione is an essential molecule for antioxidant activity in physiological processes.
Just as significant, this pathway is also involved in the production of energy through adenosine triphosphate (ATP) generation within the mitochondria of the cell.
The study team found that during the hijacking of host cell metabolism by SARS-CoV-2, transcription continues to occur for host metabolic enzymes but shows an overall reduction. ATP appears to be depleted, as shown by the expression of mitochondrial DNA.
SARS-CoV-2 Dysregulation Of One-Carbon Pathways
The study findings showed that purine biosynthesis de novo appears to increase, with rising levels of intermediates and a reduction in intracellular folate. Notably, serine donates one-carbon units for this process, taking part in folate metabolism. Many other metabolites in the cell that participate in either sulfur-containing amino acid pathways or one-carbon metabolism also showed a reduction following SARS-CoV-2 infection.
Effects On Methionine
It was found that within the infected cell, methionine and related sulfur amino acids showed a reduction. Conversely, such cells did not show any reductions in SAM, cysteine, and oxidized glutathione, all of which participate in one-carbon metabolism. The implication is that SARS-CoV-2 takes over folate and one-carbon pathways for its intracellular replication.
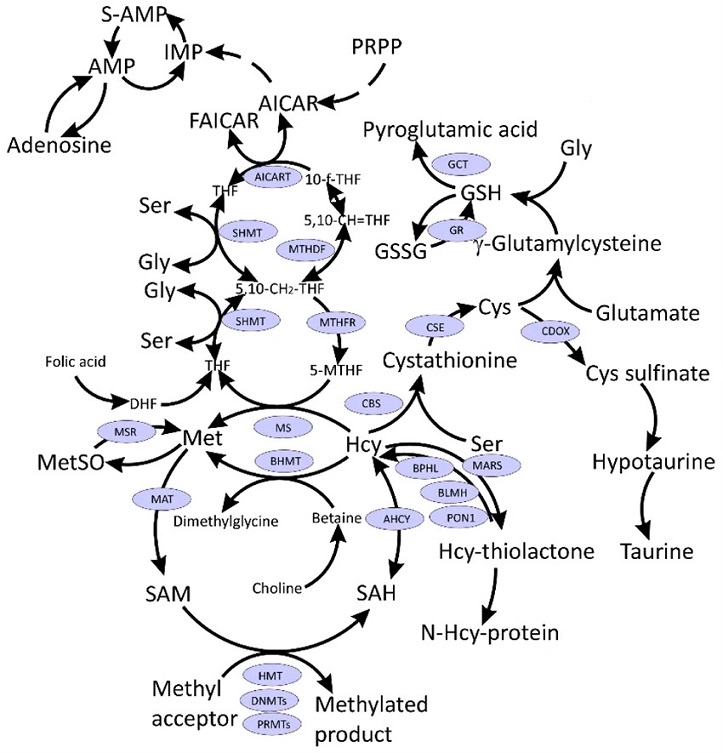 Interconnections between metabolism of folate, one-carbon, and sulfur compounds. Indicated metabolites are discussed in the text. CysGly, a product of GSH catabolism, affected in COVID-19 and discussed in the text, is not shown.
Interconnections between metabolism of folate, one-carbon, and sulfur compounds. Indicated metabolites are discussed in the text. CysGly, a product of GSH catabolism, affected in COVID-19 and discussed in the text, is not shown.
The study findings showed that a rise in SAM levels, as well as a rise in SAM/S-adenosyl homocysteine (SAH), suggests lung damage has occurred, while the former is highest in critically ill patients but is linked to a better outcome.
It was noted that the level of dimethylglycine, which is produced from homocysteine during its conversion of methionine, did not show any association with the product concerned.
Certain past studies have showed that methionine increases, whereas others have reported its reduction; however, an increase in methionine levels is more common in patients with critical COVID-19, whereas its reduction is more often associated with mild illness. The reduction in methionine sulfoxide levels, which is seen consistently in COVID-19, indicates an increase in oxidant stress following infection.
Glutathione Levels
It was also found that glutathione levels are also affected in COVID-19, with low levels indicating oxidative stress and lung damage. This is the most important antioxidant in human physiology and is depleted in high-risk conditions for COVID-19. The level of SAM is also associated with a higher SAM to GSH ratio and higher homocysteine levels.
Interestingly, in certain past studies, it was found that cysteine was increased in serum, as Cys-Gly produced by the metabolism of GSH is lower with lung damage. Meanwhile, glycine, being a part of GSH biosynthesis, showed varying directions of change in these patients, as with cysteine.
