US NIH Discovers New Eye Macular Dystrophy Disease Caused By New TIMP3 Variants. Interestingly, SARS-CoV-2 Is Known To Affect TIMP3 Transcription!
Source: Medical News - Macular Dystrophy Aug 23, 2022 3 years, 9 months, 2 days, 19 hours, 3 minutes ago
Researchers from the U.S. National Eye Institute which is part of the U.S. National Institutes of Health at Maryland-USA have discovered a new type of
macular dystrophy, which is a cause of central vision loss that is caused by a new type of mutation of the human gene TIMP3.
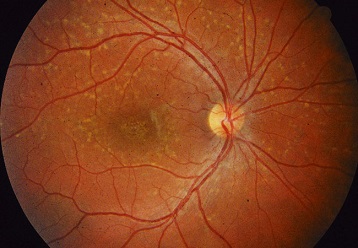
An existing eye disease called Sorsby fundus dystrophy is a typically adult-onset maculopathy with high risk for choroidal neovascularization. Sorsby fundus dystrophy, inherited as an autosomal dominant fully penetrant trait, is associated with TIMP3 variants that cause protein aggregation in the extracellular matrix.
The study was aimed at
evaluating the phenotype and underlying biochemical mechanism of disease-causing TIMP3 variants altering the N-terminal signal peptide in 2 families who have early-onset diffuse maculopathy without choroidal neovascularization with cosegregation of TIMP3 variants in the signal peptide sequence.
The case series study of 2 families with early-onset diffuse maculopathy was conducted at the National Eye Institute, National Institutes of Health Clinical Center. Data were collected and analyzed from October 2009 to December 2021.
Clinical imaging and molecular genetic testing were performed in 2 families with macular dystrophy. Cosegregation analysis of TIMP3 variants was performed in affected and unaffected family members. Candidate TIMP3 signal peptide variants were assessed for cleavage defects after transfection.
The study findings showed that eleven individuals from the 2 families with early-onset diffuse maculopathy without choroidal neovascularization harbor TIMP3 variants (L10H or G12R) in the N-terminal signaling peptide.
Subsequent cosegregation with phenotype was confirmed in additional family members. Biochemical analysis confirmed defects in both protein maturation and extracellular deposition.
The study findings showed that TIMP3 variants altering signal peptide function deviated from classic Sorsby fundus dystrophy both in phenotypic features and underlying mechanism.
The study findings suggest atypical patient presentations are caused by TIMP3 signal peptide defects, associated with impaired cleavage and deposition into the extracellular matrix, implicating a novel macular dystrophy disease.
The study findings were published in the peer reviewed journal: JAMA Ophthalmology.
https://jamanetwork.com/journals/jamaophthalmology/article-abstract/2793168
Thailand
Medical News would like to bring up that incidences of rare forms of macular dystrophy have been increasing in the last 20 months and interestingly the SARS-CoV-2 is able to affect transcription of many human host genes and also cause epigenetic changes to many of these genes including the TIMP3 gene.
/www.ncbi.nlm.nih.gov/pmc/articles/PMC7359430/">https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7359430/
https://link.springer.com/article/10.1007/s00011-021-01487-6
The new eye disease that damages the macula, a small region of the light-sensing retina required for sharp, central vision has yet to be named.
Typically, macular dystrophies are disorders that often result in central vision loss due to abnormalities in various genes, including ABCA4, BEST1, PRPH2, and TIMP3.
Many individuals with Sorsby Fundus Dystrophy, a hereditary eye disorder that is specifically linked with TIMP3 variations, often develop symptoms in adulthood. Due to choroidal neovascularization, which is the growth of new, irregular blood vessels behind the retina that leak fluid and disrupt vision, they often experience abrupt changes in visual acuity.
It is already known that TIMP3 is a protein that helps regulate retinal blood flow and is secreted from the retinal pigment epithelium (RPE), a layer of tissue that nourishes and supports the retina’s light-sensing photoreceptors. All TIMP3 gene mutations reported are in the mature protein after it has been “cut” from RPE cells in a process called cleavage.
Dr Bin Guan, Ph.D. the lead author, told Thailand
Medical News, “We found it surprising that two patients had TIMP3 variants not in the mature protein, but in the short signal sequence the gene uses to ‘cut’ the protein from the cells. We showed these variants prevent cleavage, causing the protein to be stuck in the cell, likely leading to retinal pigment epithelium toxicity.”
