Long COVID News: Many With Long COVID Especially Females Will Manifest Aortic and Arterial Stiffness And Also Diastolic Dysfunction
Long COVID News - Aortic and Arterial Stiffness Feb 16, 2023 3 years, 3 months, 1 week, 4 days, 16 hours, 35 minutes ago
Long COVID News: A new study by researchers from University of Medicine and Pharmacy “Victor Babes” Timisoara-Romania, County Emergency Hospital “Pius Brinzeu”- Romania and the Academy of Romanian Scientists have found that many Post COVID and Long COVID individuals are likely to manifest aortic and arterial stiffness and also diastolic dysfunction.
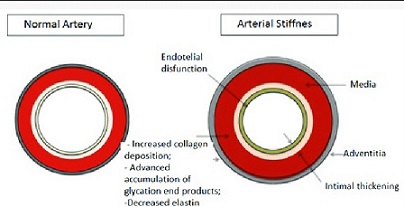
The study findings might also help answer why many are also experiencing either isolated or prolonged diastolic hypertension, a query that Thailand Medical News had covered in one of our past
Long COVID News coverages.
https://www.thailandmedical.news/news/breaking-thailand-medical-news-finds-that-sars-cov-2-infections-increases-risk-of-isolated-diastolic-hypertension-urgent-studies-warranted
According to the Romanian study team, abnormally increased arterial and aortic stiffness (AS and AoS), which are often associated with diastolic dysfunction (DD), represent common alterations in COVID-19.
The study team aimed to assess, by transthoracic echocardiography (TTE) and pulse-wave velocity (PWV), the frequency of these dysfunctions in patients with post-acute COVID-19 syndrome and to highlight potential correlations between their severity and multiple clinical and laboratory parameters.
A total of 121 women were included in our study, all of whom were younger than 55 and had been diagnosed with post-COVID-19 syndrome. Of those women, 67 also had metabolic syndrome (MS) (group A), whereas the other 54 did not (group B); 40 age-matched healthy subjects were used as controls (group C).
The study findings showed that patients in group A had worse values of indexes characterizing AS and AoS and had more frequent DD compared to those from group B and group C (p < 0.0001).
The statistical analysis evidenced significant associations between these indexes and the time that had elapsed since COVID-19 diagnosis, the factors that characterize the severity of the acute disease and those that specify MS.
Multivariate regression analysis identified the following as the main independent predictors for DD: values of the AoS index, the C-reactive protein, and the triglyceride–glucose index.
The study findings concluded that altered AS, AoS, and DD are common in patients with post-COVID-19 syndrome, especially with concurrent MS, and these parameters are apparently associated not only with the severity and time elapsed since COVID-19 diagnosis but also with MS.
The study findings were published in the peer reviewed journal: Biomedicines
https://www.mdpi.com/2227-9059/11/2/492
The pathophysiological pathways through which the SARS-CoV-2 virus affects the human organism have become progressively apparent. Its direct action, by binding to the angiotensin-converting enzyme 2 (ACE2) receptors from the surface of the cellular wall, resulting in their downregulation, has been described since the early stages.
Nevertheless, the immunol
ogically mediated effects from an over-activation of innate and adaptive immunity, which result in an increased release of acute phase reactants, such as pro-inflammatory cytokines, especially interleukine-6 (IL-6), interleukine-1β (IL-1β), ferritin, and C-reactive protein (CRP), together with the activation of macrophages….play a crucial role in the development of multiple systemic injuries.
Also, it is worth mentioning that COVID-19 also precipitates the development of β-cell injury and insulin resistance (IR) through the release of IL-1β and tumor necrosis factor α (TNF-α).
Hence, in individuals already suffering from metabolic dysfunctions (MD), which are known to be associated also with an increased pro-inflammatory state and multiple metabolic alterations, among which IR plays a crucial role, the supplementary inflammatory burden triggered by SARS-CoV-2 infection frequently predisposes patients to the development of multisystem complications.
Besides the deleterious effects on the respiratory system, COVID-19 is often associated with a large spectrum of cardiovascular complications, which may persist during the recovery from this illness, causing multiple cardiac dysfunctions.