Homocysteine Levels
The study findings also showed that homocysteine is elevated in COVID-19; however, there remains limited information regarding whether this effect is due to older age and male sex in COVID-19 cases as compared to controls remains unanswered. The MTHFR 677T gene is more common in certain ethnic groups that have a higher-than-expected incidence of COVID-19 and related mortality.
Importantly, homocysteine may trigger thrombosis or coagulopathy, both of which are complications common in COVID-19.
Certain scientists have suggested that genotyping COVID-19 for certain single nucleotide polymorphisms would help identify those with the highest risk of complications from clotting; however, this is currently not supported by experimental data.
Methylmalonic Acid(MMA) And Choline
Methylmalonic acid (MMA), which fell to 3% of the baseline level, is a catabolic product formed during the breakdown of certain amino acids and could play an antiviral and anti-inflammatory role. The reduction of MMA in adults fits thus supports a potential mechanism that causes increased severity of COVID-19 in adults.
The study findings also found that in adults but not COVID-19 positive children, choline and its metabolites were found at lower levels.
Renin-Angiotensin System (RAS)
It is already known that the renin-angiotensin system (RAS) is key to the regulation of cardiovascular function and renal health. It may also cause hypertension, diabetes, and obesity, all of which are risk factors for COVID-19.
The angiotensin-converting enzyme (ACE) and RAS balance each other to prevent undesirable effects on the cardiovascular system. Moreover, high ACE2 levels at admission have been used to predict a higher risk of severe illness.
Importantly the one-carbon cycles linked to homocysteine act through the RAS system as well, hence any dysregulation could affect cardiovascular health.
The Folate Cycle
It has been found that the folate cycle is key to one-carbon metabolism. Therapeutics and drugs that inhibit dihydrofolate reductase, as well as other steps in one-carbon transfer, have been found to block viral replication.
Potential Therapeutics
Interestingly, folic acid could also be a furin inhibitor, thus preventing SARS-CoV-2 infection as furin activity is required for the proteolytic cleavage of the viral spike protein that precedes lung cell entry by the virus.
Numerous treatment strategies suggested for COVID-19 are currently being examined.
Interestingly, screening pathways indicate the potential utility of folic acid as an inhibitor of spike-ACE2 receptor binding. Indeed, this was the leading nutraceutical with this predicted activity.
Also, 5-methyltetrahydrofolate was found to be a ligand of the key viral enzyme PLpro, while folic acid derivatives bound to the NSP15 protein. In all these cases, binding energies were comparable or superior to currently known drugs that inhibit these pathways.
Antifolate drugs such as methotrexate have been found to have an antiviral effect by blocking purine biosynthesis. This drug may act in synergy with other existing antivirals to block viral replication and the secretion of infectious virions.
Conclusions
The findings of many studies regarding the relationship between one-carbon metabolism and SARS-CoV-2 replication within human host cells are often conflicting.
Importantly however, these studies have demonstrated the potential role of glutathione, methionine sulfoxide, and choline in this process. These differences could be due to the confounding effects of non-matched age, sex, or ethnicity between cases and controls in various studies.
Yet another possible reason for inconsistent findings could be the difference in COVID-19 classification systems as to the severity of illness and differences in the time points at which samples were taken. For example, one review found that SAM levels could differ in cases as compared to controls simply by manipulating the study selected by the classification system used.
The study team proposes that among potential therapeutic approaches, the three aforementioned metabolites that are potentially impacted by COVID-19 may be worthy of further study. Thus, measures to normalize their levels may mitigate the severity of COVID-19 and improve patient outcomes. This includes N-acetyl cysteine to increase glutathione levels, preferably with glycine as well.
Such a resulting antioxidant boost may help restore methionine sulfoxide levels to normal as well. More work will be needed to establish the therapeutic utility of these strategies.
For more on the
Latest SARS-CoV-2 Research, keep on logging to Thailand
Medical News.
Read Also:
https://www.thailandmedical.news/news/breaking-covid-19-research-russian-study-indicates-that-glutathione-deficiency-affects-covid-19-susceptibility,-nac-supplements-helps
https://www.thailandmedical.news/news/breaking-study-shows-that-sars-cov-2-infections-lead-to-increased-levels-of-oxidative-stress,-oxidant-damage-and-glutathione-deficiency

 Interconnections between metabolism of folate, one-carbon, and sulfur compounds. Indicated metabolites are discussed in the text. CysGly, a product of GSH catabolism, affected in COVID-19 and discussed in the text, is not shown.
Interconnections between metabolism of folate, one-carbon, and sulfur compounds. Indicated metabolites are discussed in the text. CysGly, a product of GSH catabolism, affected in COVID-19 and discussed in the text, is not shown.