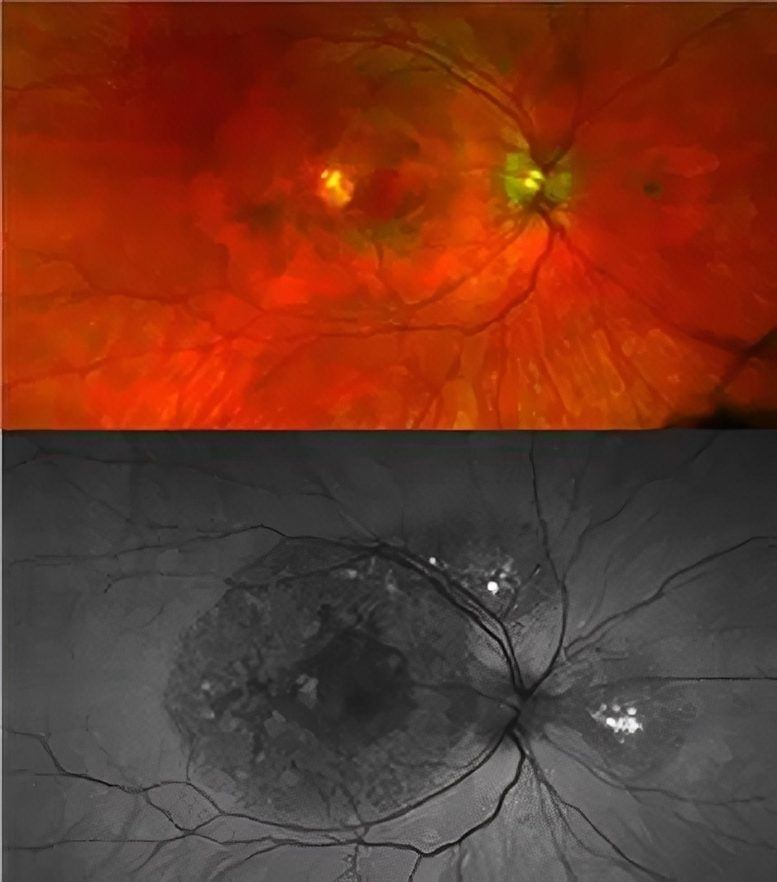 Retinal images of a patient with a TIMP3 mutation causing atypical symptoms. While there is visible damage in the retina (dark circles), there is no choroidal neovascularization present. Credit: National Eye Institute
The study team followed these findings with clinical evaluations and genetic testing of family members to verify that the two new TIMP3 variants are connected to this atypical maculopathy.
Retinal images of a patient with a TIMP3 mutation causing atypical symptoms. While there is visible damage in the retina (dark circles), there is no choroidal neovascularization present. Credit: National Eye Institute
The study team followed these findings with clinical evaluations and genetic testing of family members to verify that the two new TIMP3 variants are connected to this atypical maculopathy.
Dr Cathy Cukras, M.D., Ph.D., a Lasker tenure-track investigator and medical retina specialist who clinically evaluated the patients added, “
Affected individuals had scotomas, or blind spots, and changes in their maculas indicative of disease, but, for now, they have preserved central vision and no choroidal neovascularization, unlike typical Sorsby Fundus Dystrophy.”
The U.S. NIH’s NEI Ophthalmic Genomics Laboratory gathers and manages specimens and diagnostic data from patients who have been recruited into multiple studies within the NEI clinical program to facilitate research of rare eye diseases, including Sorsby Fundus Dystrophy.
Senior author, Dr Rob Hufnagel, M.D., Ph.D., senior author, and director of the Ophthalmic Genomics Laboratory at NEI further added, “Discovering novel disease mechanisms, even in known genes like TIMP3, may help patients that have been looking for the correct diagnosis, and will hopefully lead to new therapies for them.”
Thailand
Medical News would like to add that with the SARS-CoV-2 virus being able to affect many human host cellular pathways, cause dysfunctional regulation of various genes and even epigenetic changes etc, we can expect to see many novel diseases and medical conditions arising.
For more on
macular dystrophy diseases, keep on logging to Thailand
Medical News.
Read also:
https://www.thailandmedical.news/news/breaking-swedish-study-shows-that-sars-cov-2-causes-epigenetic-changes-to-various-genes-in-human-host
https://www.thailandmedical.news/news/breaking-hypothesis-that-majority-exposed-to-sars-cov-2-will-have-shortened-lifespans-validated-by-study-showing-nsp2-impairs-human-4ehp-gigyf2-comple
https://www.thailandmedical.news/news/great-news-study-shows-that-sars-cov-2-protein-nsp13-is-able-to-cause-dna-damage-and-dysregulate-tumor-suppressor-gene-p53,-increasing-cancer-risk
https://www.thailandmedical.news/news/urgent-studies-needed-to-assess-if-sars-cov-2-can-cause-missense-mutations-in-human-dna-and-give-rise-to-new-medical-conditions-never-seen-before
https://www.thailandmedical.news/news/new-study-by-cornell-university-shows-sars-cov-2-causes-epigenetic-changes-to-innate-immune-cells-and-their-progenitors,-contributing-to-long-covid