Most of these cardiovascular complications are consequences of the direct effects of the virus on the myocytes and endothelial cells (mediated via the ACE2 receptors), but especially, of the inflammation-modulated injuries (cytokine-release syndrome).
https://pubmed.ncbi.nlm.nih.gov/35414837/
Importantly, endothelial dysfunction has been identified as one of the most precocious and frequent alterations during COVID-19, which result from multiple and interconnected pathophysiological pathways.
Numerous studies have discussed that the SARS-CoV-2 virus has an increased inotropism for capillaries and small vessels, resulting in the elevation of arterial stiffness (AS).
https://pubmed.ncbi.nlm.nih.gov/35414837/
https://www.ahajournals.org/doi/full/10.1161/CIRCULATIONAHA.120.047971
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9595447/
Metabolic dysfunction or MD, which is characterized by IR and/or even metabolic syndrome (MS), both of which are frequently encountered in patients recovering from COVID-19, can precipitate arterial stiffness or AS.
https://www.sciencedirect.com/science/article/pii/S2213858721002448
It has been known that the downregulation of ACE2 receptors causes a reduction in arterial vasodilation, followed by increased systemic vascular resistance, AS, and higher blood pressure (BP) values, resulting in an elevation of the afterload. This elevation, in turn, causes the development of left ventricular hypertrophy (LVH) and the occurrence of diastolic dysfunction (DD), as well as elevated pressure in the left atria (LAP) and pulmonary veins, leading to heart failure with preserved ejection fraction (HFpEF).
https://pubmed.ncbi.nlm.nih.gov/35283631/
https://amsad.termedia.pl/Effect-of-COVID-19-on-endothelial-function-evaluated-with-flow-mediated-dilation,150638,0,2.html
https://jamanetwork.com/journals/jamacardiology/article-abstract/2763843
Because AS, which is measured either in the aorta or the peripheral arteries, has emerged as a surrogate marker for vascular aging and atherosclerosis, independently predicting an increased risk for adverse cardiovascular events, one may be tempted to also estimate this risk in individuals suffering from post-acute COVID-19 syndrome.
Furthermore, arterial stiffness or AS can be assessed non-invasively by pulse wave velocity (PWV), which is currently the best-validated diagnostic method, with a high accuracy and reproducibility. It is inversely correlated with vascular compliance and predicts cardiovascular outcomes. On the other hand, several indexes reflecting aortic elasticity, the left ventricular LV mass index (LVMI) defining LVH, and the parameters required to characterize DD and appreciate cardiac performance can be easily ascertained and monitored by transthoracic echocardiography (TTE).
The occurrence of endothelial dysfunction as a consequence of a direct action of the SARS-CoV-2 virus on the ACE2 receptors of endothelial walls, but which is also modulated by precocious and delayed immunological effects, was observed from the early stages of the COVID-19 pandemic and was reported by several researchers.
https://pubmed.ncbi.nlm.nih.gov/35283631/
https://www.mdpi.com/2075-1729/12/6/781
https://www.ingentaconnect.com/content/wk/jhype/2022/00000040/a00101s1/art00449
https://www.tandfonline.com/doi/full/10.1080/07853890.2022.2136403
The impairment in arterial elasticity, which is expressed by an increase in AS and AoSI and a reduction in AoD and AoS, was a predictable consequence. It was concluded that these alterations will induce further cardiac dysfunctions, such as LVH and DD, as well as secondary injuries in other organs.
