BREAKING! COVID-19 News: SARS-CoV-2 Causes Macular Degeneration By Promoting RPE Cell Senescence Via The ROS/P53/P21 Pathway!
COVID-19 News - SARS-CoV-2 Causes Macular Degeneration Feb 06, 2023 3 years, 2 months, 3 weeks, 6 days, 9 hours, 7 minutes ago
COVID-19 News: More depressing news emerging about the effects of SARS-CoV-2 infection as a new study conducted by Chinese researchers have found the spike protein of the SARS-CoV-2 virus promotes retinal pigment epithelium (RPE) cell senescence via the ROS/P53/P21 pathway which can lead to eye macular degeneration! Macular degeneration leads to the losing of one’s central vision.
The study team comprising of medical scientists and ophthalmologists from Zhengzhou University-China, Fudan University, Shanghai-China and Henan University-China investigated the role of the SARS-COV-2 spike protein (S-protein) in regulating the senescence of RPE cells.
The study findings showed that administration or overexpression of S-protein in ARPE-19 decreased cell proliferation with cell cycle arrest at the G1 phase.
The spike proteins increased SA-β-Gal positive ARPE-19 cells with high expression of P53 and P21, senescence-associated inflammatory factors (e.g., IL-1β, IL-6, IL-8, ICAM, and VEGF), and ROS. Elimination of ROS by N-acetyl cysteine (NAC) or knocking down p21 by siRNA diminished S-protein-induced ARPE cell senescence.
It was found that both administrated and overexpressed S-protein colocalize with the ER and upregulate ER-stress-associated BIP, CHOP, ATF3, and ATF6 expression. S-protein induced P65 protein nuclear translocation. Inhibition of NF-κB by bay-11-7082 reduced S-protein-mediated expression of senescence-associated factors.
Additionally, it was shown that the intravitreal injection of S-protein upregulates senescence-associated inflammatory factors in the zebrafish retina.
The study Findings clearly showed that the S-protein of SARS-Cov-2 induces cellular senescence of ARPE-19 cells in vitro and the expression of senescence-associated cytokines in zebrafish retina in vivo likely by activating ER stress, ROS, and NF-κb.
The study findings may uncover a potential association between SARS-cov-2 infection and development of age-related macular degeneration or AMD.
The study findings were published in the peer reviewed journal: Biogerontology (Springer)
https://link.springer.com/article/10.1007/s10522-023-10019-0
Thailand Medical News in our previous
COVID-19 News coverages had already warned that COVID-19 can cause a number of eye issues.
https://www.thailandmedical.news/news/french-study-reveals-that-covid-19-causes-potentially-dangerous-eye-abnormalities-such-as-the-formation-of-nodules-in-the-macular-region
https://www.thailandmedical.news/news/covid-19-news-italian-researchers-find-that-sars-cov-2-can-affect-the-retina-in-the-eyes
;
https://www.thailandmedical.news/news/covid-19-and-eyes-doctors-report-case-of-retinal-vein-occlusion-due-to-covid-19-in-patient-more-cases-of-eyes-being-attacked-by-sars-cov-2-emerging
https://www.thailandmedical.news/news/covid-19-latest-warnings-of-more-cases-of-covid-19-related-eye-infections-emerging
https://www.thailandmedical.news/news/covid-19-symptoms-new-documented-case-shows-that-sars-cov-2-can-cause-cranial-nerve-palsy-with-resulting-double-eye-vision-diplopia
https://www.thailandmedical.news/news/breaking-german-study-shows-sars-cov-2-can-infect-and-replicate-in-retinal-cells-with-implications-of-eye-issues-for-long-covid
https://www.thailandmedical.news/news/spanish-study-finds-that-sars-cov-2-coronavirus-present-in-tears-and-ocular-secretions-of-individuals-with-covid-19
https://www.thailandmedical.news/news/californian-study-shows-that-post-covid-individuals-are-at-a-risk-of-developing-retinal-vascular-occlusions-or-eye-strokes
https://www.thailandmedical.news/news/us-nih-discovers-new-eye-macular-dystrophy-disease-caused-by-new-timp3-variants--interestingly,-sars-cov-2-is-known-to-affect-timp3-transcription
https://www.thailandmedical.news/news/covid-19-news-study-validates-that-sars-cov-2-exhibits-ocular-tropism-and-is-able-to-cause-vision-issues
https://www.thailandmedical.news/news/covid-19-news-various-new-case-reports-show-that-sar-cov-2-is-causing-lots-of-new-eye-and-vision-issues-especially-in-post-covid-phases
AMD or
age-related macular degeneration affects approximately 0.4–8.7% of individuals over the age of 50 and is a leading cause of blindness among the elderly in the developed world.