In the first months of the onset of COVID-19 in Europe, researchers had reported impaired AS levels, as confirmed by elevated values of PWV, in infected people compared to healthy subjects and non-COVID-19 patients and associated this dysfunction with a worse clinical outcome.
https://onlinelibrary.wiley.com/doi/full/10.1111/joim.13275
As a result of accompanying comorbidities, such as systemic hypertension, ischemic heart disease, T2DM, obesity, and MS, which are also associated with accelerated vascular aging, are important contributors to the severity and clinical outcome of an infection with the SARS-CoV-2 virus, it was assumed that they would supplementarily lead to further degeneration of vascular walls, as highlighted in several studies.
https://www.nature.com/articles/s42255-021-00358-y
https://pubmed.ncbi.nlm.nih.gov/35884823/
The SARS-CoV-2 virus also affects the micro-circulation, namely small vessels and capillaries, favoring the occurrence of DD. In particular, individuals with T2DM, obesity, and/or MS, who have an altered response of the innate immunity with an increased pro-inflammatory state, exhibit higher susceptibility to developing more severe injuries, a state which is conducive to enhanced severity of SARS-CoV-2 infection.
https://pubmed.ncbi.nlm.nih.gov/35414837/
The important contribution on the vascular function of previous chronic therapies (antihypertensive and anti-inflammatory drugs) but especially of drugs used to treat the acute infection (antiviral drugs, corticotherapy, immunomodulators, antibiotics, invasive respiration) should not be overlooked and has been debated in multiple studies.
https://pubmed.ncbi.nlm.nih.gov/35414837/
https://onlinelibrary.wiley.com/doi/full/10.1111/joim.13275
Many studies have raised the issue that even in young, previously healthy individuals, vascular function would need a longer time to recover and, in some cases, it would never return to normal.
https://pubmed.ncbi.nlm.nih.gov/35439042/
https://pubmed.ncbi.nlm.nih.gov/35345906/
This possibly explains some of the pathophysiological pathways responsible for the development of the post-acute and long COVID-19 syndromes.
The study team documented the increased prevalence of impaired AS, as assessed by PWV, but also of reduced AoD and AoS with an increased AoSI, which is associated with various patterns of DD in 121 female patients who developed post-acute COVID-19 syndrome during the recovery from a SARS-CoV-2 infection.
Though the study team conducted this study on younger women (under 55 years old), all of whom were selected for being premenopausal and who considered themselves to be apparently healthy prior to this infection, at a careful examination, the study team realized that 67 of them fulfilled the diagnostic criteria for MS.
The study team proceeded further to compare their results with those obtained in subjects without MS and with those obtained from an age-matched control group of healthy women who did not suffer from COVID-19 and in whom, as expected, the parameters characterizing AS, aortic elasticity, and DD were within normal limits.
Shockingly, PWV values, although significantly higher (p = 0.0148) in patients with post-COVID-19 syndrome and MS (group A) compared to those without MS (group B), were much more elevated than in individuals who did not suffer from this illness (p < 0.0001).
Similar results were also observed for the parameters characterizing aortic elasticity and DD (p < 0.0001). It is worth mentioning that by analyzing the existence of associations between these indexes and other factors characterizing SARS-CoV-2 infection, the study team observed the most powerful correlations with the time elapsed since the acute illness and its severity.
Similar results suggest that, in a shorter time period, the severity of the COVID-19 illness and the time elapsed since the infection are more important predictors for the magnitude of AS, impaired aortic elasticity, and DD than the generic determinants of MS.
Interestingly, the multivariate linear regression analysis employed in the study also revealed that the elements characterizing the acute illness were more powerful predictors for the development of AS, impaired aortic elasticity, and DD, followed by the TYG index as a surrogate for IR.
There is a high probability that, especially in people with metabolic dysfunctions, such as MS, T2DM, and/or obesity, several of the pathophysiological mechanisms involved in vascular aging and degeneration that were exacerbated from COVID-19 infection could persist, leading to further cardiovascular injuries.
The study team concluded that altered AS, reduced aortic elasticity, and DD are common findings among patients with post-COVID-19 syndrome, especially in those with a poor metabolic profile. The indexes characterizing those dysfunctions were significantly associated with the time elapsed since the acute illness and its severity, which was expressed by CRP levels and the extent of the lung injury, as well as by biological markers characterizing MS. Arterial and aortic stiffness, the initial levels of CRP, and the TyG index were identified by multivariate regression analysis as significant predictor factors for the development of DD.
For the latest
Long COVID News keep on logging to Thailand Medical News.