Most of the affected individuals (about 80–85%) suffer from dry AMD, characterized by atrophy of photoreceptor cells, ganglia cells, and retinal pigment epithelial cells (RPE).
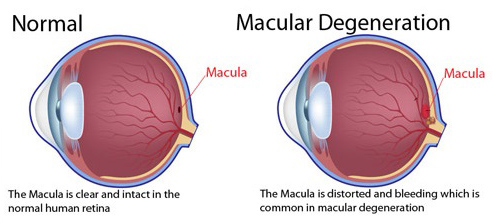
It has already been validated that an increase in the number of senescent cells in the RPE is associated with early onset AMD. This can be triggered by multiple risk factors such as UV and blue light radiation, hyperglycemia, retina ischemia, and also viral infections!
https://www.mdpi.com/1422-0067/17/9/1408
https://www.sciencedirect.com/science/article/pii/S2213231720308284
https://www.mdpi.com/1422-0067/22/3/1338
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, which attacks multiple tissues including the eye fundus, can trigger cellular senescence in infected cells. This implies that SARS-CoV-2 may be associated with retinopathy.
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)31014-X/fulltext
https://www.frontiersin.org/articles/10.3389/fphar.2020.584637/full
https://journals.asm.org/doi/full/10.1128/JVI.00794-21
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8507266/
SARS-CoV-2 virion and its S-protein promote cellular senescence by partially upregulating interferon-γ and toll-like receptor.
The study findings indicate that the S-protein of SARS-CoV-2 may be a virulent factor in triggering retinopathy.
The study team found that both administration of purified S-protein and ectopic expression of Flag-S-protein induce ARPE-19 cell senescence.
This is consistent with previous report demonstrating that the Spike protein can cause senescence in lung tumor cells in vitro.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8507266/
The senescence induction by the Spike-protein is associated with upregulation of ROS production. Removing ROS by NAC reduces S-protein-induced cellular senescence and secretion of cytokines IL-1 and IL-6.
SARS-Cov-2 has been shown to increase ROS by decreasing the glutathione (GSH) and increasing GSSG in infected cells. It accomplishes this by increasing GSH efflux and/or decreasing Cysteine uptake.
https://www.sciencedirect.com/science/article/pii/S2213231721002007
The current study findings suggest that the spike protein is one of the pathogenic factors of SARS-CoV-2 that increases ROS.
It has also been found that activation ER stress is also involved in SARS-Cov-2-induced pathological change. SARS-Cov-2 virus activates the PERK/IRE pathway via S-protein interacting with ER.
https://journals.asm.org/doi/full/10.1128/JVI.01033-07
The study team found both ectopic expression and administration of S-protein colocalized with the ER and activated ER stress by possibly activating ATF6. S protein contains ER retention peptide, and its ER localization is regulated by viral membrane proteins E and M.
The molecular mechanism underlying the traffic of administrated S-protein to ER however remain unclear. Whether its receptor proteins such as laminin, BIP are involved in the traffic of extracellular S-protein to ER remain unclear.
The study team also explored the role of S-protein in retina by artificially administering S-protein into zebrafish vitreous humor, and found that S-protein can induce the mRNA expression of senescence-associated genes without impairing retina structure.
They however did not observe DNA damage in S-protein treated zebrafish retina. This suggests the administrated S-protein can trigger the senescent phenotype, but which cell type in retina undergo senescence still remain unclear. In this zebrafish model, the study team only administrated S-protein once, and observation time is short. Prolonged observation of the regulatory effects of S-protein on retinopathy is necessary, which is still under investigation.
The study findings on the whole showed that SARS-COV-2 spike protein can induce ARPE-19 cells to undergo senescence by increasing cellular ROS or ER stress in vitro. The SARS-COV-2 spike protein may be associated with development of chronic retinal degeneration, adding yet to another eye condition that SARS-CoV-2 can cause.
For the latest
COVID-19 News, keep on logging to Thailand Medical News.
Read More:
https://www.thailandmedical.news/articles/ophthalmology-(eye-diseases)

